Abstract
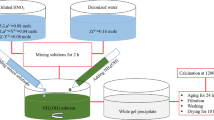
Calcium sulfate hemihydrate whiskers were synthesized successfully via one-step hydrothermal crystallization method using phosphogypsum at 130 °C for 240 min with an initial slurry mass fraction of 2.5wt%. The phase compositions, microstructures, thermal properties and molecular structures of asprepared samples were analyzed by XRD, ESEM, EDS, TG-DTA, and FT-IR. The influence of raw materials’ ball-milling time on the morphologies of whiskers was investigated. The effects of impurities on crystallization morphologies and length to diameter ratio (L/D) of calcium sulfate hemihydrate whiskers were studied. The results indicated that the calcium sulfate dihydrate crystalline could be translated directly into fibrous calcium sulfate hemihydrate whiskers. It was beneficial to form fine fiber structure when the ball-milling time of the raw material was 15 min. Aspect ratio of calcium sulfate hemihydrate whiskers decreased with increasing content of impurities. Moreover, the relative growth mechanism of whisker crystals via one-step hydrothermal crystallization method was discussed in detail.
Similar content being viewed by others
References
Canut MMC, Jacomino VMF, Bratveit K, et al. Microstructural Analyses of Phosphogypsum Generated by Brazilian Fertilizer Industries[J]. Mater. Charact., 2008, 59: 365–373
Al–Hwaiti MS, Ranville JF, Ross PE. Bioavailability and Mobility of Trace Metals in Phosphogypsum from Aqaba and Eshidiy[J]. Jordan. Chem. Erde., 2010, 70: 283–291
Bituha T, Vuci Z, Marovic G, et al. A New Approach to Determine the Phosphogypsum Spread from the Deposition Site into the Environment [J]. J. Hazard. Mater., 2013, 261: 584–592
Contreras M, Pérez–López R, Bolívar JP, et al. Fractionation and Fluxes of Metals and Radionuclides During the Recycling Process of Phosphogypsum Wastes Applied to Mineral CO2 Sequestration[J]. Waste Manage., 2015, 45: 412–419
Sahu SK, Ajmal PY, Bhangare RC, et al. Natural Radioactivity Assessment of a Phosphate Fertilizer Plant Area[J]. J. Radiat. Res. Appl. Sci., 2014, 7: 123–128
Bituha T, Vuci Z, Marovic Tayibi H, et al., Environmental Impact and Management of Phosphogypsum[J]. J. Env. Man., 2009, 90: 2 377–2 386
Yang JK, Liu WC, Zhang LL, et al. Preparation of Load–bearing Building Materials from Autoclaved Phosphogypsum[J]. Constr. Build. Mater., 2009, 23(2): 687–693
Kadirova ZC, Hojamberdiev M, Bo L, et al. Ion Uptake Properties of Low–cost Inorganic Sorption Materials in the CaO–Al2O3–SiO2 System Prepared from Phosphogypsum and Kaolin[J]. J. Clean. Prod., 2014, 83: 483–490
El–Afifi EM, Hilal MA, Attallah MF, et al. Characterization of Phosphogypsum Wastes Associated with Phosphoric Acid and Fertilizers Production[J]. J. Environ. Radioactiv., 2009, 100: 407–412
He H, Dong FQ, He P, et al. Effect of Glycerol on the Preparation of Phosphogypsum–based CaSO4·0.5H2O Whiskers[J]. J. Mater. Sci., 2014, 49: 1 957–1 963
Zhu ZC, Xu L, Chen G, et al. Optimization on Tribological Properties of Aramid Fibre and CaSO4 Whisker Reinfored Non–metallic Friction Material with Analytic Hierarchy Process and Preference Ranking Organization Method for Enrichment Evaluations[J]. Mater. Des., 2010, 31: 551–555
Mwaba MG, Gu JJ, Golriz MR. Effect of Magnetic Field on Calcium Sulfate Crystal Morphology[J]. J. Cryst. Growth, 2007, 303: 381–386
Xu A, Li H, Luo K, et al. Formation of Calcium Sulfate Whiskers from CaCO3–bearing Desulfurization Gypsum[J]. Res. Chem. Intermed., 2011, 37: 449–455
Zhu ZC, Xu L, Chen G, et al. Effect of Different Whiskers on the Physical and Tribological Properties of Non–metallic Friction Materials [J]. Mater. Des., 2011, 32: 54–61
Zhu XF, Wang YB, Wang X, et al. Integration of Preparation and Stabilization for Hemihydrate Calcium Sulfate Whiskers[J]. Adv. Mater. Res., 2011, 239–242: 3 047–3 077
Hamdona SK, Al–Hadad UA. Crystallization of Calcicalcium Sulfate Dihydrate in the Presence of Some Metal Ions[J]. J. Cryst. Growth, 2007, 299: 146–151
Liu J, Reni L, Wei Q, et al. Fabrication and Characterization of Polycaprolactone/Calcium Sulfate Whisker Composites[J]. Express Polym. Lett., 2011, 5: 742–752
Capadona JR, Shanmuganathan K, Trittschuh S, et al. Polymer Nanocomposites with Nanowhiskers Isolated from Microcrystalline Cellulose [J]. Biomacromolecules, 2009, 10: 712–716
Zhao W, Wu Y, Xu J, et al. Effect of Ethylene Glycol on Hydrothermal Formation of Calcium Sulfate Hemihydrate Whiskers with High Aspect Ratios[J]. RSC. Adv., 2015, 5: 50 544–50 548
Wang YQ, Li YC, Yuan A, et al. Preparation of Calcium Sulfate Whiskers by Carbide Slag through Hydrothermal Method[J]. Cryst. Res. Technol., 2014, 49: 800–807
Wang X, Jin B, Yang LS, et al. Effct of CuCl2 on Hydrothermal Crystallization of Calcium Sulfate Whiskers Prepared from FGD Gypsum [J]. Cryst. Res. Technol., 2015, 50: 633–640
Luo KB, Xiang CM, Li HP, et al. Influence of Temperature and Solution Composition on the Formation of Calcium Sulfates[J]. Particuology, 2010, 8: 240–244
Anto PL, Anto RJ, Varhhese HT, et al. FT–Raman and SERS Spectra of Anilinium Sulfate[J]. Ramac. Spectrosc., 2009, 40: 1 810–1 815
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by the National High-tech Research and Development Program of China (2011AA06A106)
Rights and permissions
About this article
Cite this article
Wang, S., Chen, D. & Zhang, K. Preparation, Characterization, and Formation Mechanism of Calcium Sulfate Hemihydrate Whiskers. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 33, 1407–1415 (2018). https://doi.org/10.1007/s11595-018-1983-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-018-1983-9