Abstract
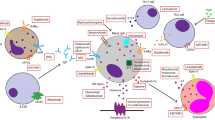
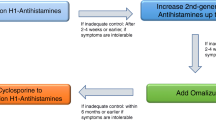
Chronic spontaneous urticaria (CSU) is characterized by recurring wheals that last 6 weeks or longer without an identifiable cause. The estimated point prevalence of CSU worldwide is 1%. Furthermore, it has a significant impact on quality of life in both adults and pediatric patients and their families. Although it is most often a self-limited disease, some patients have urticaria refractory to first-line treatment: second-generation H1 antihistamines. In these patients, the use of targeted monoclonal antibodies is necessary. While omalizumab is the only Food and Drug Administration-approved monoclonal antibody for CSU, others, including ligelizumab, dupilumab, benralizumab, and several orally administered Bruton’s tyrosine kinase inhibitors, are also promising therapeutics for reducing the morbidity of CSU. Novel therapies, among others discussed here, are rapidly being developed with new trials and therapeutics being released nearly monthly. Thus, we performed a scoping literature review of randomized controlled trials studying targeted therapies for CSU. We also discuss the pathophysiology, diagnosis, prognosis, and future research directions in CSU.
Similar content being viewed by others
References
Maurer M, Abuzakouk M, Bérard F et al (2017) The burden of chronic spontaneous urticaria is substantial: real-world evidence from ASSURE-CSU. Allergy 72(12):2005–2016. https://doi.org/10.1111/all.13209
Fricke J, Ávila G, Keller T et al (2020) Prevalence of chronic urticaria in children and adults across the globe: systematic review with meta-analysis. Allergy 75(2):423–432. https://doi.org/10.1111/all.14037
Balp MM, Weller K, Carboni V et al (2018) Prevalence and clinical characteristics of chronic spontaneous urticaria in pediatric patients. Pediatr Allergy Immunol 29(6):630–636. https://doi.org/10.1111/pai.12910
Saini SS, Kaplan AP (2018) Chronic spontaneous urticaria: the devil’s itch. J Allergy Clin Immunol Pract 6(4):1097–1106. https://doi.org/10.1016/j.jaip.2018.04.013
Sharma VK, Gupta V, Pathak M, Ramam M (2017) An open-label prospective clinical study to assess the efficacy of increasing levocetirizine dose up to four times in chronic spontaneous urticaria not controlled with standard dose. J Dermatol Treat 28(6):539–543. https://doi.org/10.1080/09546634.2016.1246705
Sotés PI, Armisén M, Usero-Bárcena T et al (2021) Efficacy and safety of up-dosing antihistamines in chronic spontaneous urticaria: a systematic review of the literature. J Investig Allergol Clin Immunol 31(4):282–291. https://doi.org/10.18176/jiaci.0649
Zuberbier T, Abdul Latiff AH, Abuzakouk M et al (2022) The international EAACI/GA2LEN/EuroGuiDerm/APAAACI guideline for the definition, classification, diagnosis, and management of urticaria. Allergy 77(3):734–766. https://doi.org/10.1111/all.15090
Bernstein JA, Lang DM, Khan DA et al (2014) The diagnosis and management of acute and chronic urticaria: 2014 update. J Allergy Clin Immunol 133(5):1270–1277. https://doi.org/10.1016/j.jaci.2014.02.036
Endo T, Toyoshima S, Kanegae K et al (2019) Identification of biomarkers for predicting the response to cyclosporine A therapy in patients with chronic spontaneous urticaria. Allergol Int 68(2):270–273. https://doi.org/10.1016/j.alit.2018.09.006
Sussman G, Hébert J, Gulliver W et al (2020) Omalizumab re-treatment and step-up in patients with chronic spontaneous urticaria: OPTIMA trial. J Allergy Clin Immunol Pract 8(7):2372-2378.e5. https://doi.org/10.1016/j.jaip.2020.03.022
Kaplan A, Ledford D, Ashby M et al (2013) Omalizumab in patients with symptomatic chronic idiopathic/spontaneous urticaria despite standard combination therapy. J Allergy Clin Immunol 132(1):101–109. https://doi.org/10.1016/j.jaci.2013.05.013
