Abstract
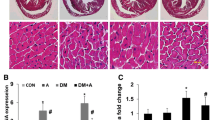
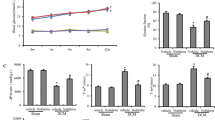
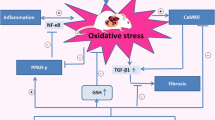
Thiazolidinediones are useful antidiabetic medications. However, their use is associated with adverse side effects like edema, heart failure and bone fractures. In this study, we investigated the anti-ferroptosis effects of suberosin (SBR; a prenylated coumarin) in diabetic Sprague Dawley rats. Further, we assessed the effects of co-administration of SBR (30 and 90 mg/kg/day) with thiazolidinedione (TZ at 15 mg/kg) to mitigate TZ-induced cardiomyopathy in diabetic rats. Our results showed that cardiac output, stroke volume, left ventricle systolic and diastolic pressures were aggravated in diabetic rats treated with TZ alone after 4 weeks. TZ treatments induced ferroptosis as well as marked histoarchitecture disarrangements in rat cardiomyocytes. The study found that optimizing volume overload alleviated cardiac hypertrophy and mitigated left ventricular dysfunction in diabetic rats co-treated with SBR. SBR co-administration with TZ reduced MDA levels in heart tissue and serum iron concentration (biomarkers of ferroptosis), downregulated mRNA expressions of LOX, ACSL4, LPCAT3, and promoted GPX4 activity as well as upregulated mRNA levels of AKT/PI3K/GSK3β as compared to the group administered with TZ at 15 mg/kg. SBR co-administration also helped to retain the normal histoarchitecture of cardiomyocytes in diabetic rats. Hence, our results suggested that SBR is an effective supplement and could be prescribed to diabetic patients along with TZ but this requires further clinical trials.






Similar content being viewed by others
Data Availability
The data is available upon request from the authors.
Abbreviations
- LPCAT3:
-
Lysophosphatidylcholine acyltransferase 3
- ACSL4:
-
Acyl coenzyme A synthetase long-chain member 4
- LOX:
-
Lysyl oxidase
- GPX4:
-
Glutathione peroxidase 4
- AKT:
-
Protein kinase B
- PI3K:
-
Phosphoinositide 3-kinase
- GSK3β:
-
Glycogen synthase kinase-3 beta
- LVDd:
-
Left ventricular diastolic diameter
- LVDs:
-
Left ventricular systolic diameter
- LVEVd:
-
Left ventricular end-diastolic volume
- LVEVs:
-
Left ventricular end-systolic volume
- FS:
-
Fraction shortening
- EF:
-
Ejection fraction
References
Wild, S., Roglic, G., Green, A., Sicree, R., & King, H. (2004). Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care, 27(5), 1047–1053.
Sharma, V., & Patial, V. (2022). Peroxisome proliferator-activated receptor gamma and its natural agonists in the treatment of kidney diseases. Frontiers in Pharmacology, 13, 991059.
Orasanu, G., & Plutzky, J. (2009). The pathologic continuum of diabetic vascular disease. Journal of the American College of Cardiology, 53(5S), S35–S42.
Laing, S. P., Swerdlow, A. J., Slater, S. D., Burden, A. C., Morris, A., Waugh, N. R., Gatling, W., Bingley, P. J., & Patterson, C. C. (2003). Mortality from heart disease in a cohort of 23,000 patients with insulin-treated diabetes. Diabetologia, 46, 760–765.
Juurlink, D. N., Gomes, T., Lipscombe, L. L., Austin, P. C., Hux, J. E., & Mamdani, M. M. (2009). Adverse cardiovascular events during treatment with pioglitazone and rosiglitazone: Population based cohort study. BMJ, 339, b2942.
Nissen, S. E., & Wolski, K. (2007). Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. New England Journal of Medicine, 356(24), 2457–2471.
