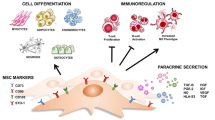
Abstract
Mesenchymal stem/stromal cells (MSCs) are remarkable tools for regenerative medicine. Therapeutic approaches using these cells can promote increased activity and viability in several cell types through diverse mechanisms such as paracrine and immunomodulatory activities, contributing substantially to tissue regeneration and functional recovery. However, biological samples of human MSCs, usually obtained from adult tissues, often exhibit variable behavior during in vitro culture, especially with respect to cell population heterogeneity, replicative senescence, and consequent loss of functionality. Accordingly, it is necessary to establish standard protocols to generate high-quality, stable cell cultures, for example, by using pluripotent stem cells (PSCs) in derivation protocols of MSC-like cells since PSCs maintain their characteristics consistently during culture. However, the available protocols seem to generate distinct populations of PSC-derivedMSCs (PSC-MSCs) with peculiar attributes, which do not always resemble bona fide primary MSCs. The present review addresses the developmental basis behind some of these derivation protocols, exposing the differences among them and discussing the functional properties of PSC-MSCs, shedding light on elements that may help determine standard characterizations and criteria to evaluate and define these cells.
Graphic Abstract





Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Viswanathan, S., Shi, Y., Galipeau, J., Krampera, M., Leblanc, K., Martin, I., & Sensebe, L. (2019). Mesenchymal stem versus stromal cells: International Society for Cell & Gene Therapy (ISCT®) Mesenchymal Stromal Cell committee position statement on nomenclature. Cytotherapy, 21(10), 1019–1024
Friedenstein, A. J., Piatetzky-Shapiro, I. I., & Petrakova, K. V. (1966). Osteogenesis in transplants of bone marrow cells. Journal of Embryology and Experimental Morphology, 16(3), 381–390
Castro-Malaspina, H., Gay, R. E., Resnick, G., Kapoor, N., Meyers, P., Chiarieri, D., & Moore, M. A. S. (1980). Characterization of human bone marrow fibroblast colony-forming cells (CFU-F) and their progeny. Blood, 56(2), 289–301
da Silva Meirelles, L., Chagastelles, P. C., & Nardi, N. B. (2006). Mesenchymal stem cells reside in virtually all post-natal organs and tissues. Journal of Cell Science, 119(11), 2204–2213
Dominici, M., Le Blanc, K., Mueller, I., Slaper-Cortenbach, I., Marini, F. C., Krause, D. S., & Horwitz, E. M. (2006). Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy, 8(4), 315–317
Bourin, P., Bunnell, B. A., Casteilla, L., Dominici, M., Katz, A. J., March, K. L., & Gimble, J. M. (2013). Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/ stem cells: a joint statement of the International Federation for Adipose Therapeutics (IFATS) and Science and the International S. Cytotherapy, 15(6), 641–648
Angulski, A. B. B., Capriglione, L. G., Batista, M., Marcon, B. H., Senegaglia, A. C., Stimamiglio, M. A., & Correa, A. (2017). The protein content of extracellular vesicles derived from expanded human umbilical cord blood-derived CD133 + and human bone marrow-derived mesenchymal stem cells partially explains why both sources are advantageous for regenerative medicine. Stem Cell Reviews and Reports, 13(2), 244–257
Uccelli, A., Mancardi, G., & Chiesa, S. (2008). Is there a role for mesenchymal stem cells in autoimmune diseases? Autoimmunity, 41(8), 592–595
Phinney, D. G. (2012). Functional heterogeneity of mesenchymal stem cells: implications for cell therapy. Journal of Cellular Biochemistry, 113(9), 2806–2812
Robert, A. W., Marcon, B. H., Dallagiovanna, B., & Shigunov, P. (2020). Adipogenesis, osteogenesis, and chondrogenesis of human mesenchymal stem/stromal cells: a comparative transcriptome approach. Frontiers in Cell and Developmental Biology, 8(561), 1–52
Horinouchi, C. D. S., Barisón, M. J., Robert, A. W., Kuligovski, C., Aguiar, A. M., & Dallagiovanna, B. (2020). Influence of donor age on the differentiation and division capacity of human adipose-derived stem cells. World Journal of Stem Cells, 12(12), 1640–1651
Russell, K. C., Phinney, D. G., Lacey, M. R., Barrilleaux, B. L., Meyertholen, K. E., & O’Connor, K. C. (2010). In vitro high-capacity assay to quantify the clonal heterogeneity in trilineage potential of mesenchymal stem cells reveals a complex hierarchy of lineage commitment. Stem Cells, 28(4), 788–798
Russell, K. C., Lacey, M. R., Gilliam, J. K., Tucker, H. A., Phinney, D. G., & O’Connor, K. C. (2011). Clonal analysis of the proliferation potential of human bone marrow mesenchymal stem cells as a function of potency. Biotechnology and Bioengineering, 108(11), 2716–2726
Galipeau, J. (2013). The mesenchymal stromal cells dilemma-does a negative phase III trial of random donor mesenchymal stromal cells in steroid-resistant graft-versus-host disease represent a death knell or a bump in the road? Cytotherapy, 15(1), 2–8
