Abstract
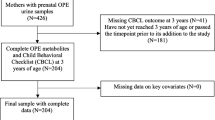
Exposure to endocrine-disrupting chemicals (EDCs) can promote infant neurodevelopmental impairment and maternal postpartum depression (PPD). However, the associations between lactation exposure to EDCs, maternal PPD, and infant neurodevelopment are unclear. Hence, we investigated these relationships in infants aged 36–42 months. We recruited 221 Korean mothers and analyzed 29 EDCs. The Edinburgh Postnatal Depression Scale (EPDS) was used to assess maternal PPD. Bayley scales of infant development; the Swanson, Nolan, and Pelham rating scale (SNAP); and the Child Behavior Checklist (CBCL) were used to assess neurodevelopment in infants exposed to the top 30% of EDC over three years. Multiple regression analyses were adjusted for maternal age, pre-pregnancy body mass index, education, income, employment, residence, and infant age and sex. The rates of infants with clinically abnormal diagnoses on neurologic developmental tests (Balyey, SNAP, and CBCL scales) ranged from 7.7 to 38.5% in this study, with the motor and hyperactivity/impulsivity areas scoring the highest among 65 boys and girls. Mono-2-ethylhexyl phthalate (MEHP) and mono-isononyl phthalate (MiNP) levels in breast milk significantly correlated with infant inattention and hyperactivity. Perfluorononanoic acid (PFNA) and perfluorooctyl sulfonate (PFOS) levels correlated significantly with motor development of BSID-III and total CBCL score which mean infant might have lower developmental status. EDC concentrations in breast milk were not associated with maternal PPD. Overall, lactational exposure to EDCs during the postpartum period can exert a negative effect on maternal PPD and infant neurodevelopment.
Similar content being viewed by others
Data availability
None.
References
Abdallah MAE, Wemken N, Drage DS, Tlustos C, Cellarius C, Cleere K, Morrison JJ, Daly S, Coggins MA, Harrad S (2020) Concentrations of perfluoroalkyl substances in human milk from Ireland: implications for adult and nursing infant exposure. Chemosphere 246:125724. https://doi.org/10.1016/j.chemosphere.2019.125724
Arbuckle TE, Fisher M, MacPherson S, Lang C, Provencher G, LeBlanc A, Hauser R, Feeley M, Ayotte P, Neisa A, Ramsay T, Tawagi G (2016) Maternal and early life exposure to phthalates: the Plastics and Personal-care Products use in Pregnancy (P4) study. Sci Total Environ 551–552:344–356. https://doi.org/10.1016/j.scitotenv.2016.02.022
Australian Breastfeeding Association. https://www.breastfeeding.asn.au/. Accessed 12 June 2022
Bever CS, Rand AA, Nording M, Taft D, Kalanetra KM, Mills DA, Breck MA, Smilowitz JT, German JB, Hammock BD (2018) Effects of triclosan in breast milk on the infant fecal microbiome. Chemosphere 203:467–473. https://doi.org/10.1016/j.chemosphere.2018.03.186
Bornehag CG, Lindh C, Reichenberg A, Wikström S, Unenge Hallerback M, Evans SF, Sathyanarayana S, Barrett ES, Nguyen RHN, Bush NR, Swan SH (2018) Association of prenatal phthalate exposure with language development in early childhood. JAMA Pediatr 172:1169–1176. https://doi.org/10.1001/jamapediatrics.2018.3115
Braun JM, Kalkbrenner AE, Calafat AM, Yolton K, Ye X, Dietrich KN, Lanphear BP (2011) Impact of early-life bisphenol A exposure on behavior and executive function in children. Pediatrics 128:873–882. https://doi.org/10.1542/peds.2011-1335
