Abstract
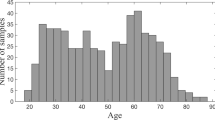
The brainstem, a small and crucial structure, is connected to the cerebrum, spinal cord, and cerebellum, playing a vital role in regulating autonomic functions, transmitting motor and sensory information, and modulating cognitive processes, emotions, and consciousness. While previous research has indicated that changes in brainstem anatomy can serve as a biomarker for aging and neurodegenerative diseases, the structural changes that occur in the brainstem during normal aging remain unclear. This study aimed to examine the age- and sex-related differences in the global and local structural measures of the brainstem in 187 healthy adults (ranging in age from 18 to 70 years) using structural magnetic resonance imaging. The findings showed a significant negative age effect on the volume of the two major components of the brainstem: the medulla oblongata and midbrain. The shape analysis revealed that atrophy primarily occurs in specific structures, such as the pyramid, cerebral peduncle, superior and inferior colliculi. Surface area and shape analysis showed a trend of flattening in the aging brainstem. There were no significant differences between the sexes or sex-by-age interactions in brainstem structural measures. These findings provide a systematic description of age associations with brainstem structures in healthy adults and may provide a reference for future research on brain aging and neurodegenerative diseases.








Similar content being viewed by others
Data availability
All data included in this study are available upon request by contact with the corresponding author.
References
Abe, O., Yamasue, H., Aoki, S., Suga, M., Yamada, H., Kasai, K., Masutani, Y., Kato, N., Kato, N., & Ohtomo, K. (2008). Aging in the CNS: Comparison of gray/white matter volume and diffusion tensor data. Neurobiology of Aging., 29, 102–116. https://doi.org/10.1016/j.neurobiolaging.2006.09.003
Attems, J., Quass, M., & Jellinger, K. A. (2007). Tau and alpha-synuclein brainstem pathology in Alzheimer disease: Relation with extrapyramidal signs. Acta Neuropathologica., 113, 53–62. https://doi.org/10.1007/s00401-006-0146-9
Avants, B., & Gee, J. C. (2004). Geodesic estimation for large deformation anatomical shape averaging and interpolation. NeuroImage, 23(Suppl 1), S139–S150. https://doi.org/10.1016/j.neuroimage.2004.07.010
Balivada, S., Ganta, C. K., Zhang, Y., Pawar, H. N., Ortiz, R. J., Becker, K. G., Khan, A. M., & Kenney, M. J. (2017). Microarray analysis of aging-associated immune system alterations in the rostral ventrolateral medulla of F344 rats. Physiological Genomics., 49, 400–415. https://doi.org/10.1152/physiolgenomics.00131.2016
Beltramo, R. (2020). A new primary visual cortex. Science, 370, 46. https://doi.org/10.1126/science.abe1482
Beltran-Castillo, S., Olivares, M. J., Contreras, R. A., Zuniga, G., Llona, I., von Bernhardi, R., & Eugenin, J. L. (2017). D-serine released by astrocytes in brainstem regulates breathing response to CO2 levels. Nature Communications, 8, 838. https://doi.org/10.1038/s41467-017-00960-3