Thomson, J. A., Itskovitz-Eldor, J., Shapiro, S. S., Waknitz, M. A., Swiergiel, Jennifer, J., Marshall, V. S., & Jones, J. M. (1998). Embryonic stem cell lines derived from human blastocysts. Science, 282(5391), 1145–1147
Xu, C., Jiang, J., Sottile, V., McWhir, J., Lebrowski, J., & Carpenter, M. K. (2004). Immortalized fibroblast-Like cells derived from human embryonic stem cells support undifferentiated cell growth. Stem Cells, 22(1), 972–980
Hwang, N. S., Varghese, S., Zhang, Z., & Elisseeff, J. (2006). Chondrogenic differentiation of human embryonic stem cell-derived cells in arginine-glycine-aspartate modified hydrogels. Tissue Engineering, 12(9), 2695–2706
Hwang, N. S., Varghese, S., Lee, H. J., Zhang, Z., Ye, Z., Bae, J., & Elisseeff, J. (2008). In vivo commitment and functional tissue regeneration using human embryonic stem cell-derived mesenchymal cells. Proceedings of the National Academy of Sciences of the United States of America, 105(52), 20641–20646
Olivier, E. N., Rybicki, A. C., & Bouhassira, E. E. (2006). Differentiation of human embryonic stem cells into bipotent mesenchymal stem cells. Stem Cells, 24(8), 1914–1922
Yen, B. L., Chang, C. J., Liu, K. J., Chen, Y. C., Hu, H. I., Bai, C. H., & Yen, M. L. (2009). Brief report-human embryonic stem cell-derived mesenchymal progenitors possess strong immunosuppressive effects toward natural killer cells as well as T lymphocytes. Stem Cells, 27(2), 451–456
Moslem, M., Valojerdi, M. R., Pournasr, B., Muhammadnejad, A., & Baharvand, H. (2013). Therapeutic potential of human induced pluripotent stem cell-derived mesenchymal stem cells in mice with lethal fulminant hepatic failure. Cell Transplantation, 22(10), 1785–1799
Trivedi, P., & Hematti, P. (2008). Derivation and immunological characterization of mesenchymal stromal cells from human embryonic stem cells. Experimental Hematology, 36(3), 350–359
Arpornmaeklong, P., Brown, S. E., Wang, Z., & Krebsbach, P. H. (2009). Phenotypic characterization, osteoblastic differentiation, and bone regeneration capacity of human embryonic stem cell-derived mesenchymal stem cells. Stem Cells and Development, 18(7), 955–968
Villa-Diaz, L. G., Brown, S. E., Liu, Y., Ross, A., Lahann, J., Parent, J. M., & Krebsbach, P. H. (2012). Derivation of functional mesenchymal stem cells from human induced pluripotent stem cells culture on synthetic polymer substrates. Stem Cells, 30(6), 1174–1181
Brown, S. E., Tong, W., & Krebsbach, P. H. (2009). The derivation of mesenchymal stem cells from human embryonic stem cells. Cells, Tissues, Organs, 189(1–4), 256–260
Karlsson, C., Emanuelsson, K., Wessberg, F., Kajic, K., Axell, M. Z., Eriksson, P. S., & Strehl, R. (2009). Human embryonic stem cell-derived mesenchymal progenitors-Potential in regenerative medicine. Stem Cell Research, 3(1), 39–50
De Peppo, G. M., Svensson, S., Lennerås, M., Synnergren, J., Stenberg, J., Strehl, R., & Karlsson, C. (2010). Human embryonic mesodermal progenitors highly resemble human mesenchymal stem cells and display high potential for tissue engineering applications. Tissue Engineering - Part A, 16(7), 2161–2182
De Peppo, G. M., Sjovall, P., Lennerås, M., Strehl, R., Hyllner, J., Thomsen, P., & Karlsson, C. (2010). Osteogenic potential of human mesenchymal stem cells and human embryonic stem cell-derived mesodermal progenitors: a tissue engineering perspective. Tissue Engineering - Part A, 16(11), 3413–3426
De Peppo, G. M., Sladkova, M., Sjövall, P., Palmquist, A., Oudina, K., Hyllner, J., & Karlsson, C. (2013). Human embryonic stem cell-derived mesodermal progenitors display substantially increased tissue formation compared to human mesenchymal stem cells under dynamic culture conditions in a packed Bed/Column bioreactor. Tissue Engineering - Part A, 19(1–2), 175–187
Li, O., Tormin, A., Sundberg, B., Hyllner, J., Le Blanc, K., & Scheding, S. (2013). Human embryonic stem cell-derived mesenchymal stroma cells (hES-MSCs) engraft in vivo and support hematopoiesis without suppressing immune function: implications for off-the shelf ES-MSC therapies. PLoS One1, 8(1), 1–9
Stavropoulos, M. E., Mengarelli, I., & Barberi, T. (2009). Differentiation of multipotent mesenchymal precursors and skeletal myoblasts from human embryonic stem cells. Current Protocols in Stem Cell Biology, Chap, 1(Supplement 9), 1–10
Hu, J., Smith, L. A., Feng, K., Liu, X., Sun, H., & Ma, P. X. (2010). Response of human embryonic stem cell-derived mesenchymal stem cells to osteogenic factors and architectures of materials during in vitro osteogenesis. Tissue Engineering - Part A, 16(11), 3507–3514
Fu, X., Chen, Y., Xie, F. N., Dong, P., Liu, W. B., Cao, Y., & Xiao, R. (2015). Comparison of immunological characteristics of mesenchymal stem cells derived from human embryonic stem cells and bone marrow. Tissue Engineering - Part A, 21(3–4), 616–626
Barbet, R., Peiffer, I., Hatzfeld, A., Charbord, P., & Hatzfeld, J. A. (2011). Comparison of gene expression in human embryonic stem cells, hESC-derived mesenchymal stem cells and human mesenchymal stem cells. Stem Cells International, 2011(1), 1–9