Braun JM, Bellinger DC, Hauser R, Wright RO, Chen A, Calafat AM, Yolton K, Lanphear BP (2017) Prenatal phthalate, triclosan, and bisphenol A exposures and child visual-spatial abilities. Neurotoxicology 58:75–83. https://doi.org/10.1016/j.neuro.2016.11.009
Calabrese EJ and Mattson MP (2017). How does hormesis impact biology, toxicology, and medicine? npj Aging and Mechanism of Disease. 3:13 10.1038/s41514–017–0013-z
Calafat AM, Slakman AR, Silva MJ, Herbert AR, Needham LL (2004) Automated solid phase extraction and quantitative analysis of human milk for 13 phthalate metabolites. J Chromatogr B Analyt Technol Biomed Life Sci 805:49–56. https://doi.org/10.1016/j.jchromb.2004.02.006
Caparros-Gonzalez RA, Romero-Gonzalez B, Strivens-Vilchez H, Gonzalez-Perez R, Martinez-Augustin O, Peralta-Ramirez MI (2017) Hair cortisol levels, psychological stress and psychopathological symptoms as predictors of postpartum depression. PLOS ONE 12:e0182817. https://doi.org/10.1371/journal.pone.0182817
Carrizosa C, Murcia M, Ballesteros V, Costa O, Manzano-Salgado CB, Ibarluzea J, Iñiguez C, Casas M, Andiarena A, Llop S, Lertxundi A, Schettgen T, Sunyer J, Ballester F, Vrijheid M, Lopez-Espinosa MJ (2021) Prenatal perfluoroalkyl substance exposure and neuropsychological development throughout childhood: the INMA Project. J Hazard Mater 416:125185. https://doi.org/10.1016/j.jhazmat.2021.125185
Casas M, Forns J, Martínez D, Avella-García C, Valvi D, Ballesteros-Gómez A, Luque N, Rubio S, Julvez J, Sunyer J, Vrijheid M (2015) Exposure to bisphenol A during pregnancy and child neuropsychological development in the INMA-Sabadell cohort. Environ Res 142:671–679. https://doi.org/10.1016/j.envres.2015.07.024
Chen N, Li J, Li D, Yang Y, He D (2014) Chronic exposure to perfluorooctane sulfonate induces behavior defects and neurotoxicity through oxidative damages, in vivo and in vitro. PLOS ONE 9:e113453. https://doi.org/10.1371/journal.pone.0113453
Choi SK, Kim JH, Park JK, Lee KM, Kim E, Jeon WB (2013) Cytotoxicity and inhibition of intercellular interaction in N2a neurospheroids by perfluorooctanoic acid and perfluorooctanesulfonic acid. Food Chem Toxicol 60:520–529. https://doi.org/10.1016/j.fct.2013.07.070
Cox JL, Holden JM, Sagovsky R (1987) Detection of postnatal depression. Development of the 10-item Edinburgh Postnatal Depression Scale. Br J Psychiatry 150:782–786. https://doi.org/10.1192/bjp.150.6.782
Darbre PD, Harvey PW (2008) Paraben esters: review of recent studies of endocrine toxicity, absorption, esterase and human exposure, and discussion of potential human health risks. J Appl Toxicol 28:561–578. https://doi.org/10.1002/jat.1358
Darras VM (2008) Endocrine disrupting polyhalogenated organic pollutants interfere with thyroid hormone signalling in the developing brain. Cerebellum 7:26–37. https://doi.org/10.1007/s12311-008-0004-5
Doherty BT, Engel SM, Buckley JP, Silva MJ, Calafat AM, Wolff MS (2017) Prenatal phthalate biomarker concentrations and performance on the Bayley Scales of Infant Development-II in a population of young urban children. Environ Res 152:51–58. https://doi.org/10.1016/j.envres.2016.09.021
Dong J, Cong Z, You M, Fu Y, Wang Y, Wang Y, Fu H, Wei L, Chen J (2019) Effects of perinatal di (2-ethylhexyl) phthalate exposure on thyroid function in rat offspring. Environ Toxicol Pharmacol 67:53–60. https://doi.org/10.1016/j.etap.2019.01.012
