Abstract
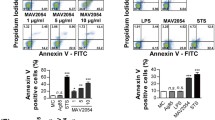
To investigate the effects and mechanisms of Mycobacterium avium MAV-5183 protein on apoptosis in mouse Ana-1 macrophages. A pET-21a-MAV-5183 recombinant plasmid was constructed. The recombinant MAV-5183 protein was cloned, expressed, purified, and identified using an anti-His-tagged antibody. Rabbits were immunized to obtain antiserum, and its potency and immunoreactivity were assessed through WB. Mouse Ana-1 macrophages were incubated with varying concentrations of MAV-5183 protein. Flow cytometry, following ANNEXIN V-FITC/PI double staining, detected apoptosis. Western Blot analysis was conducted to identify apoptosis-related molecules Caspase-9/8/3 and vesicle-related molecules ASC, NLRP3, and Cleaved-casp1. ELISA measured TNF-α and IL-6 levels in the culture supernatant. LDH activity and ROS levels were analyzed separately. RT-qPCR measured mRNA levels of Caspase-9/8/3, ASC, NLRP3, Caspase-1, IL-1β, Bax, MAPK-p38, Bcl-2, TNF-α, and IL-6. MAV-5183 protein was successfully cloned, purified, and identified. In in vitro studies on Ana-1 macrophages, MAV-5183 protein increased the expression of Caspase-9/8/3, ASC, NLRP3 (P < 0.01), induced ROS secretion (P < 0.05), and promoted inflammatory cytokine secretion (TNF-α, IL-6, P < 0.0001); however, it did not significantly affect LDH (P > 0.05). MAV-5183 also induced apoptosis in Ana-1 macrophages (P < 0.05). RT-qPCR results indicated a significant increase in mRNA expression of Caspase-9/8/3, ASC, NLRP3, TNF-α, IL-6, MAPK-p38, and pro-apoptotic factor Bax (P < 0.01), with no significant effect on Bcl-2 and IL-1β mRNA (P > 0.05). The data indicate that MAV-5183 induces macrophage apoptosis through a caspase-dependent pathway and promotes inflammatory cytokine secretion via ROS.







Similar content being viewed by others
Data Availability
The data supporting this study’s findings are available from the corresponding author upon reasonable request.
Abbreviations
- MAV:
-
Mycobacterium avium
- RT-qPCR:
-
Real-time quantitative PCR
References
Kang, Y. A. & Koh, W. J. (2016). Antibiotic treatment for nontuberculous mycobacterial lung disease. Expert Review of Respiratory Medicine, 10, 557–568.
Wang, X., Chen, S., Ren, H., Chen, J., Li, J., Wang, Y., Hua, Y., Wang, X. & Huang, N. (2019). HMGN2 regulates non-tuberculous mycobacteria survival via modulation of M1 macrophage polarization. Journal of Cellular and Molecular Medicine, 23, 7985–7998.
Vergne, I., Chua, J., Singh, S. B. & Deretic, V. (2004). Cell biology of mycobacterium tuberculosis phagosome. Annual Review of Cell and Developmental Biology, 20, 367–394.
Bhattacharyya, A., Pathak, S., Basak, C., Law, S., Kundu, M. & Basu, J. (2003). Execution of macrophage apoptosis by Mycobacterium avium through apoptosis signal-regulating kinase 1/p38 mitogen-activated protein kinase signaling and caspase 8 activation. Journal of Biological Chemistry, 278, 26517–26525.
Bermudez, L. E., Parker, A. & Petrofsky, M. (1999). Apoptosis of Mycobacterium avium-infected macrophages is mediated by both tumour necrosis factor (TNF) and Fas, and involves the activation of caspases. Clinical & Experimental Immunology, 116, 94–99.
Lee, K. I., Whang, J., Choi, H. G., Son, Y. J., Jeon, H. S., Back, Y. W., Park, H. S., Paik, S., Park, J. K., Choi, C. H. & Kim, H. J. (2016). Mycobacterium avium MAV2054 protein induces macrophage apoptosis by targeting mitochondria and reduces intracellular bacterial growth. Scientific Reports, 6, 37804.
Lewis, M. S., Danelishvili, L., Rose, S. J., & Bermudez, L. E. (2019). MAV_4644 interaction with the host cathepsin Z protects mycobacterium avium subsp. hominissuis from rapid macrophage killing. Microorganisms, 7, 144.
Bermudez, L. E., Danelishvili, L., Babrack, L. & Pham, T. (2015). Evidence for genes associated with the ability of Mycobacterium avium subsp. hominissuis to escape apoptotic macrophages. Frontiers in Cellular and Infection Microbiology, 5, 63.