Nettis E, Cegolon L, Di Leo E et al (2018) Omalizumab in chronic spontaneous urticaria: efficacy, safety, predictors of treatment outcome, and time to response. Ann Allergy Asthma Immunol 121(4):474–478. https://doi.org/10.1016/j.anai.2018.06.014
Bernstein JA, Kavati A, Tharp MD et al (2018) Effectiveness of omalizumab in adolescent and adult patients with chronic idiopathic/spontaneous urticaria: a systematic review of “real-world” evidence. Expert Opin Biol Ther 18(4):425–448. https://doi.org/10.1080/14712598.2018.1438406
Bernstein JA, Singh U, Rao MB, Berendts K, Zhang X, Mutasim D (2020) Benralizumab for chronic spontaneous urticaria. N Engl J Med 383(14):1389–1391. https://doi.org/10.1056/NEJMc2016395
Kaul M, End P, Cabanski M et al (2021) Remibrutinib (LOU064): a selective potent oral BTK inhibitor with promising clinical safety and pharmacodynamics in a randomized phase I trial. Clin Transl Sci. https://doi.org/10.1111/cts.13005
Giménez-Arnau AM, Salman A (2020) Targeted therapy for chronic spontaneous urticaria: rationale and recent progress. Drugs 80(16):1617–1634. https://doi.org/10.1007/s40265-020-01387-9
Kocatürk E, Maurer M, Metz M, Grattan C (2017) Looking forward to new targeted treatments for chronic spontaneous urticaria. Clin Transl Allergy 7:1. https://doi.org/10.1186/s13601-016-0139-2
Altrichter S, Frischbutter S, Fok JS et al (2020) The role of eosinophils in chronic spontaneous urticaria. J Allergy Clin Immunol 145(6):1510–1516. https://doi.org/10.1016/j.jaci.2020.03.005
Bansal CJ, Bansal AS (2019) Stress, pseudoallergens, autoimmunity, infection and inflammation in chronic spontaneous urticaria. Allergy Asthma Clin Immunol 15:56. https://doi.org/10.1186/s13223-019-0372-z
Bracken SJ, Abraham S, MacLeod AS (2019) Autoimmune theories of chronic spontaneous urticaria. Front Immunol 10:627. https://doi.org/10.3389/fimmu.2019.00627
Hide M, Francis DM, Grattan CE, Hakimi J, Kochan JP, Greaves MW (1993) Autoantibodies against the high-affinity IgE receptor as a cause of histamine release in chronic urticaria. N Engl J Med 328(22):1599–1604. https://doi.org/10.1056/NEJM199306033282204
Grattan CE, Francis DM, Hide M, Greaves MW (1991) Detection of circulating histamine releasing autoantibodies with functional properties of anti-IgE in chronic urticaria. Clin Exp Allergy 21(6):695–704. https://doi.org/10.1111/j.1365-2222.1991.tb03198.x
Dabija D, Tadi P (2021) Chronic urticaria. In: StatPearls. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/pubmed/32310370. Accessed 29 Sept 2021
Gu H, Li L, Gu M, Zhang G (2015) Association between Helicobacter pylori infection and chronic urticaria: a meta-analysis. Gastroenterol Res Pract 2015:486974. https://doi.org/10.1155/2015/486974
Gaig P, García-Ortega P, Enrique E, Papo M, Quer JC, Richard C (2002) Efficacy of the eradication of Helicobacter pylori infection in patients with chronic urticaria. A placebo-controlled double blind study. Allergol Immunopathol 30(5):255–258. https://doi.org/10.1016/s0301-0546(02)79133-7
Yanase Y, Takahagi S, Hide M (2018) Chronic spontaneous urticaria and the extrinsic coagulation system. Allergol Int 67(2):191–194. https://doi.org/10.1016/j.alit.2017.09.003
Cugno M, Marzano AV, Tedeschi A, Fanoni D, Venegoni L, Asero R (2009) Expression of tissue factor by eosinophils in patients with chronic urticaria. Int Arch Allergy Immunol 148(2):170–174. https://doi.org/10.1159/000155748
Ying S, Kikuchi Y, Meng Q, Kay AB, Kaplan AP (2002) TH1/TH2 cytokines and inflammatory cells in skin biopsy specimens from patients with chronic idiopathic urticaria: comparison with the allergen-induced late-phase cutaneous reaction. J Allergy Clin Immunol 109(4):694–700. https://doi.org/10.1067/mai.2002.123236