Duan C, Hare MM, Staring M, Deligiannidis KM (2019) Examining the relationship between perinatal depression and neurodevelopment in infants and children through structural and functional neuroimaging research. Int Rev Psychiatry 31:264–279. https://doi.org/10.1080/09540261.2018.1527759
Ejaredar M, Nyanza EC, Ten Eycke K, Dewey D (2015) Phthalate exposure and childrens neurodevelopment: A systematic review. Environ Res 142:51–60. https://doi.org/10.1016/j.envres.2015.06.014
Etzel T, Muckle G, Arbuckle TE, Fraser WD, Ouellet E, Séguin JR, Lanphear B, Braun JM (2018) Prenatal urinary triclosan concentrations and child neurobehavior. Environ Int 114:152–159. https://doi.org/10.1016/j.envint.2018.02.032
Evans SF, Kobrosly RW, Barrett ES, Thurston SW, Calafat AM, Weiss B, Stahlhut R, Yolton K, Swan SH (2014) Prenatal bisphenol A exposure and maternally reported behavior in boys and girls. Neurotoxicology 45:91–99. https://doi.org/10.1016/j.neuro.2014.10.003
Forns J, Iszatt N, White RA, Mandal S, Sabaredzovic A, Lamoree M, Thomsen C, Haug LS, Stigum H, Eggesbø M (2015) Perfluoroalkyl substances measured in breast milk and child neuropsychological development in a Norwegian birth cohort study. Environ Int 83:176–182. https://doi.org/10.1016/j.envint.2015.06.013
Forns J, Verner MA, Iszatt N, Nowack N, Bach CC, Vrijheid M, Costa O, Andiarena A, Sovcikova E, Høyer BB, Wittsiepe J, Lopez-Espinosa MJ, Ibarluzea J, Hertz-Picciotto I, Toft G, Stigum H, Guxens M, Liew Z, Eggesbø M (2020) Early life exposure to perfluoroalkyl substances (PFAS) and ADHD: A meta-analysis of nine European population-based studies. Environ Health Perspect 128:57002. https://doi.org/10.1289/EHP5444
Fromme H, Gruber L, Seckin E, Raab U, Zimmermann S, Kiranoglu M, Schlummer M, Schwegler U, Smolic S, Völkel W, HBMnet, (2011) Phthalates and their metabolites in breast milk—results from the Bavarian Monitoring of Breast Milk (BAMBI). Environ Int 37:715–722. https://doi.org/10.1016/j.envint.2011.02.008
Gascon M, Valvi D, Forns J, Casas M, Martínez D, Júlvez J, Monfort N, Ventura R, Sunyer J, Vrijheid M (2015) Prenatal exposure to phthalates and neuropsychological development during childhood. Int J Hyg Environ Health 218:550–558. https://doi.org/10.1016/j.ijheh.2015.05.006
Goudarzi H, Nakajima S, Ikeno T, Sasaki S, Kobayashi S, Miyashita C, Ito S, Araki A, Nakazawa H, Kishi R (2016) Prenatal exposure to perfluorinated chemicals and neurodevelopment in early infancy: the Hokkaido study. Sci Total Environ 541:1002–1010. https://doi.org/10.1016/j.scitotenv.2015.10.017
Guo J, Wu C, Zhang J, Qi X, Lv S, Jiang S, Zhou T, Lu D, Feng C, Chang X, Zhang Y, Cao Y, Wang G, Zhou Z (2020) Prenatal exposure to mixture of heavy metals, pesticides and phenols and IQ in children at 7 years of age: the SMBCS study. Environ Int 139:105692. https://doi.org/10.1016/j.envint.2020.105692
Hamid N, Junaid M, Pei DS (2021) Combined toxicity of endocrine-disrupting chemicals: a review. Ecotoxicol Environ Saf 215:112136. https://doi.org/10.1016/j.ecoenv.2021.112136
Harris MH, Oken E, Rifas-Shiman SL, Calafat AM, Ye X, Bellinger DC, Webster TF, White RF, Sagiv SK (2018) Prenatal and childhood exposure to per- and polyfluoroalkyl substances (PFASs) and child cognition. Environ Int 115:358–369. https://doi.org/10.1016/j.envint.2018.03.025
Holahan MR, Smith CA (2015) Phthalates and neurotoxic effects on hippocampal network plasticity. Neurotoxicology 48:21–34. https://doi.org/10.1016/j.neuro.2015.02.008