Giuliani, M., Oudrhiri, N., Noman, Z. M., Vernochet, A., Chouaib, S., Azzarone, B., & Bennaceur-Griscelli, A. (2011). Human mesenchymal stem cells derived from induced pluripotent stem cells down-regulate NK-cell cytolytic machinery. Blood, 118(12), 3254–3262
Gruenloh, W., Kambal, A., Sondergaard, C., McGee, J., Nacey, C., Kalomoiris, S., & Nolta, J. A. (2011). Characterization and in vivo testing of mesenchymal stem cells derived from human embryonic stem cells. Tissue Engineering - Part A, 17(11–12), 1517–1525
Harkness, L., Mahmood, A., Ditzel, N., Abdallah, B. M., Nygaard, J. V., & Kassem, M. (2011). Selective isolation and differentiation of a stromal population of human embryonic stem cells with osteogenic potential. Bone, 48(2), 231–241
Ahfeldt, T., Schinzel, R. T., Lee, Y. K., Hendrickson, D., Kaplan, A., Lum, D. H., & Cowan, C. A. (2012). Programming human pluripotent stem cells into white and brown adipocytes. Nature Cell Biology, 14(2), 209–219
Liu, Y., Goldberg, A. J., Dennis, J. E., Gronowicz, G. A., & Kuhn, L. T. (2012). One-step derivation of mesenchymal stem cell (MSC)-like cells from human pluripotent stem cells on a fibrillar collagen coating. PLoS One1, 7(3), 1–9
Yen, M. L., Hou, C. H., Peng, K. Y., Tseng, P. C., Jiang, S. S., Shun, C. T., & Kuo, M. L. (2011). Efficient derivation and concise gene expression profiling of human embryonic stem cell-derived mesenchymal progenitors (EMPs). Cell Transplantation, 20(10), 1529–1545
Zou, L., Luo, Y., Chen, M., Wang, G., Ding, M., Petersen, C. C., & Bünger, C. (2013). A simple method for deriving functional MSCs and applied for osteogenesis in 3D scaffolds. Scientific Reports, 3(2243), 1–10
Xia, Y., Ling, X., Hu, G., Zhu, Q., Zhang, J., Li, Q., & Deng, Z. (2020). Small extracellular vesicles secreted by human iPSC-derived MSC enhance angiogenesis through inhibiting STAT3-dependent autophagy in ischemic stroke. Stem Cell Research and Therapy, 11(313), 1–17
Kang, R., Luo, Y., Zou, L., Xie, L., Lysdahl, H., Jiang, X., & Bünger, C. (2014). Osteogenesis of human induced pluripotent stem cells derived mesenchymal stem cells on hydroxyapatite contained nanofibers. RSC Advances, 4(11), 5734–5739
Kang, R., Zhou, Y., Tan, S., Zhou, G., Aagaard, L., Xie, L., & Luo, Y. (2015). Mesenchymal stem cells derived from human induced pluripotent stem cells retain adequate osteogenicity and chondrogenicity but less adipogenicity. Stem Cell Research and Therapy, 6(144), 1–14
Peng, K. Y., Lee, Y. W., Hsu, P. J., Wang, H. H., Wang, Y., Liou, J. Y., & Yen, B. L. (2016). Human pluripotent stem cell (PSC)-derived mesenchymal stem cells (MSCs) show potent neurogenic capacity which is enhanced with cytoskeletal rearrangement. Oncotarget, 7(28), 43949–43959
Wang, L. T., Jiang, S. S., Ting, C. H., Hsu, P. J., Chang, C. C., Sytwu, H. K., & Yen, B. L. (2018). Differentiation of mesenchymal stem cells from human induced pluripotent stem cells results in downregulation of c-Myc and DNA replication pathways with immunomodulation toward CD4 and CD8 cells. Stem Cells, 36(1), 903–914
Zhou, Y., Liao, J., Fang, C., Mo, C., Zhou, G., & Luo, Y. (2018). One-step derivation of functional mesenchymal stem cells from human pluripotent stem cells. Bio-Protocol, 8(22), 1–21
Luo, L., Zhou, Y., Zhang, C., Huang, J., Du, J., Liao, J., & Luo, Y. (2020). Feeder-free generation and transcriptome characterization of functional mesenchymal stromal cells from human pluripotent stem cells. Stem Cell Research, 48(1), 1–13
Zhang, J., Guan, J., Niu, X., Hu, G., Guo, S., Li, Q., & Wang, Y. (2015). Exosomes released from human induced pluripotent stem cells-derived MSCs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis. Journal of Translational Medicine, 13(49), 1–14
Hu, G. W., Li, Q., Niu, X., Hu, B., Liu, J., Zhou, S. M., & Deng, Z. F. (2015). Exosomes secreted by human-induced pluripotent stem cell-derived mesenchymal stem cells attenuate limb ischemia by promoting angiogenesis in mice. Stem Cell Research and Therapy, 6(10), 1–15
Mahmood, A., Harkness, L., Abdallah, B. M., Elsafadi, M., Al-Nbaheen, M. S., Aldahmash, A., & Kassem, M. (2012). Derivation of stromal (skeletal and mesenchymal) stem-like cells from human embryonic stem cells. Stem Cells and Development, 21(17), 3114–3124
Marolt, D., Campos, I. M., Bhumiratana, S., Koren, A., Petridis, P., Zhang, G., & Vunjak-Novakovic, G. (2012). Engineering bone tissue from human embryonic stem cells. Proceedings of the National Academy of Sciences of the United States of America, 109(22), 8705–8709
De Peppo, G. M., Marcos-Campos, I., Kahler, D. J., Alsalman, D., Shang, L., Vunjak-Novakovic, G., & Marolt, D. (2013). Engineering bone tissue substitutes from human induced pluripotent stem cells. Proceedings of the National Academy of Sciences of the United States of America, 110(21), 8680–8685
McGrath, M., Tam, E., Sladkova, M., Almanaie, A., Zimmer, M., & De Peppo, G. M. (2019). GMP-compatible and xeno-free cultivation of mesenchymal progenitors derived from human-induced pluripotent stem cells. Stem Cell Research and Therapy, 10(11), 1–13