Betts, M. J., Kirilina, E., Otaduy, M., Ivanov, D., Acosta-Cabronero, J., Callaghan, M. F., Lambert, C., Cardenas-Blanco, A., Pine, K., Passamonti, L., Loane, C., Keuken, M. C., Trujillo, P., Lusebrink, F., Mattern, H., Liu, K. Y., Priovoulos, N., Fliessbach, K., Dahl, M. J., … Hammerer, D. (2019). Locus coeruleus imaging as a biomarker for noradrenergic dysfunction in neurodegenerative diseases. Brain, 142, 2558–2571. https://doi.org/10.1093/brain/awz193
Biondetti, E., Gaurav, R., Yahia-Cherif, L., Mangone, G., Pyatigorskaya, N., Valabregue, R., Ewenczyk, C., Hutchison, M., Francois, C., Arnulf, I., Corvol, J. C., Vidailhet, M., & Lehericy, S. (2020). Spatiotemporal changes in substantia nigra neuromelanin content in Parkinson's disease. Brain, 143, 2757–2770. https://doi.org/10.1093/brain/awaa216
Boucetta, S., Cisse, Y., Mainville, L., Morales, M., & Jones, B. E. (2014). Discharge profiles across the sleep-waking cycle of identified cholinergic, GABAergic, and glutamatergic neurons in the pontomesencephalic tegmentum of the rat. Journal of Neuroscience, 34, 4708–4727. https://doi.org/10.1523/JNEUROSCI.2617-13.2014
Bouhrara, M., Cortina, L. E., Rejimon, A. C., Khattar, N., Bergeron, C., Bergeron, J., Melvin, D., Zukley, L., & Spencer, R. G. (2020). Quantitative age-dependent differences in human brainstem myelination assessed using high-resolution magnetic resonance mapping. NeuroImage, 206, 116307. https://doi.org/10.1016/j.neuroimage.2019.116307
Bouhrara, M., Cortina, L. E., Khattar, N., Rejimon, A. C., Ajamu, S., Cezayirli, D. S., & Spencer, R. G. (2021). Maturation and degeneration of the human brainstem across the adult lifespan. Aging, 13(11), 14862–14891. https://doi.org/10.18632/aging.203183
Brodal, P., & Bjaalie, J. G. (1992). Organization of the pontine nuclei. Neuroscience Research, 13, 83–118. https://doi.org/10.1016/0168-0102(92)90092-q
Butkovich, L. M., Houser, M. C., Chalermpalanupap, T., Porter-Stransky, K. A., Iannitelli, A. F., Boles, J. S., Lloyd, G. M., Coomes, A. S., Eidson, L. N., De Sousa, R. M., Oliver, D. L., Kelly, S. D., Chang, J., Bengoa-Vergniory, N., Wade-Martins, R., Giasson, B. I., Joers, V., Weinshenker, D., & Tansey, M. G. (2020). Transgenic mice expressing human alpha-synuclein in noradrenergic neurons develop locus ceruleus pathology and nonmotor features of Parkinson's disease. Journal of Neuroscience, 40, 7559–7576. https://doi.org/10.1523/JNEUROSCI.1468-19.2020
Caspary, D. M., Milbrandt, J. C., & Helfert, R. H. (1995). Central auditory aging: GABA changes in the inferior colliculus. Experimental Gerontology, 30, 349–360. https://doi.org/10.1016/0531-5565(94)00052-5
Kegeles, L. S., Brucato, G., Kang, U. J., Sulzer, D., Zecca, L., Abi-Dargham, A., Horga G. (2019). Neuromelanin-sensitive MRI as a noninvasive proxy measure of dopamine function in the human brain. Proceedings of the National Academy of Sciences, 116, 5108-5117.https://doi.org/10.1073/pnas.1807983116
Chadick, J. Z., Zanto, T. P., & Gazzaley, A. (2014). Structural and functional differences in medial prefrontal cortex underlie distractibility and suppression deficits in ageing. Nature Communications, 5, 4223. https://doi.org/10.1038/ncomms5223
Chapman, T. W., & Hill, R. A. (2020). Myelin plasticity in adulthood and aging. Neuroscience Letters, 715, 134645. https://doi.org/10.1016/j.neulet.2019.134645