Hornung RW, Reed LD (1990) Estimation of average concentration in the presence of nondetectable values. Appl Occup Environ Hyg 5:46–51. https://doi.org/10.1080/1047322X.1990.10389587
Jacobson MH, Stein CR, Liu M, Ackerman MG, Blakemore JK, Long SE, Pinna G, Romay-Tallon R, Kannan K, Zhu H, Trasande L (2021) Prenatal exposure to bisphenols and phthalates and postpartum depression: the role of neurosteroid hormone disruption. J Clin Endocrinol Metab 106:1887–1899. https://doi.org/10.1210/clinem/dgab199
Jacobson MH, Ghassabian A, Gore AC, Trasande L (2022) Exposure to environmental chemicals and perinatal psychopathology. Biochem Pharmacol 195:114835. https://doi.org/10.1016/j.bcp.2021.114835
Jankowska A, Polańska K, Hanke W, Wesołowska E, Ligocka D, Waszkowska M, Stańczak A, Tartaglione AM, Mirabella F, Chiarotti F, Garí M, Calamandrei G (2019) Prenatal and early postnatal phthalate exposure and child neurodevelopment at age of 7 years—Polish Mother and Child Cohort. Environ Res 177:108626. https://doi.org/10.1016/j.envres.2019.108626
Jiang Y, Zhao H, Xia W, Li Y, Liu H, Hao K, Chen J, Sun X, Liu W, Li J, Peng Y, Hu C, Li C, Zhang B, Lu S, Cai Z, Xu S (2019) Prenatal exposure to benzophenones, parabens and triclosan and neurocognitive development at 2 years. Environ Int 126:413–421. https://doi.org/10.1016/j.envint.2019.01.023
Johansson N, Fredriksson A, Eriksson P (2008) Neonatal exposure to perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) causes neurobehavioural defects in adult mice. Neurotoxicology 29:160–169. https://doi.org/10.1016/j.neuro.2007.10.008
Kang H, Choi K, Lee HS, Kim DH, Park NY, Kim S, Kho Y (2016) Elevated levels of short carbon-chain PFCAs in breast milk among Korean women: current status and potential challenges. Environ Res 148:351–359. https://doi.org/10.1016/j.envres.2016.04.017
Kapadia R, Yi JH, Vemuganti R (2008) Mechanisms of anti-inflammatory and neuroprotective actions of PPAR-gamma agonists. Front Biosci 13:1813–1826. https://doi.org/10.2741/2802
Kim SK, Lee KT, Kang CS, Tao L, Kannan K, Kim KR, Kim CK, Lee JS, Park PS, Yoo YW, Ha JY, Shin YS, Lee JH (2011) Distribution of perfluorochemicals between sera and milk from the same mothers and implications for prenatal and postnatal exposures. Environ Pollut 159:169–174. https://doi.org/10.1016/j.envpol.2010.09.008
Kim S, Eom S, Kim HJ, Lee JJ, Choi G, Choi S, Kim S, Kim SY, Cho G, Kim YD, Suh E, Kim SK, Kim S, Kim GH, Moon HB, Park J, Kim S, Choi K, Eun SH (2018) Association between maternal exposure to major phthalates, heavy metals, and persistent organic pollutants, and the neurodevelopmental performances of their children at 1 to 2 years of age- CHECK cohort study. Sci Total Environ 624:377–384. https://doi.org/10.1016/j.scitotenv.2017.12.058
Kim D, Kim JH, Seo S (2020a) Real-time measurement of indoor PM concentrations on daily change of endocrine disruptors in urine samples of new mothers. Sustainability 12:6166. https://doi.org/10.3390/su12156166
Kim JH, Kim D, Moon SM, Yang EJ (2020b) Associations of lifestyle factors with phthalate metabolites, bisphenol A, parabens, and triclosan concentrations in breast milk of Korean mothers. Chemosphere 249:126149. https://doi.org/10.1016/j.chemosphere.2020.126149
Kim JH, Kwak JM, Kang H (2021a) Web-based behavioral intervention to reduce exposure to phthalate metabolites, bisphenol A, triclosan, and parabens in mothers with young children: A randomized controlled trial. Int J Hyg Environ Health 236:113798. https://doi.org/10.1016/j.ijheh.2021.113798
