Abstract
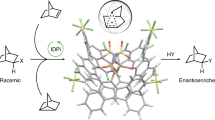
Electrostatic interaction, especially electrostatic attraction, usually plays critical roles in controlling the reactivity and selectivity in catalytic transformations; however, the like-charge repulsion, which is ubiquitous in physical systems, is rarely applied in reaction control. Herein we disclosed an unexpected like-charge repulsion induced enantio-control mode in primary aminecatalyzed fluorination reactions with 1-fluoro-2,4,6-trimethylpyridinium triflate. The ionic reaction works favorably in both highly polar (methanol) and non-polar (hexane) solvents, a seldom observed phenomenon in asymmetric catalysis. Erying plot analysis showed that an inversion temperature existed in MeOH, which was explained by the solvent-solute cluster model under different temperatures. Density functional theory (DFT) study and energy decomposition analysis (EDA) verified that the like-charge repulsion takes effect in polar solvent methanol, while in nonpolar solvents, the steric repulsion associated with ion-pair was found to be the major effect for the observed enantioselectivity.

Similar content being viewed by others
References
O’Donnell MJ. Acc Chem Res, 2004, 37: 506–517
Lygo B, Andrews BI. Acc Chem Res, 2004, 37: 518–525
Hashimoto T, Maruoka K. Chem Rev, 2007, 107: 5656–5682
Ooi T, Maruoka K. Angew Chem Int Ed, 2007, 46: 4222–4266
Jew S, Park H. Chem Commun, 2009, 7090
Hashimoto T, Maruoka K. in Asymmetric Phase Transfer Catalysis. Maruoka K, Eds. New York: Wiley-VCH, 2008. 1–8
Shirakawa S, Maruoka K. in Catalytic Asymmetric Synthesis. 3rd ed. Ojima I, Eds. New York: Wiley-VCH, 2010. 95–119
Ooi T, Maruoka K. Acc Chem Res, 2004, 37: 526–533
Dolling UH, Davis P, Grabowski EJJ. J Am Chem Soc, 1984, 106: 446–447
Hamilton GL, Kanai T, Toste FD. J Am Chem Soc, 2008, 130: 14984–14986
Lv J, Zhang Q, Zhong X, Luo S. J Am Chem Soc, 2015, 137: 15576–15583
Mayer S, List B. Angew Chem Int Ed, 2006, 45: 4193–4195
Kennedy CR, Lehnherr D, Rajapaksa NS, Ford DD, Park Y, Jacobsen EN. J Am Chem Soc, 2016, 138: 13525–13528
Ford DD, Lehnherr D, Kennedy CR, Jacobsen EN. ACS Catal, 2016, 6: 4616–4620
Ford DD, Lehnherr D, Kennedy CR, Jacobsen EN. J Am Chem Soc, 2016, 138: 7860–7863
Wasa M, Liu RY, Roche SP, Jacobsen EN. J Am Chem Soc, 2014, 136: 12872–12875
Bergonzini G, Schindler CS, Wallentin CJ, Jacobsen EN, Stephenson CRJ. Chem Sci, 2014, 5: 112–116
Birrell JA, Desrosiers JN, Jacobsen EN. J Am Chem Soc, 2011, 133: 13872–13875
Brown AR, Kuo WH, Jacobsen EN. J Am Chem Soc, 2010, 132: 9286–9288
Raheem IT, Thiara PS, Peterson EA, Jacobsen EN. J Am Chem Soc, 2007, 129: 13404–13405
Brak K, Jacobsen EN. Angew Chem Int Ed, 2013, 52: 534–561
Aoki E, Sato D, Fujiwara K, Ikeguchi M. Biochemistry, 2017, 56: 2139–2148
Leclercq L, Bauduin P, Nardello-Rataj V. Langmuir, 2017, 33: 3395–3403
Pivetta M, Rusponi S, Brune H. Phys Rev B, 2018, 98: 115417
You Y, Zhang L, Luo S. Chem Sci, 2017, 8: 621–626
Cui L, You Y’, Mi X, Luo S. J Org Chem, 2018, 83: 4250–4256
Wang Y, Zhou H, Yang K, You C, Zhang L, Luo S. Org Lett, 2019, 21: 407–411
Reichardt C, Welton T. Sovlents and Solvent Effect in Organic Chemistry. Weinheim: Wiley-VCH, 2011
Jain S, Vanka K. J Phys Chem A, 2020, 124: 8019–8028
Abadie MA, Trivelli X, Medina F, Duhal N, Kouach M, Linden B, Génin E, Vandewalle M, Capet F, Roussel P, Del Rosal I, Maron L, Agbossou-Niedercorn F, Michon C. Chem Eur J, 2017, 23: 10777–10788
Selim KB, Martel A, Laurent MY, Lhoste J, Py S, Dujardin G. J Org Chem, 2014, 79: 3414–3426
Yu H, Xie F, Ma Z, Liu Y, Zhang W. Org Biomol Chem, 2012, 10: 5137–5142