Coscia, D. M., Narr, K. L., Robinson, D. G., Hamilton, L. S., Sevy, S., Burdick, K. E., Gunduz-Bruce, H., McCormack, J., Bilder, R. M., & Szeszko, P. R. (2009). Volumetric and shape analysis of the thalamus in first-episode schizophrenia. Human Brain Mapping., 30, 1236–1245. https://doi.org/10.1002/hbm.20595
D'Antoni, A. V. (2016). Gray's anatomy, the anatomical basis of clinical practice, 41st edition. Clinical Anatomy, 29, 264–265. https://doi.org/10.1002/ca.22677
De Marco, M., & Venneri, A. (2018). Volume and connectivity of the ventral tegmental area are linked to neurocognitive signatures of alzheimer's disease in humans. Journal of Alzheimers Disease, 63, 167–180. https://doi.org/10.3233/JAD-171018
Deoni, S. C., O’Muircheartaigh, J., Elison, J. T., Walker, L., Doernberg, E., Waskiewicz, N., Dirks, H., Piryatinsky, I., Dean, D. C., 3rd, & Jumbe, N. L. (2016). White matter maturation profiles through early childhood predict general cognitive ability. Brain Structure & Function, 221(2), 1189–1203. https://doi.org/10.1007/s00429-014-0947-x
Ferreira-Pinto, M. J., Ruder, L., Capelli, P., & Arber, S. (2018). Connecting circuits for supraspinal control of locomotion. Neuron, 100, 361–374. https://doi.org/10.1016/j.neuron.2018.09.015
Franke, B., Vasquez, A. A., Veltman, J. A., Brunner, H. G., Rijpkema, M., & Fernandez, G. (2010). Genetic variation in CACNA1C, a gene associated with bipolar disorder, influences brainstem rather than gray matter volume in healthy individuals. Biological Psychiatry, 68, 586–588. https://doi.org/10.1016/j.biopsych.2010.05.037
Gahm, J. K., Shi, Y., Alzheimer’s Disease Neuroimaging Initiative. (2018). Riemannian metric optimization on surfaces (RMOS) for intrinsic brain mapping in the Laplace-Beltrami embedding space. Medical Image Analysis, 46, 189–201. https://doi.org/10.1016/j.media.2018.03.004
Ge, X., Shi, Y., Li, J., Zhang, Z., Lin, X., Zhan, J., Ge, H., Xu, J., Yu, Q., Leng, Y., Teng, G., Feng, L., Meng, H., Tang, Y., Zang, F., Toga, A. W., & Liu, S. (2015). Development of the human fetal hippocampal formation during early second trimester. NeuroImage, 119, 33–43. https://doi.org/10.1016/j.neuroimage.2015.06.055
Grabert, K., Michoel, T., Karavolos, M. H., Clohisey, S., Baillie, J. K., Stevens, M. P., Freeman, T. C., Summers, K. M., & McColl, B. W. (2016). Microglial brain region-dependent diversity and selective regional sensitivities to aging. Nature Neuroscience, 19, 504–516. https://doi.org/10.1038/nn.4222
Guerrini, L., Lolli, F., Ginestroni, A., Belli, G., Della, N. R., Tessa, C., Foresti, S., Cosottini, M., Piacentini, S., Salvi, F., Plasmati, R., De Grandis, D., Siciliano, G., Filla, A., & Mascalchi, M. (2004). Brainstem neurodegeneration correlates with clinical dysfunction in SCA1 but not in SCA2. A quantitative volumetric, diffusion and proton spectroscopy MR study. Brain, 127, 1785–1795. https://doi.org/10.1093/brain/awh201
Gur, R. C., Turetsky, B. I., Matsui, M., Yan, M., Bilker, W., Hughett, P., & Gur, R. E. (1999). Sex differences in brain gray and white matter in healthy young adults: Correlations with cognitive performance. Journal of Neuroscience, 19, 4065–4072.
