Abstract
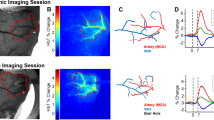
Whole-brain irradiation (WBI, also known as whole-brain radiation therapy) is a mainstay treatment modality for patients with multiple brain metastases. It is also used as a prophylactic treatment for microscopic tumors that cannot be detected by magnetic resonance imaging. WBI induces a progressive cognitive decline in ~ 50% of the patients surviving over 6 months, significantly compromising the quality of life. There is increasing preclinical evidence that radiation-induced injury to the cerebral microvasculature and accelerated neurovascular senescence plays a central role in this side effect of WBI. To better understand this side effect, male C57BL/6 mice were first subjected to a clinically relevant protocol of fractionated WBI (5 Gy, two doses per week, for 4 weeks). Nine months post the WBI treatment, we applied two-photon microscopy and Doppler optical coherence tomography to measure capillary red-blood-cell (RBC) flux, capillary morphology, and microvascular oxygen partial pressure (PO2) in the cerebral somatosensory cortex in the awake, head-restrained, WPI-treated mice and their age-matched controls, through a cover-glass-sealed chronic cranial window. Thanks to the extended penetration depth with the fluorophore — Alexa680, measurements of capillary blood flow properties (e.g., RBC flux, speed, and linear density) in the cerebral subcortical white matter were enabled. We found that the WBI-treated mice exhibited a significantly decreased capillary RBC flux in the white matter. WBI also caused a significant reduction in capillary diameter, as well as a large (although insignificant) reduction in segment density at the deeper cortical layers (e.g., 600–700 μm), while the other morphological properties (e.g., segment length and tortuosity) were not obviously affected. In addition, we found that PO2 measured in the arterioles and venules, as well as the calculated oxygen saturation and oxygen extraction fraction, were not obviously affected by WBI. Lastly, WBI was associated with a significant increase in the erythrocyte-associated transients of PO2, while the changes of other cerebral capillary PO2 properties (e.g., capillary mean-PO2, RBC-PO2, and InterRBC-PO2) were not significant. Collectively, our findings support the notion that WBI results in persistent cerebral white matter microvascular impairment, which likely contributes to the WBI-induced brain injury and cognitive decline. Further studies are warranted to assess the WBI-induced changes in brain tissue oxygenation and malfunction of the white matter microvasculature as well.








Similar content being viewed by others
References
Graus F, Walker RW, Allen JC. Brain metastases in children. J Pediatr. 1983;103:558–61.
Nayak L, Lee EQ, Wen PY. Epidemiology of brain metastases. Curr Oncol Rep. 2012;14:48–54.
Gaspar LE, et al. The role of whole brain radiation therapy in the management of newly diagnosed brain metastases: a systematic review and evidence-based clinical practice guideline. J Neurooncol. 2010;96:17–32.
Patil CG, et al. Whole brain radiation therapy (WBRT) alone versus WBRT and radiosurgery for the treatment of brain metastases. Cochrane Database Syst Rev. 2017;9:CD006121.
Tsao MN, et al. Whole brain radiotherapy for the treatment of newly diagnosed multiple brain metastases. Cochrane Database Syst Rev. 2018. https://doi.org/10.1002/14651858.CD003869.pub4.
Park K, et al. Radiotherapy for brain metastasis and long-term survival. Sci Rep. 2021;11:8046.
Lee YW, Cho HJ, Lee WH, Sonntag WE. Whole brain radiation-induced cognitive impairment: pathophysiological mechanisms and therapeutic targets. Biomol Ther. 2012;20:357–70.
Dieckmann K, Herrmann H. Are there still indications for whole brain irradiation in 2021? Memo - Mag Eur Med Oncol. 2021;14:204–7.
van Grinsven EE, Nagtegaal SHJ, Verhoeff JJC, van Zandvoort MJE. The impact of stereotactic or whole brain radiotherapy on neurocognitive functioning in adult patients with brain metastases: a systematic review and meta-analysis. Oncol Res Treat. 2021;44:622–36.
Khuntia D, Brown P, Li J, Mehta MP. Whole-brain radiotherapy in the management of brain metastasis. J Clin Oncol. 2006;24:1295–304.
Welzel G, et al. Acute neurocognitive impairment during cranial radiation therapy in patients with intracranial tumors. Strahlenther Onkol. 2008;184:647.