Shin, A. R., Lee, K. S., Lee, K. I., Shim, T. S., Koh, W. J., Jeon, H. S., Son, Y. J., Shin, S. J. & Kim, H. J. (2013). Serodiagnostic potential of Mycobacterium avium MAV2054 and MAV5183 proteins. Clinical and Vaccine Immunology, 20, 295–301.
Gidon, A., Louet, C., Rost, L. M., Bruheim, P., & Flo, T. H. (2021). The tumor necrosis factor alpha and interleukin 6 auto-paracrine signaling loop controls mycobacterium avium infection via induction of IRF1/IRG1 in human primary macrophages. mBio, 12, e0212121.
Rodrigues, M. F., Barsante, M. M., Alves, C. C., Souza, M. A., Ferreira, A. P., Amarante-Mendes, G. P., & Teixeira, H. C. (2009). Apoptosis of macrophages during pulmonary Mycobacterium bovis infection: correlation with intracellular bacillary load and cytokine levels. Immunology, 128, e691–e699.
Wojtas, B., Fijalkowska, B., Wlodarczyk, A., Schollenberger, A., Niemialtowski, M., Hamasur, B., Pawlowski, A. & Krzyzowska, M. (2011). Mannosylated lipoarabinomannan balances apoptosis and inflammatory state in mycobacteria-infected and uninfected bystander macrophages. Microbial Pathogenesis, 51, 9–21.
Lee, K. I., Choi, H. G., Son, Y. J., Whang, J., Kim, K., Jeon, H. S., Park, H. S., Back, Y. W., Choi, S., Kim, S. W., Choi, C. H., & Kim, H. J. (2016). Mycobacterium avium MAV2052 protein induces apoptosis in murine macrophage cells through Toll-like receptor 4. Apoptosis, 21, 459–472.
Ding, S., Li, X., & Gao, J. (2021). Bioinformatics analysis of MAV—5183 protein of Mycobacterium avium tuberculosis. Chinese. Journal of Pathogen Biology, 16, 1153–1157.
Sánchez, A., Espinosa, P., García, T. & Mancilla, R. (2012). The 19 kDa Mycobacterium tuberculosis lipoprotein (LpqH) induces macrophage apoptosis through extrinsic and intrinsic pathways: a role for the mitochondrial apoptosis-inducing factor. Clinical and Developmental Immunology, 2012, 950503.
Li, Y., Miltner, E., Wu, M., Petrofsky, M. & Bermudez, L. E. (2005). A Mycobacterium avium PPE gene is associated with the ability of the bacterium to grow in macrophages and virulence in mice. Cellular Microbiology, 7, 539–548.
Abate, M., Festa, A., Falco, M., Lombardi, A., Luce, A., Grimaldi, A., Zappavigna, S., Sperlongano, P., Irace, C., Caraglia, M. & Misso, G. (2020). Mitochondria as playmakers of apoptosis, autophagy and senescence. Seminars in Cell and Developmental Biology, 98, 139–153.
Ganju, N. & Eastman, A. (2002). Bcl-X(L) and calyculin A prevent translocation of Bax to mitochondria during apoptosis. Biochemical and Biophysical Research Communications, 291, 1258–1264.
Sohn, H., Kim, J. S., Shin, S. J., Kim, K., Won, C. J., Kim, W. S., Min, K. N., Choi, H. G., Lee, J. C., Park, J. K., & Kim, H. J. (2011). Targeting of Mycobacterium tuberculosis heparin-binding hemagglutinin to mitochondria in macrophages. PLoS Pathogens, 7, e1002435.
Kuai, S. G., Pei, H., Huang, L. H., Liu, Z. H., Mai, G. L., Liu, J. & Cui, Z. L. (2013). [Cell death of THP-1 induced by puried Rv3671c protein of tuberculosis and the detection of TNF-α and IL-1β in Mycobacterium tuberculosis]. Zhonghua Yu Fang Yi Xue Za Zhi 47, 444–447.
Denis, M.(1991). Tumor necrosis factor and granulocyte macrophage-colony stimulating factor stimulate human macrophages to restrict growth of virulent Mycobacterium avium and to kill avirulent M. avium: killing effector mechanism depends on the generation of reactive nitrogen intermediates. Journal of Leukocyte Biology, 49, 380–387.
Zuo, X., Wang, L., Bao, Y. & Sun, J. (2020). The ESX-1 virulence factors downregulate miR-147-3p in mycobacterium marinum-infected macrophages. Infection and Immunity, 88, e00088–20.