Guyenet, P. G., Stornetta, R. L., Souza, G., Abbott, S., & Brooks, V. L. (2020). Neuronal networks in hypertension: recent advances. Hypertension, 76, 300–311. https://doi.org/10.1161/HYPERTENSIONAHA.120.14521
Hardy, R. N., Simsek, Z. D., Curry, B., Core, S. L., Beltz, T., Xue, B., Johnson, A. K., Thunhorst, R. L., & Curtis, K. S. (2018). Aging affects isoproterenol-induced water drinking, astrocyte density, and central neuronal activation in female Brown Norway rats. Physiology & Behavior, 192, 90–97. https://doi.org/10.1016/j.physbeh.2018.03.005
Hou, Y., Dan, X., Babbar, M., Wei, Y., Hasselbalch, S. G., Croteau, D. L., & Bohr, V. A. (2019). Ageing as a risk factor for neurodegenerative disease. Nature Reviews Neurology., 15, 565–581. https://doi.org/10.1038/s41582-019-0244-7
Iglesias, J. E., Van Leemput, K., Bhatt, P., Casillas, C., Dutt, S., Schuff, N., Truran-Sacrey, D., Boxer, A., & Fischl, B. (2015). Bayesian segmentation of brainstem structures in MRI. NeuroImage, 113, 184–195. https://doi.org/10.1016/j.neuroimage.2015.02.065
Jean, A. (2001). Brain stem control of swallowing: Neuronal network and cellular mechanisms. Physiological Reviews, 81, 929–969. https://doi.org/10.1152/physrev.2001.81.2.929
Johnson, S. M., & Felder, R. B. (1993). Effects of aging on the intrinsic membrane properties of medial NTS neurons of Fischer-344 rats. Journal of Neurophysiology., 70, 1975–1987. https://doi.org/10.1152/jn.1993.70.5.1975
Kerbrat, A., Gros, C., Badji, A., Bannier, E., Galassi, F., Combes, B., Chouteau, R., Labauge, P., Ayrignac, X., Carra-Dalliere, C., Maranzano, J., Granberg, T., Ouellette, R., Stawiarz, L., Hillert, J., Talbott, J., Tachibana, Y., Hori, M., Kamiya, K., … Cohen-Adad, J. (2020). Multiple sclerosis lesions in motor tracts from brain to cervical cord: Spatial distribution and correlation with disability. Brain, 143, 2089–2105. https://doi.org/10.1093/brain/awaa162
Kroeger, D., Ferrari, L. L., Petit, G., Mahoney, C. E., Fuller, P. M., Arrigoni, E., & Scammell, T. E. (2017). Cholinergic, glutamatergic, and gabaergic neurons of the pedunculopontine tegmental nucleus have distinct effects on sleep/wake behavior in mice. Journal of Neuroscience., 37, 1352–1366. https://doi.org/10.1523/JNEUROSCI.1405-16.2016
Lambert, C., Chowdhury, R., Fitzgerald, T. H., Fleming, S. M., Lutti, A., Hutton, C., Draganski, B., Frackowiak, R., & Ashburner, J. (2013). Characterizing aging in the human brainstem using quantitative multimodal MRI analysis. Frontiers in Human Neuroscience., 7, 462. https://doi.org/10.3389/fnhum.2013.00462
Langley, J., Hussain, S., Flores, J. J., Bennett, I. J., & Hu, X. (2020). Characterization of age-related microstructural changes in locus coeruleus and substantia nigra pars compacta. Neurobiology of Aging., 87, 89–97. https://doi.org/10.1016/j.neurobiolaging.2019.11.016
Lebel, C., & Deoni, S. (2018). The development of brain white matter microstructure. NeuroImage, 182, 207–218. https://doi.org/10.1016/j.neuroimage.2017.12.097
Lee, C. Y., Mak, H. K., Chiu, P. W., Chang, H. C., Barkhof, F., & Chan, K. H. (2018). Differential brainstem atrophy patterns in multiple sclerosis and neuromyelitis optica spectrum disorders. Journal of Magnetic Resonance Imaging., 47, 1601–1609. https://doi.org/10.1002/jmri.25866
Lee, N. J., Park, I. S., Koh, I., Jung, T. W., & Rhyu, I. J. (2009). No volume difference of medulla oblongata between young and old Korean people. Brain Research., 1276, 77–82. https://doi.org/10.1016/j.brainres.2009.04.027
Liu, H., Wang, L., Geng, Z., Zhu, Q., Song, Z., Chang, R., & Lv, H. (2016). A voxel-based morphometric study of age- and sex-related changes in white matter volume in the normal aging brain. Neuropsychiatric Disease and Treatment, 12, 453–465. https://doi.org/10.2147/NDT.S90674