Lago, R. M., Singh, P. P., & Nesto, R. W. (2007). Congestive heart failure and cardiovascular death in patients with prediabetes and type 2 diabetes given thiazolidinediones: A meta-analysis of randomised clinical trials. The Lancet, 370(9593), 1129–1136.
Rosen, C. J. (2007). The rosiglitazone story—Lessons from an FDA Advisory Committee meeting. New England Journal of Medicine, 357(9), 844–846.
Song, J., Knepper, M. A., Hu, X., Verbalis, J. G., & Ecelbarger, C. A. (2004). Rosiglitazone activates renal sodium-and water-reabsorptive pathways and lowers blood pressure in normal rats. Journal of Pharmacology and Experimental Therapeutics, 308(2), 426–433.
Karalliedde, J., Buckingham, R., Starkie, M., Lorand, D., Stewart, M., Viberti, G., & Rosiglitazone Fluid Retention Study Group. (2006). Effect of various diuretic treatments on rosiglitazone-induced fluid retention. Journal of the American Society of Nephrology, 17(12), 3482–3490.
Rennings, A. J., Russel, F. G., Li, Y., Deen, P. M., Masereeuw, R., Tack, C. J., & Smits, P. (2011). Preserved response to diuretics in rosiglitazone-treated subjects with insulin resistance: A randomized double-blind placebo-controlled crossover study. Clinical Pharmacology & Therapeutics, 89(4), 587–594.
Guo, Y., Zhang, W., Zhou, X., Zhao, S., Wang, J., Guo, Y., Liao, Y., Lu, H., Liu, J., Cai, Y., & Wu, J. (2022). Roles of ferroptosis in cardiovascular diseases. Front Cardiovasc Med, 9, 911564.
Karabulut Uzunçakmak, S., Halıcı, Z., Karakaya, S., Kutlu, Z., Sağlam, Y. S., Bolat, İ., Aydın, P., & Kılıç, C. S. (2023). Suberosin alleviates sepsis-induced lung injury in a rat model of cecal ligation and puncture. Journal of InvestIgatIve surgery, 36(1), 2136802.
Kim, B. H., Kwon, J., Lee, D., & Mar, W. (2015). Neuroprotective effect of demethylsuberosin, a proteasome activator, against MPP+-induced cell death in human neuroblastoma SH-SY5Y cells. Planta Medica Letters, 2(01), e15–e18.
Latha, R. C. R., & Daisy, P. (2011). Insulin-secretagogue, antihyperlipidemic and other protective effects of gallic acid isolated from Terminalia bellerica Roxb. in streptozotocin-induced diabetic rats. Chemico-Biological Interactions, 189(1–2), 112–118.
Walum, E. (1998). Acute oral toxicity. Environmental Health Perspectives, 106(suppl 2), 497–503.
Erdem Guzel, E., Kaya Tektemur, N., Tektemur, A., & Etem Önalan, E. (2021). Carbamazepine-induced renal toxicity may be associated with oxidative stress and apoptosis in male rat. Drug and Chemical Toxicology, 46(1), 1–8.
Chang, C. S., Tsai, P. J., Sung, J. M., Chen, J. Y., Ho, L. C., Pandya, K., Maeda, N., & Tsai, Y. S. (2014). Diuretics prevent thiazolidinedione-induced cardiac hypertrophy without compromising insulin-sensitizing effects in mice. The American Journal of Pathology, 184(2), 442–453.
Ohkawa, H., Ohishi, N., & Yagi, K. (1979). Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical Biochemistry, 95(2), 351–358.
Schiller, N. B., Shah, P. M., Crawford, M., DeMaria, A., Devereux, R., Feigenbaum, H., Gutgesell, H., Reichek, N., Sahn, D., Schnittger, I., & Silverman, N. H. (1989). Recommendations for quantitation of the left ventricle by two-dimensional echocardiography. Journal of the American Society of Echocardiography, 2(5), 358–367.