Welzel G, et al. Memory function before and after whole brain radiotherapy in patients with and without brain metastases. Int J Radiat Oncol Biol Phys. 2008;72:1311–8.
Brown PD, et al. Effect of radiosurgery alone vs radiosurgery with whole brain radiation therapy on cognitive function in patients with 1 to 3 brain metastases: a randomized clinical trial. JAMA. 2016;316:401–9.
Meyers CA, et al. Neurocognitive function and progression in patients with brain metastases treated with whole-brain radiation and motexafin gadolinium: results of a randomized phase III trial. J Clin Oncol Off J Am Soc Clin Oncol. 2004;22:157–65.
Greene-Schloesser D, et al. Radiation-induced brain injury: a review. Front Oncol. 2012;2:73.
Yabluchanskiy A, et al. Pharmacological or genetic depletion of senescent astrocytes prevents whole brain irradiation-induced impairment of neurovascular coupling responses protecting cognitive function in mice. GeroScience. 2020;42:409–28.
Lamproglou I, et al. Total body 4.5 Gy gamma irradiation-induced early delayed learning and memory dysfunction in the rat. Cell Mol Biol Noisy Gd Fr. 2001;47:453–7.
Shi L, et al. Spatial learning and memory deficits after whole-brain irradiation are associated with changes in NMDA receptor subunits in the hippocampus. Radiat Res. 2006;166:892–9.
Soussain C, et al. CNS complications of radiotherapy and chemotherapy. The Lancet. 2009;374:1639–51.
Warrington JP, Csiszar A, Mitschelen M, Lee YW, Sonntag WE. Whole brain radiation-induced impairments in learning and memory are time-sensitive and reversible by systemic hypoxia. PLoS ONE. 2012;7:e30444.
Ungvari Z, et al. Cerebromicrovascular dysfunction predicts cognitive decline and gait abnormalities in a mouse model of whole brain irradiation-induced accelerated brain senescence. GeroScience. 2017;39:33–42.
Robbins ME, Bourland JD, Cline JM, Wheeler KT, Deadwyler SA. A model for assessing cognitive impairment after fractionated whole-brain irradiation in nonhuman primates. Radiat Res. 2011;175:519–25.
Hanbury DB, et al. Pathology of fractionated whole-brain irradiation in rhesus monkeys ( Macaca mulatta ). Radiat Res. 2015;183:367–74.
Brown WR, Thore CR, Moody DM, Robbins ME, Wheeler KT. Vascular damage after fractionated whole-brain irradiation in rats. Radiat Res. 2005;164:662–8.
Shi L, et al. Maintenance of white matter integrity in a rat model of radiation-induced cognitive impairment. J Neurol Sci. 2009;285:178–84.
Connor M, et al. Regional susceptibility to dose-dependent white matter damage after brain radiotherapy. Radiother Oncol. 2017;123:209–17.
Iadecola C. The neurovascular unit coming of age: a journey through neurovascular coupling in health and disease. Neuron. 2017;96:17–42.
Ashpole NM, et al. Systemic influences contribute to prolonged microvascular rarefaction after brain irradiation: a role for endothelial progenitor cells. Am J Physio - Heart Circ Physiol. 2014;307:H858–68.
Lee WH, Cho HJ, Sonntag WE, Lee YW. Radiation attenuates physiological angiogenesis by differential expression of VEGF, Ang-1, tie-2 and Ang-2 in rat brain. Radiat Res. 2011;176:753–60.
Warrington JP, et al. Whole brain radiation-induced vascular cognitive impairment: mechanisms and implications. J Vasc Res. 2013;50:445–57.
Warrington JP, et al. Cerebral microvascular rarefaction induced by whole brain radiation is reversible by systemic hypoxia in mice. Am. J. Physiol.-Heart Circ. Physiol. 2011;300:H736–44.
Correa DD, et al. Longitudinal cognitive assessment in patients with primary CNS lymphoma treated with induction chemotherapy followed by reduced-dose whole-brain radiotherapy or autologous stem cell transplantation. J Neurooncol. 2019;144:553–62.
Stokes TB, et al. White matter changes in breast cancer brain metastases patients who undergo radiosurgery alone compared to whole brain radiation therapy plus radiosurgery. J Neurooncol. 2015;121:583–90.
Cheng H, Chen H, Lv Y, Chen Z, Li CR. Prospective memory impairment following whole brain radiotherapy in patients with metastatic brain cancer. Cancer Med. 2018;7:5315–21.