Luft, A. R., Skalej, M., Schulz, J. B., Welte, D., Kolb, R., Burk, K., Klockgether, T., & Voight, K. (1999). Patterns of age-related shrinkage in cerebellum and brainstem observed in vivo using three-dimensional MRI volumetry. Cerebral Cortex, 9, 712–721. https://doi.org/10.1093/cercor/9.7.712
Majrashi, N. A., Ahearn, T. S., & Waiter, G. D. (2020). Brainstem volume mediates seasonal variation in depressive symptoms: A cross sectional study in the UK Biobank cohort. Science and Reports, 10, 3592. https://doi.org/10.1038/s41598-020-60620-3
Malone, I. B., Leung, K. K., Clegg, S., Barnes, J., Whitwell, J. L., Ashburner, J., Fox, N. C., & Ridgway, G. R. (2015). Accurate automatic estimation of total intracranial volume: A nuisance variable with less nuisance. NeuroImage, 104, 366–372. https://doi.org/10.1016/j.neuroimage.2014.09.034
Mander, B. A., Rao, V., Lu, B., Saletin, J. M., Lindquist, J. R., Ancoli-Israel, S., Jagust, W., & Walker, M. P. (2013). Prefrontal atrophy, disrupted NREM slow waves and impaired hippocampal-dependent memory in aging. Nature Neuroscience., 16, 357–364. https://doi.org/10.1038/nn.3324
Namavar, Y., Barth, P. G., Kasher, P. R., van Ruissen, F., Brockmann, K., Bernert, G., Writzl, K., Ventura, K., Cheng, E. Y., Ferriero, D. M., Basel-Vanagaite, L., Eggens, V. R., Krageloh-Mann, I., De Meirleir, L., King, M., Graham, J. J., von Moers, A., Knoers, N., Sztriha, L., … Poll-The, B. T. (2011). Clinical, neuroradiological and genetic findings in pontocerebellar hypoplasia. Brain, 134, 143–156. https://doi.org/10.1093/brain/awq287
Nolan, P. C., & Waldrop, T. G. (1996). Ventrolateral medullary neurons show age-dependent depolarizations to hypoxia in vitro. Brain Research Developmental Brain Research, 91, 111–120. https://doi.org/10.1016/0165-3806(95)00166-2
Nordenskjold, R., Malmberg, F., Larsson, E. M., Simmons, A., Ahlstrom, H., Johansson, L., & Kullberg, J. (2015). Intracranial volume normalization methods: Considerations when investigating gender differences in regional brain volume. Psychiatry Research, 231, 227–235. https://doi.org/10.1016/j.pscychresns.2014.11.011
Nordenskjold, R., Malmberg, F., Larsson, E. M., Simmons, A., Brooks, S. J., Lind, L., Ahlstrom, H., Johansson, L., & Kullberg, J. (2013). Intracranial volume estimated with commonly used methods could introduce bias in studies including brain volume measurements. NeuroImage, 83, 355–360. https://doi.org/10.1016/j.neuroimage.2013.06.068
Pal, I., Paltati, C., Kaur, C., Shubhi, S., Kumar, P., Jacob, T. G., Bhardwaj, D. N., & Roy, T. S. (2019). Morphological and neurochemical changes in GABAergic neurons of the aging human inferior colliculus. Hearing Research, 377, 318–329. https://doi.org/10.1016/j.heares.2019.02.005
Pawar, H. N., Balivada, S., & Kenney, M. J. (2017). Does aging alter the molecular substrate of ionotropic neurotransmitter receptors in the rostral ventral lateral medulla? - A short communication. Experimental Gerontology., 91, 99–103. https://doi.org/10.1016/j.exger.2017.03.001
Petrusic, I., Dakovic, M., & Zidverc-Trajkovic, J. (2019). Volume alterations of brainstem subregions in migraine with aura. Neuroimage Clin., 22, 101714. https://doi.org/10.1016/j.nicl.2019.101714
Pivetta, C., Esposito, M. S., Sigrist, M., & Arber, S. (2014). Motor-circuit communication matrix from spinal cord to brainstem neurons revealed by developmental origin. Cell, 156, 537–548. https://doi.org/10.1016/j.cell.2013.12.014
Poewe, W., Seppi, K., Tanner, C. M., Halliday, G. M., Brundin, P., Volkmann, J., Schrag, A. E., & Lang, A. E. (2017). Parkinson disease. Nature Reviews Disease Primers, 3, 17013. https://doi.org/10.1038/nrdp.2017.13
