Abstract
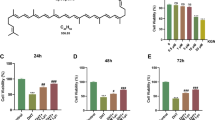
Polycystic ovarian syndrome (PCOS) is an endocrine syndrome in women of reproductive age. Berberine (BBR) is a Chinese herbal monomer that exhibits many pharmacological properties related to PCOS treatment. This study aims to analyze the effect of BBR on a cell model of PCOS and the underlying mechanism. Human ovarian granulosa (KGN) cells were treated with dihydrotestosterone (DHT) to mimic a PCOS cell model. The RNA expression of circ_0097636, miR-186-5p, and sirtuin3 (SIRT3) was determined by quantitative real-time polymerase chain reaction (qRT-PCR). Protein expression was detected by western blotting. Cell viability was analyzed by CCK-8 assay. Cell proliferation and apoptosis were investigated by 5-ethynyl-2′-deoxyuridine (EdU) assay and flow cytometry assay, respectively. The levels of interleukin-6 (IL-6), IL-1β, and tumor necrosis factor-α (TNF-α) were analyzed by enzyme-linked immunosorbent assays (ELISAs). Fe2+ concentration was assessed by an iron assay kit. Oxidative stress was assessed by detecting reactive oxygen species (ROS) level and malondialdehyde (MDA) level using commercial kits. The association of miR-186-5p with circ_0097636 and SIRT3 was identified by dual-luciferase reporter assay and RNA pull-down assay. Circ_0097636 expression was downregulated in the follicular fluid of PCOS patients and DHT-treated KGN cells when compared with control groups. BBR treatment partially relieved the DHT-induced inhibitory effect on cell proliferation and promoted effects on cell apoptosis, inflammation, ferroptosis, and oxidative stress in KGN cells. Additionally, circ_0097636 bound to miR-186-5p, and SIRT3 was identified as a target gene of miR-186-5p in KGN cells. BBR treatment ameliorated DHT-induced KGN cell injury by upregulating circ_0097636 and SIRT3 expression and downregulating miR-186-5p expression. Moreover, circ_0097636 overexpression protected KGN cells from DHT-induced injury by increasing SIRT3 expression. BBR ameliorated DHT-induced KGN cell injury and ferroptosis by regulating the circ_0097636/miR-186-5p/SIRT3 pathway.









Similar content being viewed by others
Data Availability
Not applicable
References
Ganie, M. A., Vasudevan, V., Wani, I. A., Baba, M. S., Arif, T., & Rashid, A. (2019). Epidemiology, pathogenesis, genetics & management of polycystic ovary syndrome in India. The Indian Journal of Medical Research, 150(4), 333–344.
Aversa, A., La Vignera, S., Rago, R., Gambineri, A., Nappi, R. E., Calogero, A. E., & Ferlin, A. (2020). Fundamental concepts and novel aspects of polycystic ovarian syndrome: Expert consensus resolutions. Frontiers in Endocrinology, 11, 516.
Palomba, S., Santagni, S., Falbo, A., & La Sala, G. B. (2015). Complications and challenges associated with polycystic ovary syndrome: Current perspectives. International Journal of Women's Health, 7, 745–763.
Wang, J., Wu, D., Guo, H., & Li, M. (2019). Hyperandrogenemia and insulin resistance: The chief culprit of polycystic ovary syndrome. Life Sciences, 236, 116940.
Ionescu, O. M., Frincu, F., Mehedintu, A., Plotogea, M., Cirstoiu, M., Petca, A., Varlas, V., & Mehedintu, C. (2023). Berberine-A promising therapeutic approach to polycystic ovary syndrome in infertile/pregnant women. Life, 13(1), 125.
Ramezani Tehrani, F., & Amiri, M. (2019). Polycystic ovary syndrome in adolescents: Challenges in diagnosis and treatment. International Journal of Endocrinology and Metabolism, 17(3), e91554.
Wang, K., Feng, X., Chai, L., Cao, S., & Qiu, F. (2017). The metabolism of berberine and its contribution to the pharmacological effects. Drug Metabolism Reviews, 49(2), 139–157.
Zhang, S. W., Zhou, J., Gober, H. J., Leung, W. T., & Wang, L. (2021). Effect and mechanism of berberine against polycystic ovary syndrome. Biomedicine & Pharmacotherapy, 138, 111468.