Benavides-Vallve, C., Corbacho, D., Iglesias-Garcia, O., Pelacho, B., Albiasu, E., Castano, S., Munoz-Barrutia, A., Prosper, F., & Ortiz-de-Solorzano, C. (2012). New strategies for echocardiographic evaluation of left ventricular function in a mouse model of long-term myocardial infarction. PLoS ONE. https://doi.org/10.1371/journal.pone.0041691
Bahari, S., Zeighami, H., Mirshahabi, H., Roudashti, S., & Haghi, F. (2017). Inhibition of Pseudomonas aeruginosa quorum sensing by subinhibitory concentrations of curcumin with gentamicin and azithromycin. Journal of Global Antimicrobial Resistance, 10, 21–28.
Mohammadi, A., Mehrzad, J., Mahmoudi, M., & Schneider, M. (2014). Environmentally relevant level of aflatoxin B1 dysregulates human dendritic cells through signaling on key toll-like receptors. International Journal of Toxicology, 33(3), 175–186.
Rassouli, A., Shihmani, B., Mehrzad, J., & Shokrpoor, S. (2022) The immunomodulatory effect of minocycline on gene expression of inflammation related cytokines in lipopolysaccharide-treated human peripheral blood mononuclear cells. Animal Biotechnology, 1–7.
Wittekind, D. (2003). Traditional staining for routine diagnostic pathology including the role of tannic acid. 1. Value and limitations of the hematoxylin-eosin stain. Biotechnic & Histochemistry, 78(5), 261–270.
Golfakhrabadi, F., Abdollahi, M., Ardakani, M. R. S., Saeidnia, S., Akbarzadeh, T., Ahmadabadi, A. N., Ebrahimi, A., Yousefbeyk, F., Hassanzadeh, A., & Khanavi, M. (2014). Anticoagulant activity of isolated coumarins (suberosin and suberenol) and toxicity evaluation of Ferulago carduchorum in rats. Pharmaceutical Biology, 52(10), 1335–1340.
Xu, J., Zhang, C., & Khanna, A. (2009). Wideband high-frequency echocardiography to evaluate myocardial infarct size. Journal of Ultrasound in Medicine, 28(11), 1527–1534.
Zhou, F., Zhong, W., Xue, J., Gu, Z. L., & Xie, M. L. (2012). Reduction of rat cardiac hypertrophy by osthol is related to regulation of cardiac oxidative stress and lipid metabolism. Lipids, 47(10), 987–994.
Horita, S., Nakamura, M., Satoh, N., Suzuki, M., & Seki, G. (2015). Thiazolidinediones and edema: Recent advances in the pathogenesis of thiazolidinediones-induced renal sodium retention. PPAR Research, 2015, 646423.
Patel, S. S., & Goyal, R. K. (2011). Cardioprotective effects of gallic acid in diabetes-induced myocardial dysfunction in rats. Pharmacognosy research, 3(4), 239.
Xu, S. (2019). Iron and atherosclerosis: The link revisited. Trends in Molecular Medicine, 25(8), 659–661.
Dixon, S. J., Lemberg, K. M., Lamprecht, M. R., Skouta, R., Zaitsev, E. M., Gleason, C. E., Patel, D. N., Bauer, A. J., Cantley, A. M., Yang, W. S., & Morrison, B. (2012). Ferroptosis: An iron-dependent form of nonapoptotic cell death. Cell, 149(5), 1060–1072.
Hua, Z., Ma, K., Liu, S., Yue, Y., Cao, H., & Li, Z. (2020). LncRNA ZEB1-AS1 facilitates ox-LDL-induced damage of HCtAEC cells and the oxidative stress and inflammatory events of THP-1 cells via miR-942/HMGB1 signaling. Life Sciences, 247, 117334.
Liang, R. K., Zhao, Y. Y., Shi, M. L., Zhang, G., Zhao, Y. J., Zhang, B. G., & Liang, R. J. (2021). Skimmin protects diabetic cardiomyopathy in streptozotocin-induced diabetic rats. Kaohsiung Journal of Medical Sciences, 37(2), 136–144.