Austin JF, Kim SG, Sinz CJ, Xiao WJ, MacMillan DWC. Proc Natl Acad Sci USA, 2004, 101: 5482–5487
Cainelli G, Galletti P, Giacomini D. Chem Soc Rev, 2009, 38: 990–1001
Buschmann H, Scharf HD, Hoffmann N, Esser P. Angew Chem Int Ed, 1991, 30: 477–515
Garzan A, Jaganathan A, Salehi Marzijarani N, Yousefi R, Whitehead DC, Jackson JE, Borhan B. Chem Eur J, 2013, 19: 9015–9021
Ilg MK, Wolf LM, Mantilli L, Farès C, Thiel W, Fürstner A. Chem Eur J, 2015, 21: 12279–12284
Talsi EP, Rybalova TV, Bryliakov KP. ACS Catal, 2015, 5: 4673–4679
Cainelli G, Giacomini D, Galletti P, Marini A. Angew Chem Int Ed, 1996, 35: 2849–2852
Cainelli G, Giacomini D, Galletti P, Orioli P, Paradisi F. Eur J Org Chem, 2000, 2000: 3619–3626
Cainelli G, Giacomini D, Galletti P, Orioli P. Eur J Org Chem, 2001, 2001: 4509–4515
Cainelli G, Giacomini D, Galletti P, Quintavalla A. Eur J Org Chem, 2002, 2002: 3153–3161
Olsson MHM, Warshel A. J Am Chem Soc, 2004, 126: 15167–15179
Cainelli G, Galletti P, Giacomini D, Gualandi A, Quintavalla A. HCA, 2003, 86: 3548–3559
Kitamoto Y, Kuruma Y, Suzuki K, Hattori T. J Org Chem, 2015, 80: 521–527
Kato M, Hirao S, Nakano K, Sato M, Yamanaka M, Sohtome Y, Nagasawa K. Chem Eur J, 2015, 21: 18606–18612
Lage-Estebanez I, del Olmo L, López R, García de la Vega JM. J Comput Chem, 2017, 38: 530–540
Knorr A, Stange P, Fumino K, Weinhold F, Ludwig R. ChemPhysChem, 2016, 17: 458–462
Strate A, Niemann T, Michalik D, Ludwig R. Angew Chem Int Ed, 2017, 56: 496–500
Khudozhitkov AE, Neumann J, Niemann T, Zaitsau D, Stange P, Paschek D, Stepanov AG, Kolokolov DI, Ludwig R. Angew Chem Int Ed, 2019, 58: 17863–17871
Holthoff JM, Weiss R, Rosokha SV, Huber SM. Chem Eur J, 2021, 27: 16530–16542
Zhao Y, Truhlar DG. Acc Chem Res, 2008, 41: 157–167
Zhao Y, Truhlar DG. Theor Chem Account, 2008, 120: 215–241
Xie S, Lopez SA, Ramström O, Yan M, Houk KN. J Am Chem Soc, 2015, 137: 2958–2966
Yang YF, Yu P, Houk KN. J Am Chem Soc, 2017, 139: 18213–18221
Huang M, Zhang L, Pan T, Luo S. Science, 2022, 375: 869–874
Cai M, Xu K, Li Y, Nie Z, Zhang L, Luo S. J Am Chem Soc, 2021, 143: 1078–1087
Becke AD. J Chem Phys, 1993, 98: 5648–5652
Lee C, Yang W, Parr RG. Phys Rev B, 1988, 37: 785–789
Weigend F, Ahlrichs R. Phys Chem Chem Phys, 2005, 7: 3297–3305
Marenich AV, Cramer CJ, Truhlar DG. J Phys Chem B, 2009, 113: 6378–6396
Ribeiro RF, Marenich AV, Cramer CJ, Truhlar DG. J Phys Chem B, 2011, 115: 14556–14562
Lam Y, Houk KN. J Am Chem Soc, 2014, 136: 9556–9559
Xu C, Zhang L, Luo S. Angew Chem Int Ed, 2014, 53: 4149–4153
Lu T, Chen F. J Comput Chem, 2012, 33: 580–592
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22373056, 22031006), the Haihe Laboratory of Sustainable Chemical Transformations (YYJC202113), and the National Science & Technology Fundamental Resource Investigation Program of China (2018FY201200). L.Z. was supported by the National Program of Top-notch Young Professionals.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at https://chem.scichina.com and https://link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Electronic supplementary material
11426_2023_1746_MOESM1_ESM.pdf
Understanding How Charge-charge Interaction Affects the Stereochemistry of Enamine Fluorination by Chiral Primary Amine Catalysis
Rights and permissions
About this article
Cite this article
Han, Y., Yang, K., Zhang, L. et al. Understanding how charge-charge interaction affects the stereochemistry of enamine fluorination by chiral primary amine catalysis. Sci. China Chem. 66, 2828–2835 (2023). https://doi.org/10.1007/s11426-023-1746-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-023-1746-7