Abstract
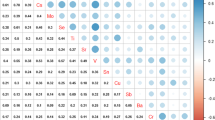
In the real world, humans are exposed to multiple metal(loid)s (designated hereafter metals) that contain essential metals as well as toxic metals. Exposure to the metal mixture was assumed to be associated with renal function impairment; however, there is no consensus on available studies. Therefore, we here explored the association between multiple metals exposure and indicators of renal function in the general population from southeastern China. A total of 11 metals with 6 human essential metals and 5 toxic metals were determined in the selected 720 subjects. In addition, serum uric acid (SUA), serum creatinine (SCR), and the estimated glomerular filtration rate (eGFR) were measured or calculated as indicators of renal function. Using multiple flexible statistical models of generalized linear model, elastic net regression, and Bayesian kernel machine regression, the joint as well as the individual effect of metals within the mixture, and the interactions between metals were explored. When exposed to the metal mixture, the statistically non-significantly increased SUA, the significantly increased SCR, and the significantly declined eGFR were observed. In addition, the declined renal function may be primarily attributed to lead (Pb), arsenic (As), and nickel (Ni) exposure. Finally, interactions, such as the synergistic effect between Pb and Mo on SUA, whereas the antagonistic effect between Ni and Cd on SCR and eGFR were identified. Our finding suggests that combined exposure to multiple metals would impair renal function. Therefore, reducing exposure to toxic heavy metals of Pb, As, and Cd and limiting exposure to the human essential metal of Ni would protect renal function.






Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study were not publicly available but are available from the corresponding author upon reasonable request.
References
Agency for Toxic Substances and Disease Registry A (2022) The ATSDR 2022 substance priority list. ATSDR https://www.atsdr.cdc.gov/spl/index.html#2022spl
Ali S et al (2019) Renal toxicity of heavy metals (cadmium and mercury) and their amelioration with ascorbic acid in rabbits. Environ Sci Pollut Res 26:3909–3920. https://doi.org/10.1007/s11356-018-3819-8
Arabi T et al (2022) Obesity-related kidney disease: beyond hypertension and insulin-resistance. Front Endocrinol (lausanne) 13:1095211. https://doi.org/10.3389/fendo.2022.1095211
CDC (2022) Biomonitoring Data Tables for Environmental Chemicals, https://www.cdc.gov/exposurereport/data_tables.html
Collaboration GCKD (2020) Global, regional, and national burden of chronic kidney disease, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 395:709–733. https://doi.org/10.1016/s0140-6736(20)30045-3
Delanaye P et al (2022) The new 2021 CKD-EPI equation without race in a European Cohort of renal transplanted patients. Transplantation 106:2443–2447. https://doi.org/10.1097/tp.0000000000004234
Foreman KJ et al (2018) Forecasting life expectancy, years of life lost, and all-cause and cause-specific mortality for 250 causes of death: reference and alternative scenarios for 2016–40 for 195 countries and territories. Lancet 392:2052–2090. https://doi.org/10.1016/s0140-6736(18)31694-5
Friedman J, Hastie T, Tibshirani R (2010) Regularization paths for generalized linear models via coordinate descent. J Stat Softw 33:1–22. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2929880/
Gao W et al (2023) Exposure to cadmium, lead, mercury, and arsenic and uric acid levels: results from NHANES 2007–2016. Biol Trace Elem Res 201:1659–1669. https://doi.org/10.1007/s12011-022-03309-0
Guo J et al (2020) Prenatal exposure to mixture of heavy metals, pesticides and phenols and IQ in children at 7 years of age: The SMBCS study. Environ Int 139:105692. https://doi.org/10.1016/j.envint.2020.105692
Hamrahian SM et al (2017) Hypertension in chronic kidney disease. Adv Exp Med Biol 956:307–325. https://doi.org/10.1007/5584_2016_84
Hoste L et al (2014) A new equation to estimate the glomerular filtration rate in children, adolescents and young adults. Nephrol Dial Transplant 29:1082–1091. https://doi.org/10.1093/ndt/gft277
Inker LA et al (2021) New creatinine- and cystatin C-based equations to estimate GFR without race. N Engl J Med 385:1737–1749. https://doi.org/10.1056/NEJMoa2102953
Jin R et al (2018) Associations of renal function with urinary excretion of metals: Evidence from NHANES 2003–2012. Environ Int 121:1355–1362. https://doi.org/10.1016/j.envint.2018.11.002
Joo SH et al (2022) Environmental exposure to lead, mercury, and cadmium is not associated with abnormal kidney function in Korean adolescents. Pediatr Nephrol 37:625–631. https://doi.org/10.1007/s00467-021-05215-4
Kalantar-Zadeh K et al (2021) Chronic kidney disease. The Lancet 398:786–802. https://doi.org/10.1016/S0140-6736(21)00519-5
Kawada T (2022) Blood cadmium and hyperuricemia in adults with special reference to sex difference. Biol Trace Elem Res 200:1561. https://doi.org/10.1007/s12011-021-02791-2
Lee K et al (2021) Prenatal and postnatal exposures to four metals mixture and IQ in 6-year-old children: a prospective cohort study in South Korea. Environ Int 157:106798. https://doi.org/10.1016/j.envint.2021.106798
Li A et al (2022) Novel strategies for assessing associations between selenium biomarkers and cardiometabolic risk factors: concentration, visit-to-visit variability, or individual mean? Evidence From a Repeated-Measures Study of Older Adults With High Selenium. Front Nutr 9:838613. https://doi.org/10.3389/fnut.2022.838613
Liang LX et al (2022) Joint effects of per- and polyfluoroalkyl substance alternatives and heavy metals on renal health: A community-based population study in China. Environ Res 219:115057. https://doi.org/10.1016/j.envres.2022.115057
Liu W et al (2022) Assessment for the associations of twenty-three metal(loid)s exposures with early cardiovascular damage among Chinese urban adults with five statistical methods: Insight into assessing health effect of multipollutant exposure. Chemosphere 307:135969. https://doi.org/10.1016/j.chemosphere.2022.135969
Liu W et al (2023) Exposures to volatile organic compounds, serum vitamin D, and kidney function: association and interaction assessment in the US adult population. Environ Sci Pollut Res Int 30:7605–7616. https://doi.org/10.1007/s11356-022-22637-1
Luo X et al (2023) Associations between co-exposure to multiple metals and renal function: a cross-sectional study in Guangxi, China. Environ Sci Pollut Res Int 30:2637–2648. https://doi.org/10.1007/s11356-022-22352-x
Use of generalized weighted quantile sum regressions of tumor necrosis factor alpha and kidney function to explore joint effects of multiple metals in blood. Int J Environ Res Public Health 19.https://doi.org/10.3390/ijerph19127399
Ma Y et al (2022) Combined exposure to multiple metals on serum uric acid in NHANES under three statistical models. Chemosphere 301:134416. https://doi.org/10.1016/j.chemosphere.2022.134416
Mazidi M et al (2022) Genetically determined blood lead is associated with reduced renal function amongst individuals with type 2 diabetes mellitus: insight from Mendelian Randomisation. J Mol Med (berl) 100:125–134. https://doi.org/10.1007/s00109-021-02152-5
Meng XL et al (2022) The association between essential trace element mixture and atherosclerotic cardiovascular disease risk among Chinese community-dwelling older adults. Environ Sci Pollut Res Int 29:90351–90363. https://doi.org/10.1007/s11356-022-22066-0
Nan Y et al (2022) Associations of nickel exposure and kidney function in U.S. adults, NHANES 2017–2018. J Trace Elem Med Biol 74:127065. https://doi.org/10.1016/j.jtemb.2022.127065
Park Y, et al (2022) Association of blood heavy metal levels and renal function in Korean adults. Int J Environ Res Public Health 19. https://doi.org/10.3390/ijerph19116646
Pottel H et al (2016) An estimated glomerular filtration rate equation for the full age spectrum. Nephrol Dial Transplant 31:798–806. https://doi.org/10.1093/ndt/gfv454
Ragsdale SW (1998) Nickel biochemistry. Curr Opin Chem Biol 2:208–215. https://doi.org/10.1016/s1367-5931(98)80062-8