Knight, V.(2022). Immunodeficiency and autoantibodies to cytokines. Journal of Applied Laboratory Medicine, 7, 151–164.
Al-Aska, A., Al-Anazi, A. R., Al-Subaei, S. S., Al-Hedaithy, M. A., Barry, M. A., Somily, A. M., Buba, F., Yusuf, U. & Al Anazi, N. A. (2011). CD4+ T-lymphopenia in HIV negative tuberculous patients at King Khalid University Hospital in Riyadh, Saudi Arabia. European Journal of Medical Research, 16, 285–288.
Nalukwago, S., Lancioni, C. L., Oketcho, J. B., Canaday, D. H. E., Boom, W. H., Ojok, L. & Mayanja-Kizza, H. (2017). The effect of interrupted anti-retroviral treatment on the reconstitution of memory and naive T cells during tuberculosis treatment in HIV patients with active pulmonary tuberculosis. African Health Sciences, 17, 954–962.
Zhang, S. Y., Li, X. B., Hou, S. G., Sun, Y., Shi, Y. R. & Lin, S. S. (2016). Cedrol induces autophagy and apoptotic cell death in A549 non-small cell lung carcinoma cells through the P13K/Akt signaling pathway, the loss of mitochondrial transmembrane potential and the generation of ROS. International Journal of Molecular Medicine, 38, 291–299.
Han, X., Kou, J., Zheng, Y., Liu, Z., Jiang, Y., Gao, Z., Cong, L. & Yang, L. (2019). ROS generated by upconversion nanoparticle-mediated photodynamic therapy induces autophagy via PI3K/AKT/ mTOR signaling pathway in M1 peritoneal macrophage. Cellular Physiology and Biochemistry, 52, 1325–1338.
Kim, G. Y., Jeong, H., Yoon, H. Y., Yoo, H. M., Lee, J. Y., Park, S. H., & Lee, C. E. (2020). Anti-inflammatory mechanisms of suppressors of cytokine signaling target ROS via NRF-2/thioredoxin induction and inflammasome activation in macrophages. BMB Reports, 53, 640–645.
Redza-Dutordoir, M. & Averill-Bates, D. A. (2016). Activation of apoptosis signalling pathways by reactive oxygen species. Biochimica et Biophysica Acta, 1863, 2977–2992.
Subbian, S., Mehta, P. K., Cirillo, S. L., & Cirillo, J. D. (2007). The Mycobacterium marinum mel2 locus displays similarity to bacterial bioluminescence systems and plays a role in defense against reactive oxygen and nitrogen species. BMC Microbiology, 7, 4.
Yabaji, S. M., Mishra, A. K., Chatterjee, A., Dubey, R. K., Srivastava, K. & Srivastava, K. K. (2017). Peroxiredoxin-1 of macrophage is critical for mycobacterial infection and is controlled by early secretory antigenic target protein through the activation of p38 MAPK. Biochemical and Biophysical Research Communications, 494, 433–439.
Yang, Y., Xu, P., He, P., Shi, F., Tang, Y., Guan, C., Zeng, H., Zhou, Y., Song, Q., Zhou, B., Jiang, S., Shao, C., Sun, J., Yang, Y., Wang, X., & Song, H. (2020). Mycobacterial PPE13 activates inflammasome by interacting with the NATCH and LRR domains of NLRP3. The Faseb Journal, 34, 12820–12833.
Wu, M. F., Shu, C. C., Wang, J. Y., Yan, B. S., Lai, H. C., Chiang, B. L., Wu, L. S., & Yu, C. J. (2019). NLRP3 inflammasome is attenuated in patients with Mycobacterium avium complex lung disease and correlated with decreased interleukin-1β response and host susceptibility. Scientific Reports, 9, 12534.
Funding
This work was supported by the National Natural Science Foundation of China (No. 81760357).
Author information
Authors and Affiliations
Contributions
Conceptualization: L.W.; Data curation: S.D., X.W., L.W.; Formal analysis: S.D., X.W.; Methodology: S.D., X.W., F.M., Z.C., X.L., J.G., and X.C.; Software: S.D., X.C.; Supervision: S.D., L.W.; Writing - original draft: S.D., X.W., F.M.; Writing - review & editing: L.W. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ding, S., Wang, X., Ma, F. et al. Effect and Mechanism of Mycobacterium avium MAV-5183 on Apoptosis of Mouse Ana-1 Macrophages. Cell Biochem Biophys (2024). https://doi.org/10.1007/s12013-024-01239-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12013-024-01239-3