Bovi JA, et al. Pretreatment volume of MRI-determined white matter injury predicts neurocognitive decline after hippocampal avoidant whole-brain radiation therapy for brain metastases: secondary analysis of NRG Oncology Radiation Therapy Oncology Group 0933. Adv Radiat Oncol. 2019;4:579–86.
Mayinger M, et al. Leukoencephalopathy after prophylactic whole-brain irradiation with or without hippocampal sparing: a longitudinal magnetic resonance imaging analysis. Eur J Cancer. 2020;124:194–203.
Yoo DH, et al. MR imaging evaluation of intracerebral hemorrhages and T2 hyperintense white matter lesions appearing after radiation therapy in adult patients with primary brain tumors. PLoS ONE. 2015;10:e0136795.
Andrews RN, et al. White matter is the predilection site of late-delayed radiation-induced brain injury in non-human primates. Radiat Res. 2019;191:217–31.
Panagiotakos G, et al. Long-term impact of radiation on the stem cell and oligodendrocyte precursors in the brain. PLoS ONE. 2007;2:e588.
Li B, Ohtomo R, Thunemann M, Adams SR, Yang J, Fu B, Yaseen MA, et al. Two-photon microscopic imaging of capillary red blood cell flux in mouse brain reveals vulnerability of cerebral white matter to hypoperfusion. J Cereb Blood Flow Metab. 2020;40(12):501–12. https://doi.org/10.1177/0271678X19831016.
Ahn SJ, et al. Label-free assessment of hemodynamics in individual cortical brain vessels using third harmonic generation microscopy. Biomed Opt Express. 2020;11:2665–78.
Esipova TV, et al. Oxyphor 2P: A high-performance probe for deep-tissue longitudinal oxygen imaging. Cell Metab. 2019;29:736-744.e7.
Li B, et al. More homogeneous capillary flow and oxygenation in deeper cortical layers correlate with increased oxygen extraction. ELife. 2019;8:e42299.
Demaria M, et al. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev Cell. 2014;31:722–33.
Goldey GJ, et al. Removable cranial windows for long-term imaging in awake mice. Nat Protoc. 2014;9:2515–38.
Komiyama T, et al. Learning-related fine-scale specificity imaged in motor cortex circuits of behaving mice. Nature. 2010;464:1182–6.
Sakadžić S, et al. Two-photon high-resolution measurement of partial pressure of oxygen in cerebral vasculature and tissue. Nat Methods. 2010;7:755–9.
Yaseen MA, et al. Multimodal optical imaging system for in vivo investigation of cerebral oxygen delivery and energy metabolism. Biomed Opt Express. 2015;6:4994–5007.
Unekawa M, et al. RBC velocities in single capillaries of mouse and rat brains are the same, despite 10-fold difference in body size. Brain Res. 2010;1320:69–73.
Li B, Lee J, Boas DA, Lesage F. Contribution of low- and high-flux capillaries to slow hemodynamic fluctuations in the cerebral cortex of mice. J Cereb Blood Flow Metab. 2016;36(8):1351–6. https://doi.org/10.1177/0271678X16649195.
Lee J, Wu W, Lesage F, Boas DA. Multiple-capillary measurement of RBC speed, flux, and density with optical coherence tomography. J. Cereb. Blood Flow Metab. Off J Int Soc Cereb Blood Flow Metab. 2013;33:1707–10.
Srinivasan VJ, et al. Quantitative cerebral blood flow with optical coherence tomography. Opt Express. 2010;18:2477.
Finikova OS, et al. Oxygen microscopy by two-photon-excited phosphorescence. ChemPhysChem. 2008;9:1673–9.
Uchida K, Reilly MP, Asakura T. Molecular stability and function of mouse hemoglobins. Zoolog Sci. 1998;15:703–6.
Lecoq J, et al. Simultaneous two-photon imaging of oxygen and blood flow in deep cerebral vessels. Nat Med. 2011;17:893–8.
Parpaleix A, Houssen YG, Charpak S. Imaging local neuronal activity by monitoring PO2 transients in capillaries. Nat Med. 2013;19:241–6.
Tahir W, Kura S, Zhu J, Cheng X, Damseh R, Tadesse F, et al. Anatomical modeling of brain vasculature in two-photon microscopy by generalizable deep learning. BME Frontiers. 2020. https://doi.org/10.34133/2020/8620932.
