Abstract
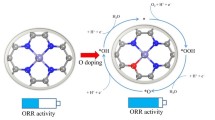
Graphene-based nanomaterials have potential as electrocatalysts for the oxygen reduction reaction (ORR) due to their unique physical and chemical properties. In this study, the mechanism of the ORR on M–N3 (M = Mn, Fe, Co, Ni, Cu) co-doped graphene with defects is investigated using dispersion-corrected density functional theory (DFT) computations. The results show that Mn-N3-Gra, Co-N3-Gra, and Ni-N3-Gra are more thermodynamically stable than Fe-N3-Gra and Cu-N3-Gra. Only Mn-N3-Gra exhibits some catalytic activity at pH < 11.327 while M-N3-Gra (M = Fe, Co, Ni, and Cu) are not suitable ORR catalysts. Under acidic conditions at pH = 0, the overpotentials of the optimal 4e− catalytic path are 0.56 V, 0.849 V, and 0.381 V for Mn-N3-Gra, Co-N3-Gra, and Ni-N3-Gra, respectively. These values are consistent with experimental results. These results indicate that Ni-N3-Gra, the optimal catalyst in this study, is comparable with Pt. The M-N3-Gra (M = Fe, Cu) catalysts cannot promote ORR under acid conditions.








Similar content being viewed by others
References
Roger AP (2000) The aluminum-air battery. Light Met Age 58:208–120
Bai L, Liu J, Gu W, Song Y, Wang F (2019) Carbon-based nanostructures vertically arrayed on layered lanthanum oxycarbonate as highly efficient catalysts for oxygen reduction reactions. ACS Appl Mater Interfaces 11:16452–16460
Ricca C, Labat F, Zavala C, Russo N, Adamo C, Merino G, Sicilia E (2017) B,N-codoped graphene as catalyst for the oxygen reduction reaction: Insights from periodic and cluster DFT calculations. J Comput Chem 39:637–647
Wu P, Du P, Zhang H, Cai C (2012) Graphyne as a promising metal-free electrocatalyst for oxygen reduction reactions in acidic fuel cells: a DFT study. J Phys Chem C 116:20472–20479
Kettel S, Wang G (2014) Reaction pathway for oxygen reduction on FeN4 embedded graphene. J Phys Chem Lett 5:452–456
Kattel S, Atanassov P, Kiefer B (2013) Catalytic activity of co-Nx/C electrocatalyst for oxygen reduction reaction: a density functional theory study. Phys Chem Chem Phys 15:148–153
Kattel S, Wang G (2013) A density functional theory study of oxygen reduction reaction on Me–N4 (Me = Fe, Co, or Ni) clusters between graphitic pores. J Mater Chem 1:10790–10797
Lu Z, Li S, Liu C, He C, Yang X, Ma D, Xu G, Yang Z (2017) Sulfur doped graphene as a promising metal-free electrocatalyst for oxygen reduction reaction: a DFT-D study. RSC Adv 7:20398–20405
Lu Z, Xu G, He C, Wang T, Yang L, Yang Z, Ma D (2015) Novel catalytic activity for oxygen reduction reaction on MnN4 embedded graphene: A dispersion-corrected density functional theory study. Carbon 84:500–508
Zhang X, Lu Z, Yang Z (2016) The mechanism of oxygen reduction reaction on CoN4 embedded graphene: a combined kinetic and atomistic thermodynamic study. Int J Hydrog Energy 41:21212–21220
Zhang J, Wang Z, Zhu Z (2014) The inherent kinetic electrochemical reduction of oxygen into H2O on FeN4-carbon: a density functional theory study. J Power Sources 255:65–69
Liu X, Dai L (2016) Carbon-based metal-free catalysts. Nat Rev Mater 1:1–12
Bai L, Duan Z, Wen X, Si R, Guan J (2019) Atomically dispersed manganese-based catalysts for efficient catalysis of oxygen reduction reaction. Appl Catal B Environ 257:117930
Bai L, Duan Z, Wen X, Guan J (2019) Bifunctional atomic iron-based catalyst for oxygen electrode reactions. J Catal 378:353–362
Wen X, Duan Z, Bai L, Guan J (2019) Atomic scandium and nitrogen-codoped graphene for oxygen reduction reaction. J Power Sources 431:265–273
Zhang L, Xia Z (2011) Mechanisms of oxygen reduction reaction on nitrogen-doped graphene for fuel cells. J Phys Chem C 115:11170–11176
Luo Z, Lim S, Tian Z, Shang J, Lai L, MacDonald B, Fu C, Shen Z, Yu T, Lin J (2011) Pyridinic N doped graphene: synthesis, electronic structure, and electrocatalytic property. J Mater Chem 21:8038–8044
Zhang P, Sun F, Xiang Z, Shen Z, Yun J, Cao D (2014) ZIF-derived in situ nitrogen-doped porous carbons as efficient metal-free electrocatalysts for oxygen reduction reaction. Energy Environ Sci 7:442–450
Lin Y, Su D (2014) Fabrication of nitrogen-modified annealed nanodiamond with improved catalytic activity. ACS Nano 8:7823–7833
Wang D, Su D (2014) Heterogeneous nanocarbon materials for oxygen reduction reaction. Energy Environ Sci 7:576–591
Maiti UN, Lee WJ, Lee JM, Oh Y, Kim JY, Kim JE, Shim J, Han TH, Kim SO (2014) 25th anniversary article: chemically modified/doped carbon nanotubes & graphene for optimized nanostructures & nano-devices. Adv Mater 26:40–67
Lima FHB, Zhang J, Shao MH, Sasaki K, Vukmirovic MB, Ticianelli EA, Adzic RR (2007) Catalytic activity−d-band center correlation for the O2 reduction reaction on platinum in alkaline solutions. J Phys Chem C 111:404–410