Toyoda M, Maruyama T, Morohashi M, Bhawan J (1996) Free eosinophil granules in urticaria: a correlation with the duration of wheals. Am J Dermatopathol 18(1):49–57. https://doi.org/10.1097/00000372-199602000-00008
Hon KL, Leung AKC, Ng WGG, Loo SK (2019) Chronic urticaria: an overview of treatment and recent patents. Recent Pat Inflamm Allergy Drug Discov 13(1):27–37. https://doi.org/10.2174/1872213X13666190328164931
Church MK, Maurer M, Simons FER et al (2010) Risk of first-generation H(1)-antihistamines: a GA(2)LEN position paper. Allergy 65(4):459–466. https://doi.org/10.1111/j.1398-9995.2009.02325.x
Phinyo P, Koompawichit P, Nochaiwong S, Tovanabutra N, Chiewchanvit S, Chuamanochan M (2021) Comparative efficacy and acceptability of licensed dose second-generation antihistamines in chronic spontaneous urticaria: a network meta-analysis. J Allergy Clin Immunol Pract 9(2):956-970.e57. https://doi.org/10.1016/j.jaip.2020.08.055
Staevska M, Popov TA, Kralimarkova T et al (2010) The effectiveness of levocetirizine and desloratadine in up to 4 times conventional doses in difficult-to-treat urticaria. J Allergy Clin Immunol 125(3):676–682. https://doi.org/10.1016/j.jaci.2009.11.047
Siebenhaar F, Degener F, Zuberbier T, Martus P, Maurer M (2009) High-dose desloratadine decreases wheal volume and improves cold provocation thresholds compared with standard-dose treatment in patients with acquired cold urticaria: a randomized, placebo-controlled, crossover study. J Allergy Clin Immunol 123(3):672–679. https://doi.org/10.1016/j.jaci.2008.12.008
van den Elzen MT, van Os-Medendorp H, van den Brink I et al (2017) Effectiveness and safety of antihistamines up to fourfold or higher in treatment of chronic spontaneous urticaria. Clin Transl Allergy. https://doi.org/10.1186/s13601-017-0141-3
Nayak AS, Berger WE, LaForce CF et al (2017) Randomized, placebo-controlled study of cetirizine and loratadine in children with seasonal allergic rhinitis. Allergy Asthma Proc 38(3):222–230. https://doi.org/10.2500/aap.2017.38.4050
Gupta S, Khalilieh S, Kantesaria B, Banfield C (2007) Pharmacokinetics of desloratadine in children between 2 and 11 years of age. Br J Clin Pharmacol 63(5):534–540. https://doi.org/10.1111/j.1365-2125.2006.02810.x
Meltzer EO, Scheinmann P, Rosado Pinto JE et al (2004) Safety and efficacy of oral fexofenadine in children with seasonal allergic rhinitis—a pooled analysis of three studies. Pediatr Allergy Immunol 15(3):253–260. https://doi.org/10.1111/j.1399-3038.2004.00167.x
Pampura AN, Papadopoulos NG, Spičák V, Kurzawa R (2011) Evidence for clinical safety, efficacy, and parent and physician perceptions of levocetirizine for the treatment of children with allergic disease. Int Arch Allergy Immunol 155(4):367–378. https://doi.org/10.1159/000321181
Novák Z, Yáñez A, Kiss I et al (2016) Safety and tolerability of bilastine 10 mg administered for 12 weeks in children with allergic diseases. Pediatr Allergy Immunol 27(5):493–498. https://doi.org/10.1111/pai.12555
Vena GA, Cassano N, Colombo D, Peruzzi E, Pigatto P, Neo-I-30 Study Group (2006) Cyclosporine in chronic idiopathic urticaria: a double-blind, randomized, placebo-controlled trial. J Am Acad Dermatol 55(4):705–709. https://doi.org/10.1016/j.jaad.2006.04.078
Deacock SJ (2008) An approach to the patient with urticaria. Clin Exp Immunol 153(2):151–161. https://doi.org/10.1111/j.1365-2249.2008.03693.x
Agache I, Rocha C, Pereira A et al (2021) Efficacy and safety of treatment with omalizumab for chronic spontaneous urticaria: a systematic review for the EAACI Biologicals Guidelines. Allergy 76(1):59–70. https://doi.org/10.1111/all.14547