Damseh R, Delafontaine-Martel P, Pouliot P, Cheriet F, Lesage F. Laplacian flow dynamics on geometric graphs for anatomical modeling of cerebrovascular networks. IEEE Trans Med Imaging. 2021;40:381–94.
Gui C, et al. A prospective evaluation of whole brain volume loss and neurocognitive decline following hippocampal-sparing prophylactic cranial irradiation for limited-stage small-cell lung cancer. J Neurooncol. 2019;144:351–8.
Takeshita Y, et al. Early volume reduction of the hippocampus after whole-brain radiation therapy: an automated brain structure segmentation study. Jpn J Radiol. 2020;38:118–25.
Shibamoto Y, et al. Incidence of brain atrophy and decline in Mini-Mental State Examination score after whole-brain radiotherapy in patients with brain metastases: a prospective Study. Int J Radiat Oncol. 2008;72:1168–73.
Lyons DG, Parpaleix A, Roche M, Charpak S. Mapping oxygen concentration in the awake mouse brain. ELife. 2016;5:e12024.
Moeini M, et al. Compromised microvascular oxygen delivery increases brain tissue vulnerability with age. Sci Rep. 2018;8:8219.
Blinder P, et al. The cortical angiome: an interconnected vascular network with noncolumnar patterns of blood flow. Nat Neurosci. 2013;16:889–97.
Ballabh P, Braun A, Nedergaard M. Anatomic analysis of blood vessels in germinal matrix, cerebral cortex, and white matter in developing infants. Pediatr Res. 2004;56:117–24.
Cavaglia M, et al. Regional variation in brain capillary density and vascular response to ischemia. Brain Res. 2001;910:81–93.
Müller K, Courtois G, Ursini MV, Schwaninger M. New insight into the pathogenesis of cerebral small-vessel diseases. Stroke. 2017;48:520–7.
Chojdak-Łukasiewicz J, Dziadkowiak E, Zimny A, Paradowski B. Cerebral small vessel disease: a review. Adv. Clin. Exp Med Off Organ Wroclaw Med Univ. 2021;30:349–56.
Pantoni L, Poggesi A, Inzitari D. The relation between white-matter lesions and cognition. Curr Opin Neurol. 2007;20:390–7.
Horsfield MA, Jones DK. Applications of diffusion-weighted and diffusion tensor MRI to white matter diseases – a review. NMR Biomed. 2002;15:570–7.
Nakao S, Yamamoto T, Kimura S, Mino T, Iwamoto T. Brain white matter lesions and postoperative cognitive dysfunction: a review. J Anesth. 2019;33:336–40.
Golub AS, Pittman RN. Erythrocyte-associated transients in PO2 revealed in capillaries of rat mesentery. Am J Physiol Heart Circ Physiol. 2005;288:H2735-2743.
Cabrales P, Intaglietta M. Time-dependant oxygen partial pressure in capillaries and tissue in the hamster window chamber model. Antioxid Redox Signal. 2007;9:845–53.
Barker MC, Golub AS, Pittman RN. Erythrocyte-associated transients in capillary PO2: an isovolemic hemodilution study in the rat spinotrapezius muscle. Am J Physiol Heart Circ Physiol. 2007;292:H2540-2549.
Wälchli T, et al. Hierarchical imaging and computational analysis of three-dimensional vascular network architecture in the entire postnatal and adult mouse brain. Nat Protoc. 2021;16:4564–610.
Akiyama K, Tanaka R, Sato M, Takeda N. Cognitive dysfunction and histological findings in adult rats one year after whole brain irradiation. Neurol Med Chir (Tokyo). 2001;41:590–8.
Nakagaki H, Brunhart G, Kemper TL, Caveness WF. Monkey brain damage from radiation in the therapeutic range. J Neurosurg. 1976;44:3–11.
Andrews RN, et al. Cerebrovascular remodeling and neuroinflammation is a late effect of radiation-induced brain injury in non-human primates. Radiat Res. 2017;187:599–611.
Constanzo J, et al. Diffusion MRI monitoring of specific structures in the irradiated rat brain. Magn Reson Med. 2018;80:1614–25.
Peiffer AM, Shi L, Olson J, Brunso-Bechtold JK. Differential effects of radiation and age on diffusion tensor imaging in rats. Brain Res. 2010;1351:23–31.
Correa DD, et al. Cognitive functions in survivors of primary central nervous system lymphoma. Neurology. 2004;62:548–55.