Li X, Li Q, Cheng J, Liu L, Yan Q, Wu Y, Zhang X, Wang Z, Qiu Q, Luo Y (2016) Conversion of dinitrogen to ammonia by FeN3-embedded graphene. J Am Chem Soc 138:8706–8709
Sun X, Li K, Yin C, Wang Y, He F, Tang H, Wu Z (2017) CoN3 embedded graphene, a potential catalyst for the oxygen reduction reaction from a theoretical perspective. Phys Chem Chem Phys 19:17670–17676
Hawkins JM, Meyer A, Solow MA (1993) Osmylation of C70: reactivity versus local curvature of the fullerene spheroid. J Am Chem Soc 115:7499–7500
Sabirov DS, Bulgakov RG (2011) Reactivity of fullerenes family towards radicals in terms of local curvature. Comput Theor Chem 963:185–190
Choho K, Langenaeker W, De Woude GV, Gerrlings P (1995) Reactivity of fullerenes. Quantum-chemical descriptors versus curvature. J Mol Struct THEOCHEM 338:293–301
Wang Y, Jiao M, Song W, Wu Z (2017) Doped fullerene as a metal-free electrocatalyst for oxygen reduction reaction: a first-principles study. Carbon. 114:393–401
Gao S, Wei X, Fan H, Li L, Geng K, Wang J (2015) Nitrogen-doped carbon shell structure derived from natural leaves as a potential catalyst for oxygen reduction reaction. Nano Energy 13:518–526
Delley B (1990) An all-electron numerical method for solving the local density functional for polyatomic molecules. J Chem Phys 92:508–517
Delley B (2000) From molecules to solids with the DMol3 approach. J Chem Phys 113:7756–7764
Lee C, Yang W, Parr RG (1988) Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789
Grimme S (2006) Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J Comput Chem 27:1787–1799
Norskov JK, Rossmeisl J, Logadottir A, Lindqvist L (2004) Origin of the overpotential for oxygen reduction at a fuel-cell cathode. J Phys Chem B 108:17886–17892
Mulliken RS (1995) Electronic population analysis on LCAO–MO molecular wave functions. I J Chem Phys 23:1841–1846
Yang N, Li L, Li J, Ding W, Wei Z (2018) Modulating oxygen reduction activity of heteroatoms doped carbon catalysts via triple effect: charge, spin density and ligand effect. Chem Sci 9:5795–5804
Orellana W (2013) Catalytic properties of transition metal–N4 moieties in graphene for the oxygen reduction reaction: evidence of spin-dependent mechanisms. J Phys Chem C 117:9812–9818
Mahesh DB, Geunsik L, Jae SL (2017) Density functional theory (DFT) calculations for oxygen reduction reaction mechanisms on metal-, nitrogen- co-doped graphene(M-N2-G (M = Ti, Cu, Mo, Nb and Ru)) electrocatalysts. Electrochim Acta 228:619–627
Kabir S, Artyushkova K, Kiefer B, Atanassov P (2015) Computational and experimental evidence for a new TM-N3/C moiety family in non-PGM electrocatalysts. Phys Chem Chem Phys 17:17785–17789
Zhang X, Yang Z, Lu Z, Wang W (2018) Bifunctional CoNx embedded graphene electrocatalysts for OER and ORR: a theoretical evaluation. Carbon. 130:112–119
Liu K, Qiao Z, Hwang S, Liu Z, Zhang H, Su D, Xu H, Wu G, Wang G (2019) Mn- and N- doped carbon as promising catalysts for oxygen reduction reaction: theoretical prediction and experimental validation. Appl Catal B 243:195–203
Jung WS, Kim Y, Noh Y, Han H, Park S, Lee J, Kim WB (2018) Comparative investigation of nitrogen species in transition metals incorporated carbon catalysts for the oxygen reduction reaction. Chem Phys Lett 708:42–47
Cheng Q, Han S, Mao K, Chen C, Yang L, Zou Z, Gu M, Hu Z, Yang H (2018) Co nanoparticle embedded in atomically-dispersed Co-N-C nanofibers for oxygen reduction with high activity and remarkable durability. Nano Energy 52:485–493
Duan Z, Wang G (2011) A first principles study of oxygen reduction reaction on a Pt(111) surface modified by a subsurface transition metal M (M = Ni, Co, or Fe). Phys Chem Chem Phys 13:20178–20187
Duan Z, Wang G (2013) Comparison of reaction energetics for oxygen reduction reactions on Pt(100), Pt(111), Pt/Ni(100), and Pt/Ni(111) surfaces: a first-principles study. J Phys Chem C 117:6284–6292
Funding
The authors acknowledge the financial support of the KEY LABORATORY OF METAL CELL OF SICHUAN PROVINCE. The calculations are performed on resources provided by the High Performance Computing Center of Tianjin University and those provided by the School of Chemical Engineering and Technology in Tianjin University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 7100 kb)
Rights and permissions
About this article
Cite this article
Gao, Q. A DFT study of the ORR on M–N3 (M = Mn, Fe, Co, Ni, or Cu) co-doped graphene with moiety-patched defects. Ionics 26, 2453–2465 (2020). https://doi.org/10.1007/s11581-019-03376-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-019-03376-9