Raininko, R., Autti, T., Vanhanen, S. L., Ylikoski, A., Erkinjuntti, T., & Santavuori, P. (1994). The normal brain stem from infancy to old age. A morphometric MRI Study. Neuroradiology, 36, 364–368. https://doi.org/10.1007/BF00612119
Rorabaugh, J. M., Chalermpalanupap, T., Botz-Zapp, C. A., Fu, V. M., Lembeck, N. A., Cohen, R. M., & Weinshenker, D. (2017). Chemogenetic locus coeruleus activation restores reversal learning in a rat model of Alzheimer's disease. Brain, 140, 3023–3038. https://doi.org/10.1093/brain/awx232
Ruder, L., & Arber, S. (2019). Brainstem circuits controlling action diversification. Annual Review of Neuroscience., 42, 485–504. https://doi.org/10.1146/annurev-neuro-070918-050201
Rusch, C. T., Bolsterli, B. K., Kottke, R., Steinfeld, R., & Boltshauser, E. (2020). Pontocerebellar hypoplasia: a pattern recognition approach. Cerebellum, 19, 569–582. https://doi.org/10.1007/s12311-020-01135-5
Sanchis-Segura, C., Ibanez-Gual, M. V., Adrian-Ventura, J., Aguirre, N., Gomez-Cruz, A. J., Avila, C., & Forn, C. (2019). Sex differences in gray matter volume: How many and how large are they really? Biology of Sex Differences., 10, 32. https://doi.org/10.1186/s13293-019-0245-7
Sanchis-Segura, C., Ibanez-Gual, M. V., Aguirre, N., Cruz-Gomez, A. J., & Forn, C. (2020). Effects of different intracranial volume correction methods on univariate sex differences in grey matter volume and multivariate sex prediction. Science and Reports, 10, 12953. https://doi.org/10.1038/s41598-020-69361-9
Sander, L., Pezold, S., Andermatt, S., Amann, M., Meier, D., Wendebourg, M. J., Sinnecker, T., Radue, E. W., Naegelin, Y., Granziera, C., Kappos, L., Wuerfel, J., Cattin, P., Schlaeger, R., & Initiative, A. D. N. (2019). Accurate, rapid and reliable, fully automated MRI brainstem segmentation for application in multiple sclerosis and neurodegenerative diseases. Human Brain Mapping, 40(14), 4091–4104. https://doi.org/10.1002/hbm.24687
Sheikhbahaei, S., Turovsky, E. A., Hosford, P. S., Hadjihambi, A., Theparambil, S. M., Liu, B., Marina, N., Teschemacher, A. G., Kasparov, S., Smith, J. C., & Gourine, A. V. (2018). Astrocytes modulate brainstem respiratory rhythm-generating circuits and determine exercise capacity. Nature Communications, 9, 370. https://doi.org/10.1038/s41467-017-02723-6
Shi, Y., Lai, R., Krishna, S., Sicotte, N., Dinov, I., Toga, A. W. (2008). Anisotropic laplace-beltrami eigenmaps: bridging reeb graphs and skeletons. Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, 2008, 1-7.https://doi.org/10.1109/CVPRW.2008.4563018
Shi, Y., Morra, J. H., Thompson, P. M., & Toga, A. W. (2009). Inverse-consistent surface mapping with Laplace-Beltrami eigen-features. Information Processing in Medical Imaging, 21, 467–478. https://doi.org/10.1007/978-3-642-02498-6_39
Shi, Y., Lai, R., Wang, D. J., Pelletier, D., Mohr, D., Sicotte, N., & Toga, A. W. (2014). Metric optimization for surface analysis in the Laplace-Beltrami embedding space. IEEE Transactions on Medical Imaging, 33, 1447–1463. https://doi.org/10.1109/TMI.2014.2313812
Subramanian, M., Hahn-Townsend, C., Clark, K. A., MohanKumar, S. M., & MohanKumar, P. S. (2015). Chronic estrogen exposure affects gene expression in the rostral ventrolateral medulla of young and aging rats: Possible role in hypertension. Brain Research, 1627, 134–142. https://doi.org/10.1016/j.brainres.2015.09.007
Szot, P. (2012). Common factors among Alzheimer's disease, Parkinson's disease, and epilepsy: Possible role of the noradrenergic nervous system. Epilepsia, 53(Suppl 1), 61–66. https://doi.org/10.1111/j.1528-1167.2012.03476.x