DeAngelis LM, Delattre JY, Posner JB. Radiation-induced dementia in patients cured of brain metastases. Neurology. 1989;39:789–96.
Duan F, et al. Whole-brain changes in white matter microstructure after radiotherapy for nasopharyngeal carcinoma: a diffusion tensor imaging study. Eur. Arch. Oto-Rhino-Laryngol. Off. J. Eur. Fed. Oto-Rhino-Laryngol. Soc. EUFOS Affil. Ger Soc Oto-Rhino-Laryngol - Head Neck Surg. 2016;273:4453–9.
Frytak S, et al. Magnetic resonance imaging for neurotoxicity in long-term survivors of carcinoma. Mayo Clin Proc. 1985;60:803–12.
King TZ, Wang L, Mao H. Disruption of white matter integrity in adult survivors of childhood brain tumors: correlates with long-term intellectual outcomes. PLoS ONE. 2015;10:e0131744.
Conill C, et al. Incidence of radiation-induced leukoencephalopathy after whole brain radiotherapy in patients with brain metastases. Clin Transl Oncol. 2007;9:590–5.
Bompaire F, et al. New insights in radiation-induced leukoencephalopathy: a prospective cross-sectional study. Support. Care Cancer Off. J Multinatl Assoc Support Care Cancer. 2018;26:4217–26.
Rubinstein LJ, Herman MM, Long TF, Wilbur JR. Disseminated necrotizing leukoencephalopathy: a complication of treated central nervous system leukemia and lymphoma. Cancer. 1975;35:291–305.
Lee JS, et al. Neurotoxicity in long-term survivors of small cell lung cancer. Int J Radiat Oncol Biol Phys. 1986;12:313–21.
Lee YY, Nauert C, Glass JP. Treatment-related white matter changes in cancer patients. Cancer. 1986;57:1473–82.
Leng X, et al. Application of a machine learning method to whole brain white matter injury after radiotherapy for nasopharyngeal carcinoma. Cancer Imaging Off. Publ Int Cancer Imaging Soc. 2019;19:19.
Monaco EA, et al. Leukoencephalopathy after whole-brain radiation therapy plus radiosurgery versus radiosurgery alone for metastatic lung cancer. Cancer. 2013;119:226–32.
Schuitema I, et al. Accelerated aging, decreased white matter integrity, and associated neuropsychological dysfunction 25 years after pediatric lymphoid malignancies. J Clin Oncol Off J Am Soc Clin Oncol. 2013;31:3378–88.
Szerlip N, et al. Factors impacting volumetric white matter changes following whole brain radiation therapy. J Neurooncol. 2011;103:111–9.
Vigliani MC, et al. Dementia following treatment of brain tumors with radiotherapy administered alone or in combination with nitrosourea-based chemotherapy: a clinical and pathological study. J Neurooncol. 1999;41:137–49.
Wang S, et al. Longitudinal diffusion tensor magnetic resonance imaging study of radiation-induced white matter damage in a rat model. Cancer Res. 2009;69:1190–8.
Moretti R, Caruso P. An iatrogenic model of brain small-vessel disease: post-radiation encephalopathy. Int J Mol Sci. 2020;21:6506.
Hattori H, Takeda M, Kudo T, Nishimura T, Hashimoto S. Cumulative white matter changes in the gerbil brain under chronic cerebral hypoperfusion. Acta Neuropathol (Berl). 1992;84:437–42.
Dalby RB, Eskildsen SF, Videbech P, Frandsen J, Mouridsen K, Sørensen L, et al. Oxygenation differs among white matter hyperintensities, intersected fiber tracts and unaffected white matter. Brain Commun. 1(1):fcz033. https://doi.org/10.1093/braincomms/fcz033.
Brown WR, Moody DM, Thore CR, Challa VR, Anstrom JA. Vascular dementia in leukoaraiosis may be a consequence of capillary loss not only in the lesions, but in normal-appearing white matter and cortex as well. J Neurol Sci. 2007;257:62–6.
Joutel A, et al. Cerebrovascular dysfunction and microcirculation rarefaction precede white matter lesions in a mouse genetic model of cerebral ischemic small vessel disease. J Clin Invest. 2010;120:433–45.
Miura M, et al. High prevalence of small vessel disease long after cranial irradiation. J Clin Neurosci Off J Neurosurg Soc Australas. 2017;46:129–35.
