Abstract
Background
Ticks are blood-sucking ectoparasites and transmit various types of protozoal, bacterial, and viral diseases in a wild as well as domestic animals and humans globally. Only a few published reports are avaliable on the prevalence of tick-borne diseases in sheep and goats in Pakistan.
Aim and objective
The aim of this systematic review and meta-analysis was to evaluate the prevalence (2000–2020) of tick-borne disease (theileriosis, babesiosis, Crimean-Congo hemorrhagic fever infection, and anaplasmosis) in sheep and goats in Pakistan.
Methods
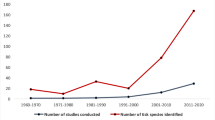
A systematic review of articles published in English language (since 2000–2020) was conducted using PubMed and Google Scholar. Diagnostic methods used in the original reference articles were PCR, PCR-RLB, microscopy, and ELISA.
Results
The overall prevalence of theileriosis, babesiosis, anaplasmosis, and Crimean-Congo hemorrhagic fever (CCHF) infections was 15.40%, 21.18%, 26.78%, and 11.62%, respectively. The prevalence of anaplasmosis was 22.06% (90/408) in sheep, 21.11% (76/360) in goats, and 40% (120/300) in both sheep and goats with substantial differences (P < 0.001). The prevalence of babesiosis among sheep was 29.88% (104/348) with highly significant differences (P < 0.001), in goats was 29.88% (25/60) with slightly significant differences (P < 0.031%), and in both sheep and goats were 7% (21/300) with highly significant differences (P < 0.001) according to subgroup analysis. The percentage of prevalence of theileriosis was 17.70% (207/1169) in sheep with highly substantial differences (P < 0.001), 4.51% (31/687) in goats with significant differences (P < 0.133), and 25% (125/500) in both sheep and goats with a significant difference among them (P < 0.001). The prevalence of CCHF among sheep was 18.63% (149/800) and 4.63% (37/800) in goats, respectively. The widely used detection method was microscopy (45.56%) followed by ELISA (38.38%), PCR (12.56%), and PCR-RLB (3.48%) test, respectively. This is a comprehensive report on the status of tick-borne disease in sheep and goats in Pakistan.
Conclusion
Based on our results, among tick-borne diseases anaplasmosis had the highest prevalence rate in sheep and goats. Due to its high prevalence, control measures should be taken to diagnose and prevent it.



Similar content being viewed by others
Availability of data and materials
All the relevant data are available in the manuscript.
Notes
Where x is the number of positive case and n is the total number of examined cases.
References
Kakar MN, Kakarsulemankhel JK (2008) Prevalence of endo (trematodes) and ectoparasites in cows and buffaloes of Quetta, Pakistan. Pak Vet J 28:34–36
Dehaghi MM, Fathi S, Asl EN, Nezhad HA (2011) Prevalence of ixodid ticks on cattle and sheep southeast of Iran. Trop Anim Health Prod 43:459–61. https://doi.org/10.1007/s11250-010-9715-y
Vahedi-Noori N, Rahbari S, Bokaei S (2012) The Seasonal Activity of Ixodes ricinus Tick in Amol, Mazandaran Province, Northern Iran. J Arthropod Borne Dis 6:129–135
Ahmed J, Alp H, Aksin M, Seitzer U (2007) Current status of ticks in Asia. Parasitol Res 101:S159–S162. https://doi.org/10.1007/s00436-007-0696-3
Ramzan M, Unsar NU, Syed HMB, Ghulam M, Alamgir AK (2018) Knowledge, attitude and practices of herdsmen about ticks and tick–borne diseases in district Multan. Pak Entomol 40:13–18
Government of Pakistan (GOP) 2019–20. Pakistan economic survey. Ministry of Finance. www.finance.gov.pk
Atif FA, Khan MS, Iqbal HJ, Roheen T (2012) Prevalence of tick-borne diseases in Punjab (Pakistan) and hematological profile of Anaplasma marginale infection in indigenous and crossbred cattle. Pak J Sci 64:11–15
Torioni de Echaide S, Knowles DP, McGuire TC, Palmer GH, Suarez CE, McElwain TF (1998) Detection of cattle naturally infected with Anaplasma marginale in a region of endemicity by nested PCR and a competitive enzyme-linked immunosorbent assay using recombinant major surface protein 5. J Clin Microbiol 36:777–782. https://doi.org/10.1128/JCM.36.3.777-782.1998