Chen, W., Zhou, H., Weir, M. D., Tang, M., Bao, C., & Xu, H. H. K. (2013). Human embryonic stem cell-derived mesenchymal stem cell seeding on calcium phosphate cement-chitosan-rgd scaffold for bone repair. Tissue Engineering - Part A, 19(7–8), 915–927
Liu, J., Chen, W., Zhao, Z., & Xu, H. H. K. (2013). Reprogramming of mesenchymal stem cells derived from iPSCs seeded in biofunctionalized calcium phosphate scaffold for bone engineering. Biomaterials, 34(32), 1–22
Wang, P., Liu, X., Zhao, L., Weir, M. D., Sin, J., Chen, W., & Xu, H. H. K. (2015). Bone tissue engineering via human induced pluripotent, umbilical cord and bone marrow mesenchymal stem cells in rat cranium. Acta Biomaterialia, 18(1), 236–248
Hynes, K., Menicanin, D., Han, J., Marino, V., Mrozik, K., Gronthos, S., & Bartold, P. M. (2013). Mesenchymal stem cells from iPS cells facilitate periodontal regeneration. Journal of Dental Research, 92(9), 833–839
Hynes, K., Menicanin, D., Mrozik, K., Gronthos, S., & Bartold, P. M. (2014). Generation of functional mesenchymal stem cells from different induced pluripotent stem cell lines. Stem Cells and Development, 23(10), 1084–1096
Ng, J., Hynes, K., White, G., Sivanathan, K. N., Vandyke, K., Bartold, P. M., & Gronthos, S. (2016). Immunomodulatory properties of induced pluripotent stem cell-derived mesenchymal cells. Journal of Cellular Biochemistry, 117(1), 2844–2853
Billing, A. M., Hamidane, B., Dib, H., Cotton, S. S., Bhagwat, R. J., Kumar, A. M., & Graumann, P. (2016). Comprehensive transcriptomic and proteomic characterization of human mesenchymal stem cells reveals source specific cellular markers. Scientific Reports, 6(21507), 1–15
Raynaud, C. M., Halabi, N., Elliott, D. A., Pasquier, J., Elefanty, A. G., Stanley, E. G., & Rafii, A. (2013). Human embryonic stem cell derived mesenchymal progenitors express cardiac markers but do not form contractile cardiomyocytes. PLoS One1, 8(1), 1–13
Theinhan, W., Liu, J., Tang, M., Chen, W., Cheng, L., & Xu, H. H. K. (2013). Induced pluripotent stem cell-derived mesenchymal stem cell seeding on biofunctionalized calcium phosphate cements. Bone Research, 4(1), 371–384
Diederichs, S., & Tuan, R. S. (2014). Functional comparison of human-induced pluripotent stem cell-derived mesenchymal cells and bone marrow-derived mesenchymal stromal cells from the same donor. Stem Cells and Development, 23(14), 1594–1610
Umezaki, Y., Hashimoto, Y., Nishishita, N., Kawamata, S., & Baba, S. (2015). Human gingival integration-free iPSCs; a source for MSC-like cells. International Journal of Molecular Sciences, 16(6), 13633–13648
Gao, W. X., Sun, Y. Q., Shi, J., Li, C. L., Fang, S., Bin, Wang, D., & Fu, Q. L. (2017). Effects of mesenchymal stem cells from human induced pluripotent stem cells on differentiation, maturation, and function of dendritic cells. Stem Cell Research and Therapy, 8(48), 1–16
Lin, Y. D., Fan, X. L., Zhang, H., Fang, S., Bin, Li, C. L., Deng, M. X., & Fu, Q. L. (2018). The genes involved in asthma with the treatment of human embryonic stem cell-derived mesenchymal stem cells. Molecular Immunology, 95(1), 47–55
Kim, S., Lee, S. K., Kim, H., & Kim, T. M. (2018). Exosomes secreted from induced pluripotent stem cell-derived mesenchymal stem cells accelerate skin cell proliferation. International Journal of Molecular Sciences, 19(3119), 1–16
Wang, D., Sun, Y. Q., Gao, W. X., Fan, X. L., Shi, J. B., & Fu, Q. L. (2018). An in vitro and in vivo study of the effect of dexamethasone on immunoinhibitory function of induced pluripotent stem cell-derived mesenchymal stem cells. Cell Transplantation, 27(9), 1340–1351
Billing, A. M., Dib, S. S., Bhagwat, A. M., da Silva, I. T., Drummond, R. D., Hayat, S., & Graumann, J. (2019). A systems-level characterization of the differentiation of human embryonic stem cells into mesenchymal stem cells. Molecular and Cellular Proteomics, 18(10), 1950–1966
Xu, M., Shaw, G., Murphy, M., & Barry, F. (2019). Induced pluripotent stem cell-derived mesenchymal stromal cells are functionally and genetically different from bone marrow-derived mesenchymal stromal cells. Stem Cells, 37(6), 754–765
Cha, B. H., Kim, J. S., Bello, A., Lee, G. H., Kim, D. H., Kim, B. J., & Lee, S. H. (2020). Efficient isolation and enrichment of mesenchymal stem cells from human embryonic stem cells by utilizing the interaction between integrin α5β1 and fibronectin. Advanced Science, 7(17), 1–15
Kwon, D., Ahn, H. J., Han, M. J., Ji, M., Ahn, J., Seo, K. W., & Kang, K. S. (2020). Human leukocyte antigen class I pseudo-homozygous mesenchymal stem cells derived from human induced pluripotent stem cells. Stem Cell Reviews and Reports, 16(4), 792–808
Wu, J., Song, D., Li, Z., Guo, B., Xiao, Y., Liu, W., & Hao, J. (2020). Immunity-and-matrix-regulatory cells derived from human embryonic stem cells safely and effectively treat mouse lung injury and fibrosis. Cell Research, 0(1–16), 794–809
Frobel, J., Hemeda, H., Lenz, M., Abagnale, G., Joussen, S., Denecke, B., & Wagner, W. (2014). Epigenetic rejuvenation of mesenchymal stromal cells derived from induced pluripotent stem cells. Stem Cell Reports, 3(1), 1–9