Tae, W. S., Sung, J. H., Baek, S. H., Lee, C. N., & Kim, B. J. (2020). Shape analysis of the subcortical nuclei in amyotrophic lateral sclerosis without cognitive impairment. Journal of Clinical Neurology, 16, 592–598. https://doi.org/10.3988/jcn.2020.16.4.592
Thompson, P. M., Hayashi, K. M., De Zubicaray, G. I., Janke, A. L., Rose, S. E., Semple, J., Hong, M. S., Herman, D. H., Gravano, D., Doddrell, D. M., & Toga, A. W. (2004). Mapping hippocampal and ventricular change in Alzheimer disease. NeuroImage, 22, 1754–1766. https://doi.org/10.1016/j.neuroimage.2004.03.040
Walhovd, K. B., Westlye, L. T., Amlien, I., Espeseth, T., Reinvang, I., Raz, N., Agartz, I., Salat, D. H., Greve, D. N., Fischl, B., Dale, A. M., & Fjell, A. M. (2011). Consistent neuroanatomical age-related volume differences across multiple samples. Neurobiology of Aging, 32, 916–932. https://doi.org/10.1016/j.neurobiolaging.2009.05.013
Wyss-Coray, T. (2016). Ageing, neurodegeneration and brain rejuvenation. Nature, 539, 180–186. https://doi.org/10.1038/nature20411
Xu, F., Ge, X., Shi, Y., Zhang, Z., Tang, Y., Lin, X., Teng, G., Zang, F., Gao, N., Liu, H., Toga, A. W., & Liu, S. (2020). Morphometric development of the human fetal cerebellum during the early second trimester. NeuroImage, 207, 116372. https://doi.org/10.1016/j.neuroimage.2019.116372
Yang, Y., Lee, J., Kim, G. (2020). Integration of locomotion and auditory signals in the mouse inferior colliculus. ELife, 9. https://doi.org/10.7554/eLife.52228
Yushkevich, P. A., Piven, J., Hazlett, H. C., et al. (2006). User-guided 3D active contour segmentation of anatomical structures: Significantly improved efficiency and reliability. NeuroImage, 31(3), 1116–1128. https://doi.org/10.1016/j.neuroimage.2006.01.015
Ziegler, D. A., Wonderlick, J. S., Ashourian, P., Hansen, L. A., Young, J. C., Murphy, A. J., Koppuzha, C. K., Growdon, J. H., & Corkin, S. (2013). Substantia nigra volume loss before basal forebrain degeneration in early Parkinson disease. JAMA Neurology, 70(2), 241–247. https://doi.org/10.1001/jamaneurol.2013.597
Funding
This work was supported by the National Natural Science Foundation of China (No. 31872802, 81301280), China Scholarship Council (No. 201506225068), Major Scientific and Technological Innovation Project of Shandong Province (No.2017CXGC1501), Postdoctoral Science Foundation of China (2015 M582098), Key Research Development Program of Shandong Province (2017GSF218077), Fundamental Research Funds for the Central Universities (2021JCG012).
Author information
Authors and Affiliations
Contributions
Minqi Hu: Conceptualization, Methodology, Segmentation, Software, Data curation, Writing—original draft, Writing—review & editing. Feifei Xu: Methodology, Software, Data curation, Visualization, Writing—review & editing. Shizhou Liu: Data curation, Segmentation, Writing—original draft. Yuan Yao: Data curation, Writing- review & editing. Qing Xia: Data acquisition, Data curation, Investigation. Caiting Zhu: Data curation, Writing—original draft. Qaiser Zubair: Writing- review & editing. Xinwen Zhang: Validation, Segmentation. Haiyan Tang: Validation, Segmentation. Shuwei Liu: Data acquisition, Conceptualization, Supervision. Yuchun Tang: Data acquisition, Conceptualization, Methodology, Formal analysis, Writing—original draft, Writing—review & editing, Supervision, Funding acquisition.
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Competing interests
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, M., Xu, F., Liu, S. et al. Aging pattern of the brainstem based on volumetric measurement and optimized surface shape analysis. Brain Imaging and Behavior 18, 396–411 (2024). https://doi.org/10.1007/s11682-023-00840-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-023-00840-z