Kim JH, Shin HS, Lee WH (2021b) Impact of endocrine-disrupting chemicals in breast milk on postpartum depression in Korean mothers. Int J Environ Res Public Health 18:4444. https://doi.org/10.3390/ijerph18094444
Kim JH, Moon N, Lee JW, Mehdi Q, Yun MH, Moon HB (2023) Time-course trend and influencing factors for per- and polyfluoroalkyl substances in the breast milk of Korean mothers. Chemosphere 310:136688. https://doi.org/10.1016/j.chemosphere.2022.136688
Korea FDA (2015). Risk Assess PFOA PFOS:11–1471057–000126–01
Kougias DG, Sellinger EP, Willing J, Juraska JM (2018) Perinatal exposure to an environmentally relevant mixture of phthalates results in a lower number of neurons and synapses in the medial prefrontal cortex and decreased cognitive flexibility in adult male and female rats. J Neurosci 38:6864–6872. https://doi.org/10.1523/JNEUROSCI.0607-18.2018
Lanpher BP (2017) Low-level toxicity of chemicals: no acceptable levels? PLoS Biol 15(12):e2003066. https://doi.org/10.1371/journal.pbio.2003066
Latini G, Wittassek M, Del Vecchio A, Presta G, De Felice C, Angerer J (2009) Lactational exposure to phthalates in Southern Italy. Environ Int 35:236–239. https://doi.org/10.1016/j.envint.2008.06.002
Lee D (2018) Evidence of the possible of endocrine disrupting chemicals in human: ongoing debates and key issues. Endocrinol Metab 33:44–52. https://doi.org/10.3803/EnM.2018.33.1.44
Lee JY, Kim YT, Bang HJ, Lee SH (2014) A Preliminary study on standardizing the K-Bayley-III expressive communication scale. Commun SciDisorders 19:320–330. https://doi.org/10.12963/csd.14138
Lee DW, Kim MS, Lim YH, Lee N, Hong YC (2018a) Prenatal and postnatal exposure to di-(2-ethylhexyl) phthalate and neurodevelopmental outcomes: A systematic review and meta-analysis. Environ Res 167:558–566. https://doi.org/10.1016/j.envres.2018.08.023
Lee S, Kim S, Park J, Kim HJ, Choi G, Choi S, Kim S, Kim SY, Kim S, Choi K, Moon HB (2018b) Perfluoroalkyl substances (PFASs) in breast milk from Korea: time-course trends, influencing factors, and infant exposure. Sci Total Environ 612:286–292. https://doi.org/10.1016/j.scitotenv.2017.08.094
Li Z, Liu Q, Liu C, Li C, Li Y, Li S, Liu X, Shao J (2017) Evaluation of PFOS-mediated neurotoxicity in rat primary neurons and astrocytes cultured separately or in co-culture. Toxicol in Vitro 38:77–90. https://doi.org/10.1016/j.tiv.2016.11.002
Lien YJ, Ku HY, Su PH, Chen SJ, Chen HY, Liao PC, Chen WJ, Wang SL (2015) Prenatal exposure to phthalate esters and behavioral syndromes in children at 8 years of age: Taiwan Maternal and Infant Cohort Study. Environ Health Perspect 123:95–100. https://doi.org/10.1289/ehp.1307154
Lim YH, Bae S, Kim BN, Shin CH, Lee YA, Kim JI, Hong YC (2017) Prenatal and postnatal bisphenol A exposure and social impairment in 4-year-old children. Environ Health 16:79. https://doi.org/10.1186/s12940-017-0289-2
Lubotzky-Gete S, Ornoy A, Grotto I, Calderon-Margalit R (2021) Postpartum depression and infant development up to 24 months: A nationwide population-based study. J Affect Disord 285:136–143. https://doi.org/10.1016/j.jad.2021.02.042
Macheka LR, Abafe OA, Mugivhisa LL, Olowoyo JO (2022) Occurrence and infant exposure assessment of per and polyfluoroalkyl substances in breast milk from South Africa. Chemosphere 288:132601. https://doi.org/10.1016/j.chemosphere.2021.132601