Hollmann, J., Brecht, J., Goetzke, R., Franzen, J., Selich, A., Schmidt, M., & Wagner, W. (2020). Genetic barcoding reveals clonal dominance in iPSC-derived mesenchymal stromal cells. Stem Cell Research and Therapy, 11(105), 1–13
Fernandez-Rebollo, E., Franzen, J., Goetzke, R., Hollmann, J., Ostrowska, A., Oliverio, M., & Wagner, W. (2020). Senescence-associated metabolomic phenotype in primary and iPSC-derived mesenchymal stromal cells. Stem Cell Reports, 14(2), 201–209
Luzzani, C., Neiman, G., Garate, X., Questa, M., Solari, C., Fernandez Espinosa, D., & Miriuka, S. G. (2015). A therapy-grade protocol for differentiation of pluripotent stem cells into mesenchymal stem cells using platelet lysate as supplement. Stem Cell Research and Therapy, 6(6), 1–13
La Greca, A., Solari, C., Furmento, V., Lombardi, A., Biani, M. C., Aban, C., & Luzzani, C. (2018). Extracellular vesicles from pluripotent stem cell-derived mesenchymal stem cells acquire a stromal modulatory proteomic pattern during differentiation. Experimental and Molecular Medicine, 50(119), 1–12
Goetzke, R., Franzen, J., Ostrowska, A., Vogt, M., Blaeser, A., Klein, G., & Wagner, W. (2018). Does soft really matter? Differentiation of induced pluripotent stem cells into mesenchymal stromal cells is not influenced by soft hydrogels. Biomaterials, 156(1), 147–158
Barberi, T., Willis, L. M., Socci, N. D., & Studer, L. (2005). Derivation of multipotent mesenchymal precursors from human embryonic stem cells. PLoS Medicine, 2(6), 0554–0560
Trivedi, P., & Hematti, P. (2007). Simultaneous generation of CD34 + primitive hematopoietic cells and CD73 + mesenchymal stem cells from human embryonic stem cells cocultured with murine OP9 stromal cells. Experimental Hematology, 35(1), 146–154
Kopher, R. A., Penchev, V. R., Islam, M. S., Hill, K. L., Khosla, S., & Kaufman, D. S. (2010). Human embryonic stem cell-derived CD34 + cells function as MSC progenitor cells. Bone, 47(4), 718–728
Evseenko, D., Zhu, Y., Schenke-Layland, K., Kuo, J., Latour, B., Ge, S., & Crooks, G. M. (2010). Mapping the first stages of mesoderm commitment during differentiation of human embryonic stem cells. Proceedings of the National Academy of Sciences of the United States of America, 107(31), 13742–13747
Chin, C. J., Cooper, A. R., Lill, G. R., Evseenko, D., Zhu, Y., He, C., & Bin, … Crooks, G. M. (2016). Genetic tagging during human mesoderm differentiation reveals tripotent lateral plate mesodermal progenitors. Stem Cells, 34(5), 1239–1250
Chin, C. J., Li, S., Corselli, M., Casero, D., Zhu, Y., He, C., & Bin, … Crooks, G. M. (2018). Transcriptionally and functionally distinct mesenchymal subpopulations are generated from human pluripotent stem cells. Stem Cell Reports, 10(2), 436–446
Vodyanik, M. A., Yu, J., Zhang, X., Tian, S., Stewart, R., Thomson, J. A., & Slukvin, I. I. (2010). A mesoderm-derived precursor for mesenchymal stem and endothelial cells. Cell Stem Cell, 7(6), 718–729
Brown, P. T., Squire, M. W., & Li, W. J. (2014). Characterization and evaluation of mesenchymal stem cells derived from human embryonic stem cells and bone marrow. Cell and Tissue Research, 358(1), 149–164
Xie, W., Schultz, M. D., Lister, R., Hou, Z., Rajagopal, N., Ray, P., & Ren, B. (2013). Epigenomic analysis of multilineage differentiation of human embryonic stem cells. Cell, 153(5), 1134–1148
Bloor, A. J. C., Patel, A., Griffin, J. E., Gilleece, M. H., Radia, R., Yeung, D. T., & Rasko, J. E. J. (2020). Production, safety and efficacy of iPSC-derived mesenchymal stromal cells in acute steroid-resistant graft versus host disease: a phase I, multicenter, open-label, dose-escalation study. Nature Medicine, 26(11), 1–15
Tran, N. T., Trinh, Q. M., Lee, G. M., & Han, Y. M. (2012). Efficient differentiation of human pluripotent stem cells into mesenchymal stem cells by modulating intracellular signaling pathways in a feeder/serum-free system. Stem Cells and Development, 21(7), 1165–1175
Nakayama, C., Fujita, Y., Matsumura, W., Ujiie, I., Takashima, S., Shinkuma, S., & Shimizu, H. (2018). The development of induced pluripotent stem cell-derived mesenchymal stem/stromal cells from normal human and RDEB epidermal keratinocytes. Journal of Dermatological Science, 91(3), 301–310
Wei, H., Tan, G., Manasi, Qiu, S., Kong, G., Yong, P., & Shim, W. (2012). One-step derivation of cardiomyocytes and mesenchymal stem cells from human pluripotent stem cells. Stem Cell Research, 9(2), 87–100
Miao, Q., Shim, W., Tee, N., Lim, S. Y., Chung, Y. Y., Mia Ja, M., & Wong, K. P. (2014). iPSC-derived human mesenchymal stem cells improve myocardial strain of infarcted myocardium. Journal of Cellular and Molecular Medicine, 18(8), 1644–1654
Kimbrel, E. A., Kouris, N. A., Yavanian, G. J., Chu, J., Qin, Y., Chan, A., & Lanza, R. (2014). Mesenchymal stem cell population derived from human pluripotent stem cells displays potent immunomodulatory and therapeutic properties. Stem Cells and Development, 23(14), 1611–1624