Rondanelli, M., Infantino, V., Riva, A., Petrangolini, G., Faliva, M. A., Peroni, G., Naso, M., Nichetti, M., Spadaccini, D., Gasparri, C., et al. (2020). Polycystic ovary syndrome management: A review of the possible amazing role of berberine. Archives of Gynecology and Obstetrics, 301(1), 53–60.
Zhou, W. Y., Cai, Z. R., Liu, J., Wang, D. S., Ju, H. Q., & Xu, R. H. (2020). Circular RNA: Metabolism, functions and interactions with proteins. Molecular Cancer, 19(1), 172.
Verduci, L., Tarcitano, E., Strano, S., Yarden, Y., & Blandino, G. (2021). CircRNAs: Role in human diseases and potential use as biomarkers. Cell Death & Disease, 12(5), 468.
Wang, Y., Mo, Y., Peng, M., Zhang, S., Gong, Z., Yan, Q., Tang, Y., He, Y., Liao, Q., Li, X., et al. (2022). The influence of circular RNAs on autophagy and disease progression. Autophagy, 18(2), 240–253.
Mu, L., Sun, X., Tu, M., & Zhang, D. (2021). Non-coding RNAs in polycystic ovary syndrome: A systematic review and meta-analysis. Reproductive Biology and Endocrinology, 19(1), 10.
Chen, A. X., Jin, R. Y., Zhou, W. M., Ye, Y. J., Lu, J. L., Ren, Y. F., & Xuan, F. L. (2022). CircRNA circ_0043533 facilitates cell growth in polycystic ovary syndrome by targeting miR-1179. Reproductive Biology, 22(2), 100637.
Lu, X., Gao, H., Zhu, B., & Lin, G. (2021). Circular RNA circ_RANBP9 exacerbates polycystic ovary syndrome via microRNA-136-5p/XIAP axis. Bioengineered, 12(1), 6748–6758.
Ma, Z., Zhao, H., Zhang, Y., Liu, X., & Hao, C. (2019). Novel circular RNA expression in the cumulus cells of patients with polycystic ovary syndrome. Archives of Gynecology and Obstetrics, 299(6), 1715–1725.
Xiong, D. D., Feng, Z. B., Lai, Z. F., Qin, Y., Liu, L. M., Fu, H. X., He, R. Q., Wu, H. Y., Dang, Y. W., Chen, G., et al. (2019). High throughput circRNA sequencing analysis reveals novel insights into the mechanism of nitidine chloride against hepatocellular carcinoma. Cell Death & Disease, 10(9), 658.
Wang, T., Liu, Y., Lv, M., Xing, Q., Zhang, Z., He, X., Xu, Y., Wei, Z., & Cao, Y. (2019). miR-323-3p regulates the steroidogenesis and cell apoptosis in polycystic ovary syndrome (PCOS) by targeting IGF-1. Gene, 683, 87–100.
Li, W., Li, D., Kuang, H., Feng, X., Ai, W., Wang, Y., Shi, S., Chen, J., & Fan, R. (2020). Berberine increases glucose uptake and intracellular ROS levels by promoting Sirtuin 3 ubiquitination. Biomedicine & Pharmacotherapy, 121, 109563.
Zhang, F., Ma, T., Cui, P., Tamadon, A., He, S., Huo, C., Yierfulati, G., Xu, X., Hu, W., Li, X., et al. (2019). Diversity of the gut microbiota in dihydrotestosterone-induced PCOS rats and the pharmacologic effects of diane-35, probiotics, and berberine. Frontiers in Microbiology, 10, 175.
Wang, Z., Nie, K., Su, H., Tang, Y., Wang, H., Xu, X., & Dong, H. (2021). Berberine improves ovulation and endometrial receptivity in polycystic ovary syndrome. Phytomedicine, 91, 153654.
Shen, H. R., Xu, X., & Li, X. L. (2021). Berberine exerts a protective effect on rats with polycystic ovary syndrome by inhibiting the inflammatory response and cell apoptosis. Reproductive Biology and Endocrinology, 19(1), 3.
Shen, H. R., Xu, X., Ye, D., & Li, X. L. (2021). Berberine improves the symptoms of DHEA-induced PCOS rats by regulating gut microbiotas and metabolites. Gynecologic and Obstetric Investigation, 86(4), 388–397.
Zhang, J., Ding, N., Xin, W., Yang, X., & Wang, F. (2022). Quantitative proteomics reveals that a prognostic signature of the endometrium of the polycystic ovary syndrome women based on ferroptosis proteins. Frontiers in Endocrinology, 13, 871945.