Matthews M, Nigg JT, Fair DA (2014) Attention deficit hyperactivity disorder. Curr Top Behav Neurosci 16:235–266. https://doi.org/10.1007/7854_2013_249
Minatoya M, Kishi R (2021) A review of recent studies on bisphenol A and phthalate exposures and child neurodevelopment. Int J Environ Res Public Health 18:3585. https://doi.org/10.3390/ijerph18073585
Miodovnik A, Edwards A, Bellinger DC, Hauser R (2014) Developmental neurotoxicity of Ortho-phthalate diesters: review of human and experimental evidence. Neurotoxicology 41:112–122. https://doi.org/10.1016/j.neuro.2014.01.007
Murray L, Cooper PJ (1997) Postpartum depression and child development. Psychol Med 27:253–260. https://doi.org/10.1017/s0033291796004564
Neugebauer J, Wittsiepe J, Kasper-Sonnenberg M, Schöneck N, Schölmerich A, Wilhelm M (2015) The influence of low level pre- and perinatal exposure to PCDD/Fs, PCbs, and lead on attention performance and attention-related behavior among German school-aged children: results from the Duisburg Birth Cohort Study. Int J Hyg Environ Health 218:153–162. https://doi.org/10.1016/j.ijheh.2014.09.005
Oh KJ, Lee HL, Hong KE, Ha EH (1997) Korean version of Child Behavior Checklist (K-CBCL). ChungAng Aptitude Publishing, Seoul, Korea
Oh KJ, Kim YA (2009) Huno consulting, Korean version of the child behavior checklist for ages 1.5–5. Seoul, Korea
Oulhote Y, Steuerwald U, Debes F, Weihe P, Grandjean P (2016) Behavioral difficulties in 7-year old children in relation to developmental exposure to perfluorinated alkyl substances. Environ Int 97:237–245. https://doi.org/10.1016/j.envint.2016.09.015
Pan R, Wang C, Shi R, Zhang Y, Wang Y, Cai C, Ding G, Yuan T, Tian Y, Gao Y (2019) Prenatal bisphenol A exposure and early childhood neurodevelopment in Shandong, China. Int J Hyg Environ Health 222:896–902. https://doi.org/10.1016/j.ijheh.2019.03.002
Polanska K, Ligocka D, Sobala W, Hanke W (2014) Phthalate exposure and child development: the Polish Mother and Child Cohort Study. Early Hum Dev 90:477–485. https://doi.org/10.1016/j.earlhumdev.2014.06.006
Power MC, Webster TF, Baccarelli AA, Weisskopf MG (2013) Cross-sectional association between polyfluoroalkyl chemicals and cognitive limitation in the national health and nutrition examination survey. Neuroepidemiology 40:125–132. https://doi.org/10.1159/000342310
Qian X, Li J, Xu S, Wan Y, Li Y, Jiang Y, Zhao H, Zhou Y, Liao J, Liu H, Sun X, Liu W, Peng Y, Hu C, Zhang B, Lu S, Cai Z, Xia W (2019) Prenatal exposure to phthalates and neurocognitive development in children at two years of age. Environ Int 131:105023. https://doi.org/10.1016/j.envint.2019.105023
Rochester JR, Bolden AL, Kwiatkowski CF (2018) Prenatal exposure to bisphenol A and hyperactivity in children: A systematic review and meta-analysis. Environ Int 114:343–356. https://doi.org/10.1016/j.envint.2017.12.028
Schiller CE, Meltzer-Brody S, Rubinow DR (2015) The role of reproductive hormones in postpartum depression. CNS Spectr 20:48–59. https://doi.org/10.1017/S1092852914000480
Skogheim TS, Weyde KVF, Aase H, Engel SM, Surén P, Øie MG, Biele G, Reichborn-Kjennerud T, Brantsæter AL, Haug LS, Sabaredzovic A, Auyeung B, Villanger GD (2021) Prenatal exposure to per- and polyfluoroalkyl substances (PFAS) and associations with attention-deficit/hyperactivity disorder and autism spectrum disorder in children. Environ Res 202:111692. https://doi.org/10.1016/j.envres.2021.111692
