Abstract
Purpose of Review
The aim of this paper is to discuss strategies for prevention and management of hyperkalemia in patients with heart failure, including the role of novel therapies.
Recent Findings
Renin-angiotensin-aldosterone system (RAAS) antagonists, including angiotensin-converting enzyme inhibitors (ACEI), angiotensin receptor blockers (ARB), and mineralocorticoid receptor antagonists (MRA) decrease mortality and morbidity in heart failure but increase the risk of hyperkalemia, especially when used in combination. Prevention of hyperkalemia and its associated complications requires careful patient selection, counseling regarding dietary potassium intake, awareness of drug interactions, and regular laboratory surveillance. Recent data suggests that the risk of hyperkalemia may be further moderated through the use of combined angiotensin-neprilysin inhibitors, novel MRAs, and novel potassium binding agents.
Summary
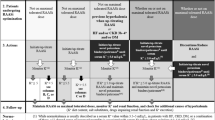
Clinicians should be mindful of the risk of hyperkalemia when prescribing RAAS inhibitors to patients with heart failure. In patients at highest risk, such as those with diabetes, the elderly, and advanced chronic kidney disease, more intensive laboratory surveillance of potassium and creatinine may be required. Novel therapies hold promise for reducing the risk of hyperkalemia and enhancing the tolerability of RAAS antagonists.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
•• Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, Colvin MM, et al. 2016 ACC/AHA/HFSA focused update on new pharmacological therapy for heart failure: an update of the 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. J Am Coll Cardiol. 2016;68:1476–88. This update to the AHA guidelines for heart failure management incorporates the use of angiotensin-receptor neprilysin inhibitors into the clinical strategies for patients with stage C heart failure with reduced ejection fraction
Granger CB, McMurray JJV, Yusuf S, Held P, Michelson EL, Olofsson B, et al. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function intolerant to angiotensin-converting-enzyme inhibitors: the CHARM-Alternative trial. Lancet. 2003;362:772–6.
Cohn JN, Tognoni G. Valsartan heart failure trial investigators. A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure N Engl J Med. 2001;345:1667–75.
Zannad F, McMurray JJV, Krum H, van Veldhuisen DJ, Swedberg K, Shi H, et al. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med. 2011;364:11–21.
Pitt B, Zannad F, Remme WJ, Cody R, Castaigne A, Perez A, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation study investigators. N Engl J Med. 1999;341:709–17.
Pfeffer MA, Braunwald E, Moyé LA, Basta L, Brown EJ, Cuddy TE, et al. Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction: results of the survival and ventricular enlargement trial. The SAVE investigators. N Engl J Med. 1992;327:669–77.
The SOLVD investigators. Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. The SOLVD Investigators. N Engl J Med. 1991;325:293–302.
Palmer BF. Managing hyperkalemia caused by inhibitors of the renin-angiotensin-aldosterone system. N Engl J Med. 2004;351:585–92.
Reardon LC, Macpherson DS. Hyperkalemia in outpatients using angiotensin-converting enzyme inhibitors. How much should we worry? Arch Intern Med. 1998;158:26–32.
Sarwar CMS, Papadimitriou L, Pitt B, Piña I, Zannad F, Anker SD, et al. Hyperkalemia in heart failure. J Am Coll Cardiol. 2016;68:1575–89.
Luo J, Brunelli SM, Jensen DE, Yang A. Association between serum potassium and outcomes in patients with reduced kidney function. Clin J Am Soc Nephrol. 2016;11:90–100.
McMurray JJV, Ostergren J, Swedberg K, Granger CB, Held P, Michelson EL, et al. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function taking angiotensin-converting-enzyme inhibitors: the CHARM-Added trial. Lancet. 2003;362:767–71.
Pfeffer MA, Swedberg K, Granger CB, Held P, McMurray JJV, Michelson EL, et al. Effects of candesartan on mortality and morbidity in patients with chronic heart failure: the CHARM-Overall programme. Lancet. 2003;362:759–66.
Yusuf S, Pfeffer MA, Swedberg K, Granger CB, Held P, McMurray JJV, et al. Effects of candesartan in patients with chronic heart failure and preserved left-ventricular ejection fraction: the CHARM-Preserved trial. Lancet. 2003;362:777–81.