Goff WL, Johnson WC, Kuttler KL (1985) Development of an indirect fluorescent antibody test, using microfluorometry as a diagnostic test for bovine anaplasmosis. Am J Vet Res 46:1080–1084
Darghouth MA, Sassi L, Gharbi M, Soudani MC, Karoui M, Krichi A (2004) Detection of natural infections with Theileria annulata on calves at first theileriosis season: comparison of the Indirect Fluorescent Antibody Test (IFAT) and blood smears. Arch Inst Pasteur Tunis 81:41–45
Bakheit MA, Schnittger L, Salih DA, Boguslawski K, Beyer D, Fadl M, Ahmed JS (2004) Application of the recombinant Theileria annulata surface protein in an indirect ELISA for the diagnosis of tropical theileriosis. Parasitol Res 92:299–302. https://doi.org/10.1007/s00436-003-1055-7
Figueroa JV, L’Hostis M, Camus E (2010) Bovine babesiosis. In: Lefevre P-C, Blancou J, Chermette R, Uilenberg G (eds) Infectious and Parasitic Diseases of Livestock: bacterial diseases, fungal diseases, parasitic diseases, vol 2. Paris, France, Lavoisier, pp 1819–1838
Figueroa JV, Chieves LP, Johnson GS, Buening GM (1993) Multiplex polymerase chain reaction based assay for the detection of Babesia bigemina, Babesia bovis and Anaplasma marginale DNA in bovine blood. Vet Parasitol 50:69–81. https://doi.org/10.1016/0304-4017(93)90008-b
Gale KR, Dimmock CM, Gartside M, Leatch G (1996) Anaplasma marginale: detection of carrier cattle by PCR-ELISA. Int J Parasitol 26:1103–1109 (PMID: 8982791)
Carelli G, Decaro N, Lorusso A, Elia G, Lorusso E, Mari V, Ceci L, Buonavoglia C (2007) Detection and quantification of Anaplasma marginale DNA in blood samples of cattle by real-time PCR. Vet Microbiol 124:107–114. https://doi.org/10.1016/j.vetmic.2007.03.022
Kim C, Iseki H, Herbas MS, Yokoyama N, Suzuki H, Xuan X, Fujisaki K, Igarashi I (2007) Development of TaqMan-based real-time PCR assays for diagnostic detection of Babesia bovis and Babesia bigemina. Am J Trop Med Hyg 7:837–841 (PMID: 17984338)
d’Oliveira C, van der Weide M, Habela MA, Jacquiet P, Jongejan F (1995) Detection of Theileria annulata in blood samples of carrier cattle by PCR. J Clin Microbiol 33:2665–2669. https://doi.org/10.1128/JCM.33.10.2665-2669
Gubbels JM, de Vos AP, van der Weide M, Viseras J, Schouls LM, de Vries E, Jongejan F (1999) Simultaneous detection of bovine Theileria and Babesia species by reverse line blot hybridization. J Clin Microbiol 37:1782–1789. https://doi.org/10.1128/JCM.37.6.1782-1789
Gomes J, Inácio J (2015) Direct detection of Theileria annulata in bovine blood samples using standard and isothermal DNA amplification approaches. Veterinary Infection Biology: Molecular Diagnostics and High-Throughput Strategies. Humana Press, New York, pp 175–182
Arksey H, O’Malley L (2005) Scoping studies: towards a methodological framework. Int J Soc Res Methodol 8:19–32
Jabbar AI, Felicia P (2015) Gameplay engagement and learning in game-based learning: A systematic review. Rev Educ Res 85:740–779
Bilgic HB, Bakırcı S, Kose O, Unlu AH, Hacılarlıoglu S, Eren H, Weir W, Karagenc T (2017) Prevalence of tick-borne haemoparasites in small ruminants in Turkey and diagnostic sensitivity of single-PCR and RLB. Parasit Vectors 10:211. https://doi.org/10.1186/s13071-017-2151-3
Soosaraei M, Haghi MM, Etemadifar F, Fakhar M, Teshnizi SH, Asfaram S, Esboei BR (2020) Status of Anaplasma spp. infection in domestic ruminants from Iran: A systematic review with meta-analysis. Parasite Epidemiol Control 11:e00173. https://doi.org/10.1016/j.parepi.2020.e00173
Qin G, Li Y, Liu J, Liu Z, Yang J, Zhang L, Liu G, Guan G, Luo J, Yin H (2016) Molecular detection and characterization of Theileria infection in cattle and yaks from Tibet Plateau Region, China. Parasitol Res 115:2647–52. https://doi.org/10.1007/s00436-016-5011-8
Haghi MM, Etemadifar F, Fakhar M, Teshnizi SH, Soosaraei M, Shokri A, Hajihasani A, Mashhadi H (2017) Status of babesiosis among domestic herbivores in Iran: a systematic review and meta-analysis. Parasitol Res 116:1101–1109. https://doi.org/10.1007/s00436-016-5368-8