Spratlen MJ, Perera FP, Lederman SA, Rauh VA, Robinson M, Kannan K, Trasande L, Herbstman J (2020) The association between prenatal exposure to perfluoroalkyl substances and childhood neurodevelopment. Environ Pollut 263:114444. https://doi.org/10.1016/j.envpol.2020.114444
Stein CR, Savitz DA, Bellinger DC (2013) Perfluorooctanoate and neuropsychological outcomes in children. Epidemiology 24:590–599. https://doi.org/10.1097/EDE.0b013e3182944432
Strøm M, Hansen S, Olsen SF, Haug LS, Rantakokko P, Kiviranta H, Halldorsson TI (2014) Persistent organic pollutants measured in maternal serum and offspring neurodevelopmental outcomes—A prospective study with long-term follow-up. Environ Int 68:41–48. https://doi.org/10.1016/j.envint.2014.03.002
Swanson JM, Kraemer HC, Hinshaw SP, Arnold LE, Conners CK, Abikoff HB, Clevenger W, Davies M, Elliott GR, Greenhill LL, Hechtman L, Hoza B, Jensen PS, March JS, Newcorn JH, Owens EB, Pelham WE, Schiller E, Severe JB, Simpson S, Vitiello B, Wells K, Wigal T, Wu M (2001) Clinical relevance of the primary findings of the MTA: success rates based on severity of ADHD and ODD symptoms at the end of treatment. J Am Acad Child Adolesc Psychiatry 40:168–179. https://doi.org/10.1097/00004583-200102000-00011
Téllez-Rojo MM, Cantoral A, Cantonwine DE, Schnaas L, Peterson K, Hu H, Meeker JD (2013) Prenatal urinary phthalate metabolites levels and neurodevelopment in children at two and three years of age. Sci Total Environ 461–462:386–390. https://doi.org/10.1016/j.scitotenv.2013.05.021
Tran NQV, Miyake K (2017) Neurodevelopmental disorders and environmental toxicants: epigenetics as an underlying mechanism. Int J Genomics 2017:7526592. https://doi.org/10.1155/2017/7526592
Tsai MS, Chen MH, Lin CC, Ng S, Hsieh CJ, Liu CY, Hsieh WS, Chen PC (2017) Children’s environmental health based on birth cohort studies of Asia. Sci Total Environ 609:396–409. https://doi.org/10.1016/j.scitotenv.2017.07.081
UvnäsMoberg K, Ekström-Bergström A, Buckley S, Massarotti C, Pajalic Z, Luegmair K, Kotlowska A, Lengler L, Olza I, Grylka-Baeschlin S, Leahy-Warren P, Hadjigeorgiu E, Villarmea S, Dencker A (2020) Maternal plasma levels of oxytocin during breastfeeding—A systematic review. PLOS ONE 15:e0235806. https://doi.org/10.1371/journal.pone.0235806
Vanden Heuvel JP, Thompson JT, Frame SR, Gillies PJ (2006) Differential activation of nuclear receptors by perfluorinated fatty acid analogs and natural fatty acids: A comparison of human, mouse, and rat peroxisome proliferator-activated receptor-alpha, -beta, and -gamma, liver X receptor-beta, and retinoid X receptor-alpha. Toxicol Sci 92:476–489. https://doi.org/10.1093/toxsci/kfl014
Vuong AM, Yolton K, Braun JM, Sjodin A, Calafat AM, Xu Y, Dietrich KN, Lanphear BP, Chen A (2020) Polybrominated diphenyl ether (PBDE) and poly- and perfluoroalkyl substance (PFAS) exposures during pregnancy and maternal depression. Environ Int 139:105694. https://doi.org/10.1016/j.envint.2020.105694
Walf AA, Frye CA (2006) A review and update of mechanisms of estrogen in the hippocampus and amygdala for anxiety and depression behavior. Neuropsychopharmacology 31:1097–1111. https://doi.org/10.1038/sj.npp.1301067
WHO. https://www.who.int/westernpacific/health-topics/breastfeeding. Accessed 12 June 2022