Vitale, S. G., Fulghesu, A. M., Mikuš, M., Watrowski, R., D'Alterio, M. N., Lin, L. T., Shah, M., Reyes-Muñoz, E., Sathyapalan, T., & Angioni, S. (2022). The translational role of miRNA in polycystic ovary syndrome: From bench to bedside-A systematic literature review. Biomedicines, 10(8), 1816.
Javadi, M., Rad, J. S., Farashah, M. S. G., & Roshangar, L. (2022). An insight on the role of altered function and expression of exosomes and microRNAs in female reproductive diseases. Reproductive Sciences, 29(5), 1395–1407.
Tan, W., Dai, F., Yang, D., Deng, Z., Gu, R., Zhao, X., & Cheng, Y. (2022). MiR-93-5p promotes granulosa cell apoptosis and ferroptosis by the NF-kB signaling pathway in polycystic ovary syndrome. Frontiers in Immunology, 13, 967151.
Wei, J., & Zhao, Y. (2020). MiR-185-5p protects against angiogenesis in polycystic ovary syndrome by targeting VEGFA. Frontiers in Pharmacology, 11, 1030.
Song, Y., Yu, G., Xiang, Y., Li, Y., Wan, L., & Tan, L. (2019). Altered miR-186 and miR-135a contribute to granulosa cell dysfunction by targeting ESR2: A possible role in polycystic ovary syndrome. Molecular and Cellular Endocrinology, 494, 110478.
Singh, C. K., Chhabra, G., Ndiaye, M. A., Garcia-Peterson, L. M., Mack, N. J., & Ahmad, N. (2018). The role of sirtuins in antioxidant and redox signaling. Antioxidants & Redox Signaling, 28(8), 643–661.
Haigis, M. C., & Sinclair, D. A. (2010). Mammalian sirtuins: Biological insights and disease relevance. Annual Review of Pathology, 5, 253–295.
Zhang, L., Han, L., Ma, R., Hou, X., Yu, Y., Sun, S., Xu, Y., Schedl, T., Moley, K. H., & Wang, Q. (2015). Sirt3 prevents maternal obesity-associated oxidative stress and meiotic defects in mouse oocytes. Cell Cycle (Georgetown, Texas), 14(18), 2959–2968.
Yu, W., Dittenhafer-Reed, K. E., & Denu, J. M. (2012). SIRT3 protein deacetylates isocitrate dehydrogenase 2 (IDH2) and regulates mitochondrial redox status. The Journal of Biological Chemistry, 287(17), 14078–14086.
Bause, A. S., & Haigis, M. C. (2013). SIRT3 regulation of mitochondrial oxidative stress. Experimental Gerontology, 48(7), 634–639.
Zhang, Q., Ren, J., Wang, F., Li, M., Pan, M., Zhang, H., & Qu, F. (2023). Chinese herbal medicine alleviates the pathogenesis of polycystic ovary syndrome by improving oxidative stress and glucose metabolism via mitochondrial Sirtuin 3 signaling. Phytomedicine, 109, 154556.
Zhang, Q., Ren, J., Wang, F., Pan, M., Cui, L., Li, M., & Qu, F. (2022). Mitochondrial and glucose metabolic dysfunctions in granulosa cells induce impaired oocytes of polycystic ovary syndrome through Sirtuin 3. Free Radical Biology & Medicine, 187, 1–16.
Funding
This work was supported by the Fund Program for the Scientific Activities of Selected Returned Overseas Professionals in Shanxi Province (No. 20230055) and the Fund Program for Research Project Supported by Shanxi Scholarship Council of China (No. 2023-183).
Author information
Authors and Affiliations
Contributions
SW designed and performed the research; YW, QQ, JfL, QC, YZ, XL, and JrL analyzed the data; SW wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Written informed consents were obtained from all participants and this study was permitted by the Ethics Committee of Fifth Hospital of Shanxi Medical University.
Consent for Publication
Not applicable
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, S., Wang, Y., Qin, Q. et al. Berberine Protects Against Dihydrotestosterone-Induced Human Ovarian Granulosa Cell Injury and Ferroptosis by Regulating the Circ_0097636/MiR-186-5p/SIRT3 Pathway. Appl Biochem Biotechnol (2023). https://doi.org/10.1007/s12010-023-04825-y
Accepted:
Published:
DOI: https://doi.org/10.1007/s12010-023-04825-y