Desai AS, Swedberg K, McMurray JJV, Granger CB, Yusuf S, Young JB, et al. Incidence and predictors of hyperkalemia in patients with heart failure: an analysis of the CHARM Program. J Am Coll Cardiol. 2007;50:1959–66.
McMurray JJV, Krum H, Abraham WT, Dickstein K, Køber LV, Desai AS, et al. Aliskiren, Enalapril, or Aliskiren and Enalapril in heart failure. N Engl J Med. 2016;374:1521–32.
Desai AS. Hyperkalemia in patients with heart failure: incidence, prevalence, and management. Curr Heart Fail Rep. 2009;6:272–80.
Vardeny O, Claggett B, Anand I, Rossignol P, Desai AS, Zannad F, et al. Incidence, predictors, and outcomes related to hypo- and hyperkalemia in patients with severe heart failure treated with a mineralocorticoid receptor antagonist. Circ Heart Fail. 2014;7:573–9.
Pitt B, Remme W, Zannad F, Neaton J, Martinez F, Roniker B, et al. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med. 2003;348:1309–21.
Juurlink DN, Mamdani MM, Lee DS, Kopp A, Austin PC, Laupacis A, et al. Rates of hyperkalemia after publication of the randomized Aldactone Evaluation study. N Engl J Med. 2004;351:543–51.
Ramadan FH, Masoodi N, El-Solh AA. Clinical factors associated with hyperkalemia in patients with congestive heart failure. J Clin Pharm Ther. 2005;30:233–9.
Jain N, Kotla S, Little BB, Weideman RA, Brilakis ES, Reilly RF, et al. Predictors of hyperkalemia and death in patients with cardiac and renal disease. Am J Cardiol. 2012;109:1510–3.
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, Drazner MH, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines. J Am Coll Cardiol. 2013;62:e147–239.
Aldactone [Internet]. FDA; Available from: http://www.accessdata.fda.gov/drugsatfda_docs/label/2008/012151s062lbl.pdf
Ray K, Dorman S, Watson R. Severe hyperkalaemia due to the concomitant use of salt substitutes and ACE inhibitors in hypertension: a potentially life threatening interaction. J Hum Hypertens. 1999;13:717–20.
Pantanowitz L. Drug-induced hyperkalemia. Am J Med. 2002;112:334–5.
Cheng TO. Herbal interactions with cardiac drugs. Arch Intern Med. 2000;160:870–1.
Perazella MA. Trimethoprim-induced hyperkalaemia: clinical data, mechanism, prevention and management. Drug Saf. 2000;22:227–36.
Kleyman TR, Roberts C, Ling BN. A mechanism for pentamidine-induced hyperkalemia: inhibition of distal nephron sodium transport. Ann Intern Med. 1995;122:103–6.
Banhara PB, Gonçalves RT, Rocha PT, Delgado AG, Leite M, Gomes CP. Tubular dysfunction in renal transplant patients using sirolimus or tacrolimus. Clin Nephrol. 2015;83:331–7.
Oster JR, Singer I, Fishman LM. Heparin-induced aldosterone suppression and hyperkalemia. Am J Med. 1995;98:575–86.
•• McMurray JJV, Packer M, Desai AS, Gong J, Lefkowitz MP, Rizkala AR, et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014;371:993–1004. The PARADIGM-HF study demonstrated that the angiotensin receptor-neprilysin inhibitor LCZ696 was superior to enalapril in reducing heart failure hospitalizations and mortality
• Desai AS, Vardeny O, Claggett B, McMurray JJV, Packer M, Swedberg K, et al. Reduced risk of hyperkalemia during treatment of heart failure with mineralocorticoid receptor antagonists by use of sacubitril/valsartan compared with enalapril: a secondary analysis of the PARADIGM-HF trial. JAMA Cardiol. 2016; This secondary analysis of PARADIGM-HF showed that there is a reduced risk of hyperkalemia in patients on ARNI and MRA compared to enalapril and MRA
Bärfacker L, Kuhl A, Hillisch A, Grosser R, Figueroa-Pérez S, Heckroth H, et al. Discovery of BAY 94-8862: a nonsteroidal antagonist of the mineralocorticoid receptor for the treatment of cardiorenal diseases. ChemMedChem. 2012;7:1385–403.