Saini SS, Bindslev-Jensen C, Maurer M et al (2015) Efficacy and safety of omalizumab in patients with chronic idiopathic/spontaneous urticaria who remain symptomatic on H1 antihistamines: a randomized, placebo-controlled study. J Invest Dermatol 135(3):925. https://doi.org/10.1038/jid.2014.512
Maurer M, Rosén K, Hsieh HJ et al (2013) Omalizumab for the treatment of chronic idiopathic or spontaneous urticaria. N Engl J Med 368(10):924–935. https://doi.org/10.1056/NEJMoa1215372
Maurer M, Kaplan A, Rosén K et al (2018) The XTEND-CIU study: long-term use of omalizumab in chronic idiopathic urticaria. J Allergy Clin Immunol 141(3):1138-1139.e7. https://doi.org/10.1016/j.jaci.2017.10.018
Ocak M, Soyer O, Buyuktiryaki B, Sekerel BE, Sahiner UM (2020) Omalizumab treatment in adolescents with chronic spontaneous urticaria: efficacy and safety. Allergol Immunopathol 48(4):368–373. https://doi.org/10.1016/j.aller.2020.03.011
Arm JP, Bottoli I, Skerjanec A et al (2014) Pharmacokinetics, pharmacodynamics and safety of QGE031 (ligelizumab), a novel high-affinity anti-IgE antibody, in atopic subjects. Clin Exp Allergy 44(11):1371–1385. https://doi.org/10.1111/cea.12400
Maurer M, Giménez-Arnau AM, Sussman G et al (2019) Ligelizumab for chronic spontaneous urticaria. N Engl J Med 381(14):1321–1332. https://doi.org/10.1056/NEJMoa1900408
Maurer M, Giménez-Arnau A, Bernstein JA et al (2022) Sustained safety and efficacy of ligelizumab in patients with chronic spontaneous urticaria: a one-year extension study. Allergy. https://doi.org/10.1111/all.15175
Harb H, Chatila TA (2020) Mechanisms of dupilumab. Clin Exp Allergy 50(1):5–14. https://doi.org/10.1111/cea.13491
Lee JK, Simpson RS (2019) Dupilumab as a novel therapy for difficult to treat chronic spontaneous urticaria. J Allergy Clin Immunol Pract 7(5):1659-1661.e1. https://doi.org/10.1016/j.jaip.2018.11.018
Goodman B, Jariwala S (2021) Dupilumab as a novel therapy to treat adrenergic urticaria. Ann Allergy Asthma Immunol 126(2):205–206. https://doi.org/10.1016/j.anai.2020.06.034
Hom S, Pisano M (2017) Reslizumab (Cinqair): an interleukin-5 antagonist for severe asthma of the eosinophilic phenotype. P T 42(9):564–568. https://www.ncbi.nlm.nih.gov/pubmed/28890642. Accessed 29 Sept 2021
Wechsler ME, Akuthota P, Jayne D et al (2017) Mepolizumab or placebo for eosinophilic granulomatosis with polyangiitis. N Engl J Med 376(20):1921–1932. https://doi.org/10.1056/NEJMoa1702079
Ortega HG, Liu MC, Pavord ID et al (2014) Mepolizumab treatment in patients with severe eosinophilic asthma. N Engl J Med 371(13):1198–1207. https://doi.org/10.1056/NEJMoa1403290
Roufosse F, Kahn JE, Rothenberg ME et al (2020) Efficacy and safety of mepolizumab in hypereosinophilic syndrome: a phase III, randomized, placebo-controlled trial. J Allergy Clin Immunol 146(6):1397–1405. https://doi.org/10.1016/j.jaci.2020.08.037
Hata D, Kawakami Y, Inagaki N et al (1998) Involvement of Bruton’s tyrosine kinase in FcεRI-dependent mast cell degranulation and cytokine production. J Exp Med 187(8):1235–1247. https://doi.org/10.1084/jem.187.8.1235
Maurer M, Berger W, Giménez-Arnau A et al (2021) Remibrutinib (LOU064) versus placebo in patients with chronic spontaneous urticaria: a randomised, double-blind, phase 2b dose-finding study. SSRN Electron J. https://doi.org/10.2139/ssrn.3968885
Metz M, Sussman G, Gagnon R et al (2021) Fenebrutinib in H antihistamine-refractory chronic spontaneous urticaria: a randomized phase 2 trial. Nat Med 27(11):1961–1969. https://doi.org/10.1038/s41591-021-01537-w
Hermes B, Prochazka AK, Haas N, Jurgovsky K, Sticherling M, Henz BM (1999) Upregulation of TNF-alpha and IL-3 expression in lesional and uninvolved skin in different types of urticaria. J Allergy Clin Immunol 103(2 Pt 1):307–314. https://doi.org/10.1016/s0091-6749(99)70506-3