Wang, X., Kimbrel, E. A., Ijichi, K., Paul, D., Lazorchak, A. S., Chu, J., & Xu, R. H. (2014). Human ESC-derived MSCs outperform bone marrow MSCs in the treatment of an EAE model of multiple sclerosis. Stem Cell Reports, 3(1), 115–130
Deng, P., Zhou, C., Alvarez, R., Hong, C., & Wang, C. Y. (2016). Inhibition of IKK/NF-κB signaling enhances differentiation of mesenchymal stromal cells from human embryonic stem cells. Stem Cell Reports, 6(4), 456–465
Yu, Y., Deng, P., Yu, B., Szymanski, J. M., Aghaloo, T., Hong, C., & Wang, C. Y. (2017). Inhibition of EZH2 promotes human embryonic stem cell differentiation into mesoderm by reducing H3K27me3. Stem Cell Reports, 9(3), 752–761
Lee, G., Kim, H., Elkabetz, Y., Shamy, A., Panagiotakos, G., Barberi, G., & Studer, T. (2007). Isolation and directed differentiation of neural crest stem cells derived from human embryonic stem cells. Nature Biotechnology, 25(12), 1468–1475
Menendez, L., Yatskievych, T. A., Antin, P. B., & Dalton, S. (2011). Wnt signaling and a smad pathway blockade direct the differentiation of human pluripotent stem cells to multipotent neural crest cells. Proceedings of the National Academy of Sciences of the United States of America, 108(48), 19240–19245
Fukuta, M., Nakai, Y., Kirino, K., Nakagawa, M., Sekiguchi, K., Nagata, S., & Toguchida, J. (2014). Derivation of mesenchymal stromal cells from pluripotent stem cells through a neural crest lineage using small molecule compounds with defined media. PLoS One1, 9(12), 1–25
Eto, S., Goto, M., Soga, M., Kaneko, Y., Uehara, Y., Mizuta, H., & Era, T. (2018). Mesenchymal stem cells derived from human iPS cells via mesoderm and neuroepithelium have different features and therapeutic potentials. PLoS One1, 13(7), 1–22
Zhang, L., Wang, H., Liu, C., Wu, Q., Su, P., Wu, D., & Zhou, J. (2018). MSX2 initiates and accelerates mesenchymal stem/stromal cell specification of hPSCs by regulating TWIST1 and PRAME. Stem Cell Reports, 11(2), 497–513
Kamiya, D., Takenaka-Ninagawa, N., Motoike, S., Kajiya, M., Akaboshi, T., Zhao, C., & Ikeya, M. … (2020). Induction of functional mesenchymal stem/stromal cells from human iPSCs via a neural crest cell lineage under xeno-free conditions. Cell Reports Medicine, 1–69. Available at SSRN: https://ssrn.com/abstract=3741231 or https://doi.org/10.2139/ssrn.3741231
Mitsuzawa, S., Zhao, C., Ikeguchi, R., Aoyama, T., Kamiya, D., Ando, M., & Ikeya, M. (2020). Pro-angiogenic scaffold-free bio three-dimensional conduit developed from human induced pluripotent stem cell-derived mesenchymal stem cells promotes peripheral nerve regeneration. Scientific Reports, 10(12034), 1–15
Yoshimatsu, M., Ohnishi, H., Zhao, C., Hayashi, Y., Kuwata, F., Kaba, S., & Omori, K. (2021). In vivo regeneration of rat laryngeal cartilage with mesenchymal stem cells derived from human induced pluripotent stem cells via neural crest cells. Stem Cell Research, 52, 1–12
Wang, X., Lazorchak, A. S., Song, L., Li, E., Zhang, Z., Jiang, B., & Xu, R. H. (2016). Immune modulatory mesenchymal stem cells derived from human embryonic stem cells through a trophoblast-like stage. Stem Cells, 34(2), 380–391
Li, E., Zhang, Z., Jiang, B., Yan, L., Park, J. W., & Xu, R. H. (2018). Generation of mesenchymal stem cells from human embryonic stem cells in a complete serum-free condition. International Journal of Biological Sciences, 14(13), 1901–1909
Yan, L., Jiang, B., Li, E., Wang, X., Ling, Q., Zheng, D., & Xu, R. H. (2018). Scalable generation of mesenchymal stem cells from human embryonic stem cells in 3D. International Journal of Biological Sciences, 14(10), 1196–1210
Lian, Q., Lye, E., Suan Yeo, K., Khia Way Tan, E., Salto-Tellez, M., Liu, T. M., & Lim, S. K. (2007). Derivation of clinically compliant MSCs from CD105+, CD24 – differentiated human ESCs. Stem Cells, 25(2), 425–436
Lian, Q., Zhang, Y., Zhang, J., Zhang, H. K., Wu, X., Zhang, Y., & Tse, H. F. (2010). Functional mesenchymal stem cells derived from human induced pluripotent stem cells attenuate limb ischemia in mice. Circulation, 121(9), 1113–1123
Zhang, Y., Liang, X., Liao, S., Wang, W., Wang, J., Li, X., & Lian, Q. (2015). Potent paracrine effects of human induced pluripotent stem cell-derived mesenchymal stem cells attenuate doxorubicin-induced cardiomyopathy. Scientific Reports, 5(11235), 1–17
Chen, T. S., Arslan, F., Yin, Y., Tan, S. S., Lai, R. C., Choo, A. B. H., & Lim, S. K. (2011). Enabling a robust scalable manufacturing process for therapeutic exosomes through oncogenic immortalization of human ESC-derived MSCs. Journal of Translational Medicine, 9(47), 1–10
Lai, R. C., Wee, R., Yeo, Y., Padmanabhan, J., Choo, A., Kleijn, D. P. V., & Lim, S. K. (2016). Isolation and characterization of exosome from human embryonic stem cell-derived C-Myc-immortalized mesenchymal stem cells. Methods in Molecular Biology, 1416(1), 477–494
Sze, S. K., de Kleijn, D. P. V., Lai, R. C., Tan, E. K. W., Zhao, H., Yeo, K. S., & Lim, S. K. (2007). Elucidating the secretion proteome of human embryonic stem cell-derived mesenchymal stem cells. Molecular and Cellular Proteomics, 6(10), 1680–1689