Kolkhof P, Delbeck M, Kretschmer A, Steinke W, Hartmann E, Bärfacker L, et al. Finerenone, a novel selective nonsteroidal mineralocorticoid receptor antagonist protects from rat cardiorenal injury. J Cardiovasc Pharmacol. 2014;64:69–78.
Naegele M, Hernandez AF, Ruschitzka F. Finerenone in heart failure: walking a fine line. Eur Heart J. 2016;37:2115–7.
Pitt B, Filippatos G, Gheorghiade M, Kober L, Krum H, Ponikowski P, et al. Rationale and design of ARTS: a randomized, double-blind study of BAY 94-8862 in patients with chronic heart failure and mild or moderate chronic kidney disease. Eur J Heart Fail. 2012;14:668–75.
Pitt B, Kober L, Ponikowski P, Gheorghiade M, Filippatos G, Krum H, et al. Safety and tolerability of the novel non-steroidal mineralocorticoid receptor antagonist BAY 94-8862 in patients with chronic heart failure and mild or moderate chronic kidney disease: a randomized, double-blind trial. Eur Heart J. 2013;34:2453–63.
Pitt B, Anker SD, Böhm M, Gheorghiade M, Køber L, Krum H, et al. Rationale and design of MinerAlocorticoid receptor antagonist tolerability study-heart failure (ARTS-HF): a randomized study of finerenone vs. eplerenone in patients who have worsening chronic heart failure with diabetes and/or chronic kidney disease. Eur J Heart Fail. 2015;17:224–32.
Filippatos G, Anker SD, Böhm M, Gheorghiade M, Køber L, Krum H, et al. A randomized controlled study of finerenone vs. eplerenone in patients with worsening chronic heart failure and diabetes mellitus and/or chronic kidney disease. Eur Heart J. 2016;37:2105–14.
Lepage L, Desforges K, Lafrance J-P. New drugs to prevent and treat hyperkalemia. Curr Opin Nephrol Hypertens. 2016;25:524–8.
Linder KE, Krawczynski MA, Laskey D. Sodium zirconium Cyclosilicate (ZS-9): a novel agent for the treatment of hyperkalemia. Pharmacotherapy. 2016;36:923–33.
Scherr L, Ogden DA, Mead AW, Spritz N, Rubin AL. Management of hyperkalemia with a cation-exchange resin. N Engl J Med. 1961;264:115–9.
Sterns RH, Grieff M, Bernstein PL. Treatment of hyperkalemia: something old, something new. Kidney Int. 2016;89:546–54.
Flinn RB, Merrill JP, Welzant WR. Treatment of the oliguric patient with a new sodium-exchange resin and sorbitol; a preliminary report. N Engl J Med. 1961;264:111–5.
Sterns RH, Rojas M, Bernstein P, Chennupati S. Ion-exchange resins for the treatment of hyperkalemia: are they safe and effective? J Am Soc Nephrol. 2010;21:733–5.
Ayoub I, Oh MS, Gupta R, McFarlane M, Babinska A, Salifu MO. Colon necrosis due to sodium polystyrene sulfonate with and without sorbitol: an experimental study in rats. PLoS One. 2015;10:e0137636.
Rogers FB, Li SC. Acute colonic necrosis associated with sodium polystyrene sulfonate (Kayexalate) enemas in a critically ill patient: case report and review of the literature. J Trauma. 2001;51:395–7.
Bomback AS, Woosley JT, Kshirsagar AV .Colonic necrosis due to sodium polystyrene sulfate (Kayexalate). Am J Emerg Med. 2009;27:753.e1–2.
Harel Z, Harel S, Shah PS, Wald R, Perl J, Bell CM. Gastrointestinal adverse events with sodium polystyrene sulfonate (Kayexalate) use: a systematic review. Am J Med. 2013;126:264.e9–24.