Naz S, Maqbool A, Ahmed S, Ashraf K, Ahmed N, Saeed K, Latif M, Iqbal J, Ali Z, Shafi K, Nagra A (2012) Prevalence of theileriosis in small ruminants in Lahore-Pakistan. J Vet Anim Sci 2:16–20
Kasi KK, Sas MA, Sauter-Louis C, von Arnim F, Gethmann JM, Schulz A, Wernike K, Groschup MH, Conraths FJ (2020) Epidemiological investigations of Crimean-Congo haemorrhagic fever virus infection in sheep and goats in Balochistan. Pakistan Ticks Tick Borne Dis 11:101324. https://doi.org/10.1016/j.ttbdis.2019.101324
Khalili M, Sakhaee E, Bagheri Amiri F, Safat AA, Afshar D, Esmaeili S (2020) Serological evidence of leptospirosis in Iran; A systematic review and meta-analysis. Microb Pathog 138:103833. https://doi.org/10.1016/j.micpath.2019
Irshad N, Qayyum M, Hussain M, Khan MQ (2010) Prevalence of tick infestation and theileriosis in sheep and goats. Pak Vet J 30:178–180
Rehman Z, Khan MS, Avais M, Aleem M, Shabbir MZ, Khan JA (2010) Prevalence of theileriosis in sheep in Okara district. Pakistan Pakistan J Zool 42:639–643
Iqbal F, Fatima M, Shahnawaz S, Naeem M, Shaikh R, Ali M, Shaikh A, Aktas M, Ali M (2011) A study on the determination of risk factors associated with babesiosis and prevalence of Babesia sp., by PCR amplification, in small ruminants from Southern Punjab (Pakistan). Parasite 18:229–234. https://doi.org/10.1051/parasite/2011183229
Shahzad W, Noor H, Ahmad MU, Munir R, Sharif Saghar M, Hassan Mushtaq M, Ahmad N, Akbar G, Mehmood F (2013) Prevalence and molecular diagnosis of babesia ovis and theileria ovis in lohi sheep at livestock experiment station (LES), Bahadurnagar, Okara, Pakistan. Iran J Parasitol 8:570–578 (PMID: 25516738)
Fatima M, Saeed S, Shaikh RS, Ali M, Iqbal F (2015) A study on molecular detection of Theileria lestoquardi by PCR amplification in apparently healthy small ruminants from five districts of Southern Punjab. Pak J Zool 47:441–446
Saeed S, Jahangir M, Fatima M, Shaikh RS, Khattak RM, Ali M, Iqbal F (2015) PCR based detection of Theileria lestoquardi in apparently healthy sheep and goats from two districts in Khyber Pukhtoon Khwa (Pakistan). Trop Biomed 32:225–232
Shah SSA, Khan MI, Rahman HU (2017) Epidemiological and hematological investigations of tick-borne diseases in small ruminants in Peshawar and Khyber agency. Pak J Adv Parasitol 4:15–22
Anwar K (2018) Epidemiology of tick-borne infection in ruminants in Peshawar. J Adv Parasitol 5:6–10
Khan A, Nasreen, Mitchell RD III, Niaz S, Ayaz S, Khattak I, Naeem H, Zaman MA (2019) Seroprevalence of Anaplasma spp. among sheep and goats in Charsadda District, Pakistan. Small Rumin Res 176:5–10. https://doi.org/10.1016/j.smallrumres.2019.05.008
Nasreen, Khan A, Niaz S, Hassan Shah M, Khan A, Ahmed H, Khattak I, Zeb J, Naeem H, Hassan MA, Ulucesme MC, Ozubek S, Aktas M (2020) Molecular detection of small ruminant piroplasmosis and first report of Theileria luwenshuni (Apicomplexa: Theileridae) in small ruminants of Pakistan. Exp Parasitol 212:107872. https://doi.org/10.1016/j.exppara.2020
Acknowledgements
The authors wish to acknowledge all researchers and their publications were used in our review for hard work on tick-borne diseases in Pakistan.
Funding
None to declare.
Author information
Authors and Affiliations
Contributions
HA designed the study. AM and TS designed the study protocols. NF, AK and MK searched the databases for related literature. KA, NH and SSK extracted the desired information. MQ did the statistical analyses. TM wrote the introduction and discussion with MSA. SS and HA critically revised and improved the technical quality of the manuscript. All the authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mushtaq, A., Shoukat, T., Mumtaz, T. et al. Tick-borne Diseases in Sheep and Goats in Pakistan: A Systematic Review and Meta-analysis. Acta Parasit. 66, 1316–1325 (2021). https://doi.org/10.1007/s11686-021-00396-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11686-021-00396-2