Wilson LH, Eliason MJ, Leiferman KM, Hull CM, Powell DL (2011) Treatment of refractory chronic urticaria with tumor necrosis factor–alfa inhibitors. J Am Acad Dermatol 64(6):1221–1222. https://doi.org/10.1016/j.jaad.2009.10.043
Silverman GJ, Weisman S (2003) Rituximab therapy and autoimmune disorders: prospects for anti-B cell therapy. Arthritis Rheum 48(6):1484–1492. https://doi.org/10.1002/art.10947
Combalia A, Losno RA, Prieto-González S, Mascaró JM (2018) Rituximab in refractory chronic spontaneous urticaria: an encouraging therapeutic approach. Skin Pharmacol Physiol 31(4):184–187. https://doi.org/10.1159/000487402
Kitajima M, Lee HC, Nakayama T, Ziegler SF (2011) TSLP enhances the function of helper type 2 cells. Eur J Immunol 41(7):1862–1871. https://doi.org/10.1002/eji.201041195
Shikotra A, Choy DF, Ohri CM et al (2012) Increased expression of immunoreactive thymic stromal lymphopoietin in patients with severe asthma. J Allergy Clin Immunol 129(1):104-111.e1−e9. https://doi.org/10.1016/j.jaci.2011.08.031
Sabag DA, Matanes L, Bejar J et al (2020) Interleukin-17 is a potential player and treatment target in severe chronic spontaneous urticaria. Clin Exp Allergy 50(7):799–804. https://doi.org/10.1111/cea.13616
Ferrer M, Giménez-Arnau A, Saldana D et al (2018) Predicting chronic spontaneous urticaria symptom return after omalizumab treatment discontinuation: exploratory analysis. J Allergy Clin Immunol Pract 6(4):1191-1197.e5. https://doi.org/10.1016/j.jaip.2018.04.003
Ertas R, Ozyurt K, Atasoy M, Hawro T, Maurer M (2018) The clinical response to omalizumab in chronic spontaneous urticaria patients is linked to and predicted by IgE levels and their change. Allergy 73(3):705–712. https://doi.org/10.1111/all.13345
Ertas R, Ozyurt K, Ozlu E et al (2017) Increased IgE levels are linked to faster relapse in patients with omalizumab-discontinued chronic spontaneous urticaria. J Allergy Clin Immunol 140(6):1749–1751. https://doi.org/10.1016/j.jaci.2017.08.007
Deza G, Bertolín-Colilla M, Sánchez S et al (2018) Basophil FcɛRI expression is linked to time to omalizumab response in chronic spontaneous urticaria. J Allergy Clin Immunol 141(6):2313-2316.e1. https://doi.org/10.1016/j.jaci.2018.02.021
Grieco T, Dies L, Sernicola A et al (2020) Potential clinical and serological predictors of chronic spontaneous urticaria relapse in patients under omalizumab treatment. Immunotherapy 12(16):1173–1181. https://doi.org/10.2217/imt-2020-0088
Flanagin A, Frey T, Christiansen SL, AMA Manual of Style Committee (2021) Updated guidance on the reporting of race and ethnicity in medical and science journals. JAMA 326(7):621–627. https://doi.org/10.1001/jama.2021.13304
Warren RB, Marsden A, Tomenson B et al (2019) Identifying demographic, social and clinical predictors of biologic therapy effectiveness in psoriasis: a multicentre longitudinal cohort study. Br J Dermatol 180(5):1069–1076. https://doi.org/10.1111/bjd.16776
Szefler SJ, Jerschow E, Yoo B et al (2021) Response to omalizumab in black and white patients with allergic asthma. J Allergy Clin Immunol Pract 9(11):4021–4028. https://doi.org/10.1016/j.jaip.2021.07.013
Funding
Dr. Greiner is supported by training grant T32 AI155385 from the U.S. National Institutes of Health. The funder had no role in the design of the study, the collection and analysis of the data, or the preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Greiner, B., Nicks, S., Adame, M. et al. Pathophysiology, Diagnosis, and Management of Chronic Spontaneous Urticaria: A Literature Review. Clinic Rev Allerg Immunol 63, 381–389 (2022). https://doi.org/10.1007/s12016-022-08952-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-022-08952-y