Zhang, J., Lian, Q., Zhu, G., Zhou, F., Sui, L., Tan, C., & Colman, A. (2011). A human iPSC model of Hutchinson Gilford Progeria reveals vascular smooth muscle and mesenchymal stem cell defects. Cell Stem Cell, 8(1), 31–45
Zhang, Y., Liao, S., Yang, M., Liang, X., Poon, M. W., Wong, C. Y., & Lian, Q. (2012). Improved cell survival and paracrine capacity of human embryonic stem cell-derived mesenchymal stem cells promote therapeutic potential for pulmonary arterial hypertension. Cell Transplantation, 21(10), 2225–2239
Fu, Q. L., Chow, Y. Y., Sun, S. J., Zeng, Q. X., Li, H. B., Shi, J. B., & Xu, G. (2012). Mesenchymal stem cells derived from human induced pluripotent stem cells modulate T-cell phenotypes in allergic rhinitis. Allergy: European Journal of Allergy and Clinical Immunology, 67(10), 1215–1222
Sun, Y. Q., Deng, M. X., He, J., Zeng, Q. X., Wen, W., Wong, D. S. H., & Fu, Q. L. (2012). Human pluripotent stem cell-derived mesenchymal stem cells prevent allergic airway inflammation in mice. Stem Cells, 30(12), 2692–2699
Cheng, P. P., Liu, X. C., Ma, P. F., Gao, C., Li, J. L., Lin, Y. Y., & Qi, Z. Q. (2015). IPSC-MSCs combined with low-dose rapamycin induced islet allograft tolerance through suppressing Th1 and enhancing regulatory T-Cell differentiation. Stem Cells and Development, 24(15), 1793–1804
Shi, H., Liang, M., Chen, W., Sun, X., Wang, X., Li, C., & Zeng, W. (2018). Human induced pluripotent stem cell-derived mesenchymal stem cells alleviate atherosclerosis by modulating inflammatory responses. Molecular Medicine Reports, 17(1), 1461–1468
Jiang, D., Xiong, G., Feng, H., Zhang, Z., Chen, P., Yan, B., & Lian, Q. (2019). Donation of mitochondria by iPSC-derived mesenchymal stem cells protects retinal ganglion cells against mitochondrial complex I defect-induced degeneration. Theranostics, 9(8), 2395–2410
Boyd, N. L., Robbins, K. R., Dhara, S. K., West, F. D., & Stice, S. L. (2009). Human embryonic stem cell-derived mesoderm-like epithelium transitions to mesenchymal progenitor cells. Tissue Engineering - Part A, 15(8), 1897–1907
Mahmood, A., Harkness, L., Schrøder, H. D., Abdallah, B. M., & Kassem, M. (2010). Enhanced differentiation of human embryonic stem cells to mesenchymal progenitors by inhibition of TGF-β/activin/nodal signaling using SB-431542. Journal of Bone and Mineral Research, 25(6), 1216–1233
Sánchez, L., Gutierrez-Aranda, I., Ligero, G., Rubio, R., Muñoz-López, M., García-Pérez, J. L., & Menendez, P. (2011). Enrichment of human ESC-derived multipotent mesenchymal stem cells with immunosuppressive and anti-inflammatory properties capable to protect against experimental inflammatory Bowel disease. Stem Cells, 29(2), 251–262
Gonzalo-Gil, E., Pérez-Lorenzo, M. J., Galindo, M., de la Díaz, R., López-Millán, B., Bueno, C., & Criado, G. (2016). Human embryonic stem cell-derived mesenchymal stromal cells ameliorate collagen-induced arthritis by inducing host-derived indoleamine 2,3 dioxygenase. Arthritis Research and Therapy, 18(77), 1–9
Spitzhorn, L. S., Megges, M., Wruck, W., Rahman, M. S., Otte, J., Degistirici, Ã, & Adjaye, J. (2019). Human iPSC-derived MSCs (iMSCs) from aged individuals acquire a rejuvenation signature. Stem Cell Research and Therapy, 10(100), 1–18
Chen, Y. S., Pelekanos, R. A., Ellis, R. L., Horne, R., Wolvetang, E. J., & Fisk, N. M. (2012). Small molecule mesengenic induction of human induced pluripotent stem cells to generate mesenchymal stem/stromal cells. Stem Cells Translational Medicine, 1(1), 83–95
Leyendecker Junior, A. (2018). TGF-β Inhibitor SB431542 promotes the differentiation of induced pluripotent stem cells and embryonic stem cells into mesenchymal-like cells. Stem Cells International, 2018(1), 1–13
Hawkins, K. E., Corcelli, M., Dowding, K., Ranzoni, A. M., Vlahova, F., Hau, K. L., & Guillot, P. V. (2018). Embryonic stem cell-derived mesenchymal stem cells (MSCs) have a superior neuroprotective capacity over fetal MSCs in the hypoxic-ischemic mouse brain. Stem Cells Translational Medicine, 7(5), 439–449
Spitzhorn, L. S., Kordes, C., Megges, M., Sawitza, I., Götze, S., Reichert, D., & Adjaye, J. (2018). Transplanted human pluripotent stem cell-derived mesenchymal stem cells support liver regeneration in Gunn rats. Stem Cells and Development, 27(24), 1702–1714
Jungbluth, P., Spitzhorn, L. S., Grassmann, J., Tanner, S., Latz, D., Rahman, M. S., & Adjaye, J. (2019). Human iPSC-derived iMSCs improve bone regeneration in mini-pigs. Bone Research, 7(32), 1–11
Zhao, Q., Gregory, C. A., Lee, R. H., Reger, R. L., Qin, L., Hai, B., & Liu, F. (2015). MSCs derived from iPSCs with a modified protocol are tumor-tropic but have much less potential to promote tumors than bone marrow MSCs. Proceedings of the National Academy of Sciences of the United States of America, 112(2), 530–535
Ullah, M., Kuroda, Y., Bartosh, T. J., Liu, F., Zhao, Q., Gregory, C., & Prockop, D. J. (2017). IPS-derived MSCS from an expandable bank to deliver a prodrug-converting enzyme that limits growth and metastases of human breast cancers. Cell Death Discovery, 3(1), 1–10