Cao, J. Y., & Dixon, S. J. (2016). Mechanisms of ferroptosis. Cellular and Molecular Life Sciences, 73(11–12), 2195–2209.
Stockwell, B. R., Angeli, J. P. F., Bayir, H., Bush, A. I., Conrad, M., Dixon, S. J., Fulda, S., Gascón, S., Hatzios, S. K., Kagan, V. E., & Noel, K. (2017). Ferroptosis: A regulated cell death nexus linking metabolism, redox biology, and disease. Cell, 171(2), 273–285.
Doll, S., Proneth, B., Tyurina, Y.Y., Panzilius, E., Kobayashi, S., Ingold, I., Irmler, M., Beckers, J., Aichler, M., Walch, A., & Prokisch, H., (2017). ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition. Nature Chemical Biology, 13(1), 91–98.
Singh, N. K., & Rao, G. N. (2019). Emerging role of 12/15-Lipoxygenase (ALOX15) in human pathologies. Progress in Lipid Research, 73, 28–45.
Anthonymuthu, T. S., Kenny, E. M., Shrivastava, I., Tyurina, Y. Y., Hier, Z. E., Ting, H. C., Dar, H. H., Tyurin, V. A., Nesterova, A., Amoscato, A. A., & Mikulska-Ruminska, K. (2018). Empowerment of 15-lipoxygenase catalytic competence in selective oxidation of membrane ETE-PE to ferroptotic death signals, HpETE-PE. Journal of the American Chemical Society, 140(51), 17835–17839.
Dixon, S. J., Winter, G. E., Musavi, L. S., Lee, E. D., Snijder, B., Rebsamen, M., Superti-Furga, G., & Stockwell, B. R. (2015). Human haploid cell genetics reveals roles for lipid metabolism genes in nonapoptotic cell death. ACS Chemical Biology, 10(7), 1604–1609.
Li, Z., Jiang, H., Ding, T., Lou, C., Bui, H. H., Kuo, M. S., & Jiang, X. C. (2015). Deficiency in lysophosphatidylcholine acyltransferase 3 reduces plasma levels of lipids by reducing lipid absorption in mice. Gastroenterology, 149(6), 1519–1529.
Yang, C., Ma, X., Wang, Z., Zeng, X., Hu, Z., Ye, Z., & Shen, G. (2017). Curcumin induces apoptosis and protective autophagy in castration-resistant prostate cancer cells through iron chelation. Drug Design, Development and Therapy, 11, 431–439.
de Oliveira, M. R., Ferreira, G. C., Schuck, P. F., & Dal Bosco, S. M. (2015). Role for the PI3K/Akt/Nrf2 signaling pathway in the protective effects of carnosic acid against methylglyoxal-induced neurotoxicity in SH-SY5Y neuroblastoma cells. Chemico-Biological Interactions, 242, 396–406.
Funding
No funding available.
Author information
Authors and Affiliations
Contributions
SI: Investigation, writing of original draft; FJ: supervision, validation and writing; IK: data analysis; TO: review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The author declares no competing interests.
Ethical Approval
The animal handling and treatment experimental protocols underwent review and monitoring by the ethical committee of the Government College University, Faisalabad, Pakistan. These protocols followed the approved protocol of the European Union of animal care and Experimentation (CEE Council 86/609). The research was conducted in adherence to the ARRIVE guidelines.
Additional information
Handling Editor: Lu Cai.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Iqbal, S., Jabeen, F., Kahwa, I. et al. Suberosin Alleviates Thiazolidinedione-Induced Cardiomyopathy in Diabetic Rats by Inhibiting Ferroptosis via Modulation of ACSL4-LPCAT3 and PI3K-AKT Signaling Pathways. Cardiovasc Toxicol 23, 295–304 (2023). https://doi.org/10.1007/s12012-023-09804-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-023-09804-7