Xu Y, Agrawal S, Cook TJ, Knipp GT (2007) Di-(2-ethylhexyl)-phthalate affects lipid profiling in fetal rat brain upon maternal exposure. Arch Toxicol 81:57–62. https://doi.org/10.1007/s00204-006-0143-8
Xu X, Hong X, Xie L, Li T, Yang Y, Zhang Q, Zhang G, Liu X (2012) Gestational and lactational exposure to bisphenol-A affects anxiety- and depression-like behaviors in mice. Horm Behav 62:480–490. https://doi.org/10.1016/j.yhbeh.2012.08.005
Xu X, Yang Y, Wang R, Wang Y, Ruan Q, Lu Y (2015) Perinatal exposure to di-(2-ethylhexyl) phthalate affects anxiety- and depression-like behaviors in mice. Chemosphere 124:22–31. https://doi.org/10.1016/j.chemosphere.2014.10.056
Ye X, Bishop AM, Needham LL, Calafat AM (2008) Automated on-line column-switching HPLC-MS/MS method with peak focusing for measuring parabens, triclosan, and other environmental phenols in human milk. Anal Chim Acta 622:150–156. https://doi.org/10.1016/j.aca.2008.05.068
Yesumanipreethi S, Nirmal Magadalenal N, Moses Inbaraj R (2021) Impact of phthalates and parabens on the neurobehavioral and reproductive function: a review. Proc Zool Soc 74:572–590. https://doi.org/10.1007/s12595-021-00408-z
Zheng G, Schreder E, Dempsey JC, Uding N, Chu V, Andres G, Sathyanarayana S, Salamova A (2021) Per- and polyfluoroalkyl substances (PFAS) in breast milk: concerning trends for current-use PFAS. Environ Sci Technol 55:7510–7520. https://doi.org/10.1021/acs.est.0c06978
Acknowledgements
The authors thank the mothers and infants who participated in this study. We would also like to acknowledge the efforts of Myoug-Hee Yun and the nurses who collected the samples at the Moyusarang Lactation Consultant Clinic and Jinhyuk Lim (Korea Cognitive Development Institute), Heeju Ha (Korea Cognitive Development Institute), Eunkyeong Lee (Daum Clinical Counseling Center), Seoyeong Jeong (Daum Clinical Counseling Center), Gui Nam Seo (Zhen-i Children Family Counseling Center), Jungin Choi (Jungin Clinic), and Sooyeon Lee (clinical psychologist).
Funding
This research was funded by the National Research Foundation of Korea, funded by the Korean Ministry of Science, ICT, grant number NRF-2021R1A2C4001788.
Author information
Authors and Affiliations
Contributions
Conceptualization, JHK and EJ; methodology, JHK, HM and NM; software, JHK and NM; validation, JHK, NM, and HM; formal analysis, NM, and HM; investigation, JHK, EJ, NM, and HM; resources, JHK; data curation, JHK and NM; writing—original draft preparation, JHK, EJ, NM, and HM; writing—review and editing, JHK, EJ, NM, and HM; visualization, JHK and NM; supervision, JHK; project administration, JHK; funding acquisition, JHK. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Institutional review board statement
This study was approved by the Institutional Review Board of Kyung Hee University, South Korea (approval number KHSIRB-18–029).
Informed consent statement
Informed consent was obtained from all subjects involved in the study.
Conflicts of interest
The authors declare no conflict of interest.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, J.H., Moon, N., Ji, E. et al. Effects of postnatal exposure to phthalate, bisphenol a, triclosan, parabens, and per- and poly-fluoroalkyl substances on maternal postpartum depression and infant neurodevelopment: a korean mother-infant pair cohort study. Environ Sci Pollut Res 30, 96384–96399 (2023). https://doi.org/10.1007/s11356-023-29292-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-29292-0