McNeill, E. P., Zeitouni, S., Pan, S., Haskell, A., Cesarek, M., Tahan, D., & Gregory, C. A. (2020). Characterization of a pluripotent stem cell-derived matrix with powerful osteoregenerative capabilities. Nature Communications, 11(3025), 1–15
Zhao, Q., Hai, B., Zhang, X., Xu, J., Koehler, B., & Liu, F. (2020). Biomimetic nanovesicles made from iPS cell-derived mesenchymal stem cells for targeted therapy of triple-negative breast cancer. Nanomedicine: Nanotechnology, Biology, and Medicine, 24, 1–11
Zhao, Q., Hai, B., Kelly, J., Wu, S., & Liu, F. (2021). Extracellular vesicle mimics made from iPS cell-derived mesenchymal stem cells improve the treatment of metastatic prostate cancer. Stem Cell Research and Therapy, 12(29), 1–13
Sheyn, D., Ben-David, S., Shapiro, G., De Mel, S., Bez, M., Ornelas, L., & Gazit, Z. (2016). Human induced pluripotent stem cells differentiate into functional mesenchymal stem cells and repair bone defects. Stem Cells Translational Medicine, 5, 1447–1460
Karam, M., Younis, I., Elareer, N. R., Nasser, S., & Abdelalim, E. M. (2020). Scalable generation of mesenchymal stem cells and adipocytes from human pluripotent stem cells. Cells, 9(710), 1–23
Zhang, L., Wei, Y., Chi, Y., Liu, D., Yang, S., Han, Z., & Li, Z. (2021). Two-step generation of mesenchymal stem/stromal cells from human pluripotent stem cells with reinforced efficacy upon osteoarthritis rabbits by HA hydrogel. Cell and Bioscience, 11(6), 1–17
Guiotto, M., Raffoul, W., Hart, A. M., Riehle, M. O., & Di Summa, P. G. (2020). Human platelet lysate to substitute fetal bovine serum in hMSC expansion for translational applications: a systematic review. Journal of Translational Medicine, 18(351), 1–14
Andrzejewska, A., Lukomska, B., & Janowski, M. (2019). Concise review: mesenchymal stem cells: from roots to boost. Stem Cells, 37(7), 855–864
Sheng, G. (2015). The developmental basis of mesenchymal stem/stromal cells (MSCs). BMC Developmental Biology, 15(44), 1–8
Slukvin, I. I., & Kumar, A. (2018). The mesenchymoangioblast, mesodermal precursor for mesenchymal and endothelial cells. Cellular and Molecular Life Sciences, 75(19), 3507–3520
Lamouille, S., Xu, J., & Derynck, R. (2014). Molecular mechanisms of epithelial-mesenchymal transition. Nature Reviews Molecular Cell Biology, 15(3), 178–196
Chen, T., You, Y., Jiang, H., & Wang, Z. Z. (2017). Epithelial–mesenchymal transition (EMT): A biological process in the development, stem cell differentiation, and tumorigenesis. Journal of Cellular Physiology, 232(12), 3261–3272
Lu, S. J., Feng, Q., Caballero, S., Chen, Y., Moore, M. A. S., Grant, M. B., & Lanza, R. (2007). Generation of functional hemangioblasts from human embryonic stem cells. Nature Methods, 4(6), 1–18
Lu, S. J., Luo, C., Holton, K., Feng, Q., Ivanova, Y., & Lanza, R. (2008). Robust generation of hemangioblastic progenitors from human embryonic stem cells. Regenerative Medicine, 3(5), 693–704
Wu, Q., Zhang, L., Su, P., Lei, X., Liu, X., Wang, H., & Zhou, J. (2015). MSX2 mediates entry of human pluripotent stem cells into mesendoderm by simultaneously suppressing SOX2 and activating NODAL signaling. Cell Research, 25(12), 1314–1332
Feng, X. H., & Derynck, R. (2005). Specificity and versatility in TGF-β signaling through smads. Annual Review of Cell and Developmental Biology, 21, 659–693
Mullen, A. C., & Wrana, J. L. (2017). TGF-β family signaling in embryonic and somatic stem-cell renewal and differentiation. Cold Spring Harbor Perspectives in Biology, 9(7), 1–33
Acknowledgements
MATOS, B.M. received a fellowship from Carlos Chagas Institute (ICC/FIOCRUZ-PR). We thank Wagner Nagib from the Communication Advisory (ASCOM/ICC) for helping to refine the illustrations of this review.
Author information
Authors and Affiliations
Contributions
The idea of the article was conceived due to difficulties to reproduce published MSC derivation protocols. Bruno Moisés de Matos and Anny Waloski Robert performed literature research and data analysis. Marco Augusto Stimamiglio wrote the MSC state of the art introduction. Alejandro Correa, Bruno Moisés de Matos and Anny Waloski Robert contributed to writing the topics about derivation protocols. Graphical abstract, Figs. 2 and 3 were idealized by Bruno Moisés de Matos. Figures 1 and 4, as well as Tables 1 and 2 were idealized and formatted by Anny Waloski Robert. The work was critically revised by all authors, especially Marco Augusto Stimamiglio and Alejandro Correa. All authors approved the present manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethical Approval
Not applicable.
Consent to Participate
Not applicable.
Consent to Publish
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Matos, B.M., Robert, A.W., Stimamiglio, M.A. et al. Pluripotent-derived Mesenchymal Stem/stromal Cells: an Overview of the Derivation Protocol Efficacies and the Differences Among the Derived Cells. Stem Cell Rev and Rep 18, 94–125 (2022). https://doi.org/10.1007/s12015-021-10258-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-021-10258-z