Lo EH, Frankel KA, Steinberg GK, DeLaPaz RL, Fabrikant JI. High-dose single-fraction brain irradiation: MRI, cerebral blood flow, electrophysiological, and histological studies. Int J Radiat Oncol Biol Phys. 1992;22:47–55.
Remler MP, Marcussen WH, Tiller-Borsich J. The late effects of radiation on the blood brain barrier. Int J Radiat Oncol Biol Phys. 1986;12:1965–9.
Nakata H, et al. Early blood-brain barrier disruption after high-dose single-fraction irradiation in rats. Acta Neurochir (Wien). 1995;136:82–6 (discussion 86-87).
Yoshida Y, et al. X-ray irradiation induces disruption of the blood-brain barrier with localized changes in claudin-5 and activation of microglia in the mouse brain. Neurochem Int. 2018;119:199–206.
Yuan H, Gaber MW, Boyd K, Wilson CM, Kiani MF, Merchant TE. Effects of fractionated radiation on the brain vasculature in a murine model: blood-brain barrier permeability, astrocyte proliferation, and ultrastructural changes. Int J Radiat Oncol Biol. Phys. 2006;66(3):860–6. https://doi.org/10.1016/j.ijrobp.2006.06.043.
Balasubramanian P, et al. Obesity-induced cognitive impairment in older adults: a microvascular perspective. Am J Physiol Heart Circ Physiol. 2021;320:H740–61.
Ungvari Z, et al. Hypertension-induced cognitive impairment: from pathophysiology to public health. Nat Rev Nephrol. 2021;17:639–54.
Toth P, et al. Age-related autoregulatory dysfunction and cerebromicrovascular injury in mice with angiotensin II-induced hypertension. J. Cereb. Blood Flow Metab. Off J Int Soc Cereb Blood Flow Metab. 2013;33:1732–42.
Tucsek Z, et al. Aging exacerbates obesity-induced cerebromicrovascular rarefaction, neurovascular uncoupling, and cognitive decline in mice. J Gerontol A Biol Sci Med Sci. 2014;69:1339–52.
Fu Z, et al. Age-dependent responses of brain myelin integrity and behavioral performance to radiation in mice. Radiat Res. 2017;188:505–16.
Li J, Xiao L, He D, Luo Y, Sun H. Mechanism of white matter injury and promising therapeutic strategies of MSCs after intracerebral hemorrhage. Front Aging Neurosci. 2021;13:632054.
Hung P-L, Huang C-C, Huang H-M, Tu D-G, Chang Y-C. Thyroxin treatment protects against white matter injury in the immature brain via brain-derived neurotrophic factor. Stroke. 2013;44:2275–83.
Fuks Z, Kolesnick R. Engaging the vascular component of the tumor response. Cancer Cell. 2005;8:89–91.
Furuse M, Nonoguchi N, Kawabata S, Miyatake S-I, Kuroiwa T. Delayed brain radiation necrosis: pathological review and new molecular targets for treatment. Med Mol Morphol. 2015;48:183–90.
Abayomi OK. Pathogenesis of irradiation-induced cognitive dysfunction. Acta Oncol. 1996;35:659–63.
Ungvari Z, et al. Ionizing radiation promotes the acquisition of a senescence-associated secretory phenotype and impairs angiogenic capacity in cerebromicrovascular endothelial cells: role of increased DNA damage and decreased DNA repair capacity in microvascular radiosensitivity. J Gerontol Ser A. 2013;68:1443–57.
Imaizumi N, Monnier Y, Hegi M, Mirimanoff R-O, Rüegg C. Radiotherapy suppresses angiogenesis in mice through TGF-betaRI/ALK5-dependent inhibition of endothelial cell sprouting. PLoS ONE. 2010;5:e11084.
Robbins MEC, Zhao W. Chronic oxidative stress and radiation-induced late normal tissue injury: a review. Int J Radiat Biol. 2004;80:251–9.
Greene-Schloesser D, Moore E, Robbins ME. Molecular pathways: radiation-induced cognitive impairment. Clin. Cancer Res. Off J Am Assoc Cancer Res. 2013;19:2294–300.
Fleckenstein K, et al. Temporal onset of hypoxia and oxidative stress after pulmonary irradiation. Int J Radiat Oncol. 2007;68:196–204.
