Abstract
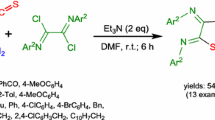
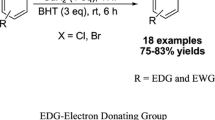
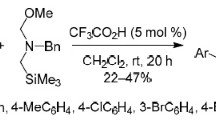
A new efficient method for the synthesis of isothiocyanates has been developed via the replacement reaction of phenyl isothiocyanate and the corresponding amines (the amino group of these amines was linked to tertiary carbon or secondary carbon) with dimethylbenzene as solvent. This reaction was carried out under the protection of nitrogen and mild condition. In addition, the yields of some products could be more than 90%. More importantly, this method has advantages with low toxicity, low cost, safety, less by-products and simple to operate. It has the potential to realize the industrial production of some complicated isothiocyanates.




Similar content being viewed by others
References
Banert K, Hagedorn M, Muller A (2001) Synthesis of new vinyl thiocyanates by [3,3] sigmatropic rearrangement of isothiocyanates. Eur J Org Chem 2001:1089–1103. https://doi.org/10.1002/1099-0690(200103)2001:6%3c1089::AID-EJOC1089%3e3.0.CO;2-N
Chen X, Li Z, Sun X, Ma HZ (2011) New method for the synthesis of sulforaphane and related isothiocyanates. Synthesis 2011:3991–3996. https://doi.org/10.1055/s-0031-1289601
Fechner J, Kaufmann M, Herz C, Eisenschmidt D, Lamy E, Kroh LW, Hanschen FS (2018) The major glucosinolate hydrolysis product in rocket (Eruca sativa L.), sativin, is 1,3-thiazepane-2-thione: elucidation of structure, bioactivity, and stability compared to other rocket isothiocyanates. Food Chem 261:57–65. https://doi.org/10.1016/j.foodchem.2018.04.023
Li ZY, Ma HZ, Han C, Xi H, Meng Q, Chen X, Sun X (2013) Synthesis of isothiocyanates by reaction of amines with phenyl chlorothionoformate via one-pot or two-step process. Synthesis 45:1667–1674. https://doi.org/10.1055/s-0033-1338744
Li ZY, Shi S, Zhuang Y, Zheng CQ, Zhang JL, Sun XQ (2015) Improvement of the synthesis of isothiocyanates. Chin J Org Chem 35:1999–2003. https://doi.org/10.6023/cjoc201503026
Márquez JM, López O, Maya L, Fuentes J, Fernández-Bolaños JG (2008) Fernández-Bolaños. Taurine isothiocyanate: a versatile intermediate for the preparation of ureas, thioureas, and guanidines. Taur Deriv Cyclodext Tetrahedron Lett 49:3912–3915. https://doi.org/10.1016/j.tetlet.2008.04.053
Murata Y, Matsumoto N, Miyata M, Kitamura Y, Kakusawa N, Matsumura M, Yasuike S (2018) One-pot reaction for the synthesis of N-substituted 2-aminobenzoxazoles using triphenylbismuth dichloride as cyclodesulfurization reagent. J Organomet Chem 859:18–23. https://doi.org/10.1016/j.jorganchem.2018.01.041
Pilipczuk T, Kusznierewicz B, Chmiel T, Przychodzen W, Bartoszek A (2017) Simultaneous determination of individual isothiocyanates in plant samples by HPLC-DAD-MS following SPE and derivatization with N-acetyl-l-cysteine. Food Chem 214:587–596. https://doi.org/10.1016/j.foodchem.2016.07.125
Sarah ML, Ya L, Michael JC, Laciak AR, Singh H, Seiner DR, Reilly TJ, Tanner JJ, Gates KS (2015) Inactivation of protein tyrosine phosphatases by dietary isothiocyanates. Bioorg Med Chem Lett 25:4549–4552. https://doi.org/10.1016/j.bmcl.2015.08.065
Srisa J, Tankam T, Sukwattanasinitt M, Wacharasindhu S (2019) Micelle-enabled one-pot guanidine synthesis in water directly from isothiocyanate using hypervalent iodine(III) reagents under mild conditions. Chem Asian J 14:3335–3343. https://doi.org/10.1002/asia.201900982
Sun N, Li B, Shao J, Mo W, Hu B, Shen Z, Hu X (2012) A general and facile one-pot process of isothiocyanates from amines under aqueous conditions. Beilstein J Org Chem 8:61–70. https://doi.org/10.3762/bjoc.8.6
Vagapova LI, Sharafutdinova DR, Burilov AR, Mustafina AR, Babaev VM, Rizvanov IK, Pudovik M (2015) A Synthesis and properties of new thiophosphorylated thioureas containing a trialkoxysilyl group. Russ J Org Chem 51:309–312. https://doi.org/10.1134/s1070428015030033
Xiao D, Powolny AA, Singh SV (2008) Benzyl isothiocyanate targets mitochondrial respiratory chain to trigger reactive oxygen species-dependent apoptosis in human breast cancer cells. J Biol Chem 283:30151–30163. https://doi.org/10.1074/jbc.m802529200
Yella R, Ghosh H, Murru S, Sahoo SK, Patel BK (2010) Efficient preparation of isothiocyanates from dithiocarbamates using bromineless brominating reagent. Synth Commun 40:2083–2096. https://doi.org/10.1080/00397910903219476
Yildiz IN, Orucemre EE, Tasdemir D, Karakucukiyidogan A, Ulasli M, Bayram H (2017) Design and synthesis of novel thioureas derived from 4-(4-Fluorophenoxy)aniline as anticancer agents: novel thiourea derivatives from 4-(4-Fluorophenoxy) aniline. J Chin Chem Soc 64:321–330. https://doi.org/10.1002/jccs.201600193
Zhang B, Shi L (2019) One-pot synthesis of isothiocyanates. Chin J Appl Chem 36:892–896
Zhang X, Lee YK, Kelley JA, Burke TR (2000) Preparation of aryl isothiocyanates via protected phenylthiocarbamates and application to the synthesis of caffeic acid (4-isothiocyanato) phenyl ester. J Org Chem 65:6237–6240. https://doi.org/10.1002/chin.200105085
Zhu SJ, Li JF (2021) Design, synthesis and herbicidal activities of p-menth-3-en-1-amine thiourea derivatives. Nat Prod Res 51:1–8. https://doi.org/10.1080/14786419.2021.1880407
Acknowledgements
This work was supported by Science and Technology Project of Chaozhou (grant No. 2019ZC01), PhD Startup Project of Hanshan Normal University (grant No. QD20181008), and The Program of Basic Capability Improvement Project of Young and Middle-aged Teachers in GuangXi Colleges and Universities (grant No. 2018KY0697).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A patent filing on the practice of this reaction [Faming Zhuanli Shenqing (2020), CN 111635345 A 20200908] has been initiated by Shouji Zhu.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhu, Sj., Li, Jf. A novel synthesis of isothiocyanates from amines and phenyl isothiocyanate via replacement reaction. Chem. Pap. 75, 4543–4547 (2021). https://doi.org/10.1007/s11696-021-01692-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-021-01692-x