Abraham SC, Bhagavan BS, Lee LA, Rashid A, Wu TT. Upper gastrointestinal tract injury in patients receiving kayexalate (sodium polystyrene sulfonate) in sorbitol: clinical, endoscopic, and histopathologic findings. Am J Surg Pathol. 2001;25:637–44.
Goutorbe P, Montcriol A, Lacroix G, Bordes J, Meaudre E, Souraud J-B. Intestinal necrosis associated with orally administered calcium polystyrene sulfonate without sorbitol. Ann Pharmacother. 2011;45:e13.
Lillemoe KD, Romolo JL, Hamilton SR, Pennington LR, Burdick JF, Williams GM. Intestinal necrosis due to sodium polystyrene (Kayexalate) in sorbitol enemas: clinical and experimental support for the hypothesis. Surgery. 1987;101:267–72.
Weir MR, Bakris GL, Bushinsky DA, Mayo MR, Garza D, Stasiv Y, et al. Patiromer in patients with kidney disease and hyperkalemia receiving RAAS inhibitors. N Engl J Med. 2015;372:211–21.
Pitt B, Anker SD, Bushinsky DA, Kitzman DW, Zannad F, Huang I-Z, et al. Evaluation of the efficacy and safety of RLY5016, a polymeric potassium binder, in a double-blind, placebo-controlled study in patients with chronic heart failure (the PEARL-HF) trial. Eur Heart J. 2011;32:820–8.
• Pitt B, Bakris GL, Bushinsky DA, Garza D, Mayo MR, Stasiv Y, et al. Effect of patiromer on reducing serum potassium and preventing recurrent hyperkalaemia in patients with heart failure and chronic kidney disease on RAAS inhibitors. Eur J heart Fail. 2015;17:1057–65. This study demonstrates reductions in serum potassium and the risk of hyperkalemia with use of patiromer among heart failure patients with chronic kidney disease treated with RAAS inhibitors
FDA approves new drug to treat hyperkalemia [Internet]. 2015. Available from: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm468546.htm
Relypsa Announces Results from Veltassa Drug-Drug Interaction Studies in Healthy Volunteers [Internet]. Redwood City, CA: Relypsa, Inc; Available from: http://investor.relypsa.com/releasedetail.cfm?releaseid=951581
Stavros F, Yang A, Leon A, Nuttall M, Rasmussen HS. Characterization of structure and function of ZS-9, a K+ selective ion trap. PLoS One. 2014;9:e114686.
Kosiborod M, Rasmussen HS, Lavin P, Qunibi WY, Spinowitz B, Packham D, et al. Effect of sodium zirconium cyclosilicate on potassium lowering for 28 days among outpatients with hyperkalemia: the HARMONIZE randomized clinical trial. JAMA. 2014;312:2223–33.
• Anker SD, Kosiborod M, Zannad F, Piña IL, McCullough PA, Filippatos G, et al. Maintenance of serum potassium with sodium zirconium cyclosilicate (ZS-9) in heart failure patients: results from a phase 3 randomized, double-blind, placebo-controlled trial. Eur J Heart Fail. 2015;17:1050–6. This sub-study of HARMONIZE showed that heart failure patients with baseline hyperkalemia treated with ZS-9 were able to maintain normokalemia.
Massie BM, Carson PE, McMurray JJ, Komajda M, McKelvie R, Zile MR, et al. Irbesartan in patients with heart failure and preserved ejection fraction. N Engl J Med. 2008;359:2456–67.
Pitt B, Pfeffer MA, Assmann SF, Boineau R, Anand IS, Claggett B, et al. Spironolactone for heart failure with preserved ejection fraction. N Engl J Med. 2013;370:13.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Ersilia M. DeFilippis declares no conflict of interest.
Akshay S. Desai has received research grants from Novartis and has been a consultant to Relypsa, Novartis, St. Jude Medical, Janssen, Sanofi, AstraZeneca, and Merck.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Pathophysiology of Myocardial Failure
Rights and permissions
About this article
Cite this article
DeFilippis, E.M., Desai, A.S. Treatment of Hyperkalemia in Heart Failure. Curr Heart Fail Rep 14, 266–274 (2017). https://doi.org/10.1007/s11897-017-0341-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11897-017-0341-0