Todorović A, Kasapović J, Pejić S, Stojiljković V, Pajović SB. Differences in antioxidative response of rat hippocampus and cortex after exposure to clinical dose of gamma-rays. Ann N Y Acad Sci. 2005;1048:369–72.
Smith KJ, Kapoor R, Felts PA. Demyelination: the role of reactive oxygen and nitrogen species. Brain Pathol Zurich Switz. 1999;9:69–92.
Peuchen S, et al. Interrelationships between astrocyte function, oxidative stress and antioxidant status within the central nervous system. Prog Neurobiol. 1997;52:261–81.
Institoris A, et al. Whole brain irradiation in mice causes long-term impairment in astrocytic calcium signaling but preserves astrocyte-astrocyte coupling. GeroScience. 2021;43:197–212.
Srinivasan VJ, Mandeville ET, Can A, Blasi F, Climov M, Daneshmand A, et al. Multiparametric, longitudinal optical coherence tomography imaging reveals acute injury and chronic recovery in experimental ischemic stroke. PLoS One. 2013;8(8):e71478. https://doi.org/10.1371/journal.pone.0071478.
Acknowledgements
The authors thank Waleed Tahir and Lei Tian from Boston University, and Rafat Damseh and Frédéric Lesage from École Polytechnique de Montréal, Université de Montréal, for their technical supports on the microvascular morphological analysis.
Funding
This work was supported by the grants from the American Heart Association, the Oklahoma Center for the Advancement of Science and Technology, the National Institute on Aging (RF1AG072295, R01AG055395, R01AG068295, R01AG070915, and K01AG073614), the National Institute of Neurological Disorders and Stroke (R01NS100782, R01NS091230, R01NS115401, U19NS123717, and RF1NS121095), the National Cancer Center (R01CA255840), the National Institute of Biomedical Imaging and Bioengineering (U24EB028941), the National Heart, Lung, and Blood Institute (U01HL133362), the National Institute of Mental Health (R00MH120053), the Oklahoma Shared Clinical and Translational Resources (U54GM104938) with an Institutional Development Award (IDeA) from NIGMS, the Presbyterian Health Foundation, the Reynolds Foundation, the NIA-supported Geroscience Training Program in Oklahoma (T32AG052363), the Oklahoma Nathan Shock Center (P30AG050911), the Cellular and Molecular GeroScience CoBRE (P20GM125528), and the Science and Technology Innovation Committee of Shenzhen Municipality (JSGG20210420091601003).
Author information
Authors and Affiliations
Contributions
B.L., A.Y., S.S., and Z.U. designed the study; A.Y. prepared the WBI model with the guidance from S.U., S.T., W.E.S., and A.C.; B.L., I.S., and S.E.E. performed the experiments; B.L., J.E.P., M.A.H.A., and J.L. analyzed the data with the guidance from S.S.; B.L. and S.S. interpreted the results with the help from Z.U. and D.A.B.; S.V. developed the phosphorescence nanoprobe Oxyphor-2P and helped with the PO2 measurements; S.R.A. synthetized Oxyphor-2P with the guidance from S.V.; B.L. synthetized the dextran-Alexa680 dye with the guidance from C.R.; B.L. and Z.U. wrote the manuscript with help from S.S. and all other authors; S.S. and B.L. developed the imaging and data processing methods; B.F. performed the animal surgeries.
Corresponding authors
Ethics declarations
Disclaimer
The funding sources had no role in the study design, in the collection, analysis and interpretation of data, in the writing of the report, or in the decision to submit the article for publication. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health, the American Heart Association, or the Presbyterian Health Foundation.
Conflict of interest
Dr. Anna Csiszar serves as Associate Editor for the Journal of Gerontology, Series A: Biological Sciences and Medical Sciences and GeroScience. Dr. Andriy Yabluchanskiy serves as Guest Editor for the American Journal of Physiology-Heart and Circulatory Physiology. Dr. Zoltan Ungvari serves as Editor-in-Chief for GeroScience and as Consulting Editor for the American Journal of Physiology-Heart and Circulatory Physiology. The authors declare no other competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Li, B., Yabluchanskiy, A., Tarantini, S. et al. Measurements of cerebral microvascular blood flow, oxygenation, and morphology in a mouse model of whole-brain irradiation-induced cognitive impairment by two-photon microscopy and optical coherence tomography: evidence for microvascular injury in the cerebral white matter. GeroScience 45, 1491–1510 (2023). https://doi.org/10.1007/s11357-023-00735-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-023-00735-3