Sabolić I et al (2010) Role of metallothionein in cadmium traffic and toxicity in kidneys and other mammalian organs. Biometals 23:897–926. https://doi.org/10.1007/s10534-010-9351-z
Saito M, et al (2016) Molecular Mechanisms of Nickel Allergy. Int J Mol Sci 17. https://doi.org/10.3390/ijms17020202
Sanders AP et al (2019) Combined exposure to lead, cadmium, mercury, and arsenic and kidney health in adolescents age 12–19 in NHANES 2009–2014. Environ Int 131:104993. https://doi.org/10.1016/j.envint.2019.104993
Satarug S et al (2010) Cadmium, environmental exposure, and health outcomes. Environ Health Perspect 118:182–190. https://doi.org/10.1289/ehp.0901234
Shiue I et al (2014) Higher urinary heavy metal, phthalate and arsenic concentrations accounted for 3–19% of the population attributable risk for high blood pressure: US NHANES, 2009–2012. Hypertens Res 37:1075–1081. https://doi.org/10.1038/hr.2014.121
Skröder H et al (2015) Kidney function and blood pressure in preschool-aged children exposed to cadmium and arsenic—potential alleviation by selenium. Environ Res 140:205–213. https://doi.org/10.1016/j.envres.2015.03.038
Su F et al (2022) Co-exposure to perfluoroalkyl acids and heavy metals mixtures associated with impaired kidney function in adults: a community-based population study in China. Sci Total Environ 839:156299. https://doi.org/10.1016/j.scitotenv.2022.156299
Tsai HJ et al (2021) Environmental pollution and chronic kidney disease. Int J Med Sci 18:1121–1129. https://doi.org/10.7150/ijms.51594
Wang T et al (2022a) Associations between plasma metal levels and mild renal impairment in the general population of Southern China. Ecotoxicol Environ Saf 247:114209. https://doi.org/10.1016/j.ecoenv.2022.114209
Wang T et al (2022b) Associations of plasma multiple metals with risk of hyperuricemia: a cross-sectional study in a mid-aged and older population of China. Chemosphere 287:132305. https://doi.org/10.1016/j.chemosphere.2021.132305
Weaver DJ Jr (2019) Uric acid and progression of chronic kidney disease. Pediatr Nephrol 34:801–809. https://doi.org/10.1007/s00467-018-3979-2
Weaver VM et al (2011a) Associations of low-level urine cadmium with kidney function in lead workers. Occup Environ Med 68:250–256. https://doi.org/10.1136/oem.2010.056077
Weaver VM et al (2011b) Differences in urine cadmium associations with kidney outcomes based on serum creatinine and cystatin C. Environ Res 111:1236–1242. https://doi.org/10.1016/j.envres.2011.07.012
Xie Y et al (2018) Analysis of the global burden of disease study highlights the global, regional, and national trends of chronic kidney disease epidemiology from 1990 to 2016. Kidney Int 94:567–581. https://doi.org/10.1016/j.kint.2018.04.011
Xu J et al (2022) Associations of metal exposure with hyperuricemia and gout in general adults. Front Endocrinol (lausanne) 13:1052784. https://doi.org/10.3389/fendo.2022.1052784
Yin H et al (2021) Nickel induces autophagy via PI3K/AKT/mTOR and AMPK pathways in mouse kidney. Ecotoxicol Environ Saf 223:112583. https://doi.org/10.1016/j.ecoenv.2021.112583
Yu L et al (2022) A review of practical statistical methods used in epidemiological studies to estimate the health effects of multi-pollutant mixture. Environ Pollut 306:119356. https://doi.org/10.1016/j.envpol.2022.119356
Zeng A et al (2022) Association between low-level blood cadmium exposure and hyperuricemia in the American general population: a cross-sectional study. Biol Trace Elem Res 200:560–567. https://doi.org/10.1007/s12011-021-02700-7
Zhou F et al (2018) Lead, cadmium, arsenic, and mercury combined exposure disrupted synaptic homeostasis through activating the Snk-SPAR pathway. Ecotoxicol Environ Saf 163:674–684. https://doi.org/10.1016/j.ecoenv.2018.07.116
Zhou F et al (2019) Toxicity assessment due to prenatal and lactational exposure to lead, cadmium and mercury mixtures. Environ Int 133:105192. https://doi.org/10.1016/j.envint.2019.105192
Zhou F et al (2020) Insights into cognitive deficits caused by low-dose toxic heavy metal mixtures and their remediation through a postnatal enriched environment in rats. J Hazard Mater 388:122081. https://doi.org/10.1016/j.jhazmat.2020.122081
Acknowledgements
All participants included in our study and all members of our study team are greatly acknowledged.
Funding
This research was supported by grants from the National Natural Science Foundation of China (Nos. 21966022, 81903360), the Key Program of Natural Science Foundation of Jiangxi Province (No. 20192ACB20025), the Natural Science Foundation of Jiangxi Province (No. 20192BAB215048), and the Science and Technology Program of Jiangxi Province Health Commission (No. 202311106).
Author information
Authors and Affiliations
Contributions
Conceptualization: Guangqin Fan, Guihua Du, and Fankun Zhou; methodology: Guangqin Fan, Guihua Du, Xiaoguang Song, Fankun Zhou, Jie Xie, and Feng Chang; investigation: Xiaoguang Song, Lu Ouyang; Qi Li, Shiying Ruan, Shaoqi Rao, Shuo Yang, and Xin Wan; formal analysis: Guangqin Fan and Guihua Du; data visualization: Guihua Du; resources: Guangqin Fan and Xiaoguang Song; writing—original draft preparation: Guihua Du; writing—review and editing: Guangqin Fan; supervision: Guangqin Fan; funding acquisition: Guangqin Fan, Fankun Zhou, and Xiaoguang Song.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All participants in this study signed informed consent by themselves or their parents if the subjects were younger than 18 years old. This study was approved by the ethical review committee of the National Institute of Environmental Health, Chinese Center for Disease Control and Prevention.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information

Fig. S1
Grouped scatter plot of the Q(height) based eGFR (Q(height)-eGFR) and eGFR for the full age spectrum (FAS-eGFR) (PNG 131 kb)

Fig. S2
Estimated coefficients and relative 95%CI between single metal exposure and level of SUA (a), SCR (b), or eGFR (c) via generalized linear regression (GLM). In this model, the control covariates were age, gender, BMI, education level, household income, marriage status, smoking or passive smoking, and drinking. *(**) represents P<0.05(0.01) (PNG 120 kb)

Fig. S3
Bi-variate exposure-response functions of the metal mixture and level of SUA (a), SCR (b), or eGFR (c). This figure shows the association of each pair of the 11 metals with levels of SUA, SCR, or eGFR when all other heavy metals are fixed at their 50th percentile (PNG 1063 kb)

Fig. S4
Estimated coefficients and relative 95%CI between metal exposure and level of SUA (a), SCR (b), or eGFR (c) via GLM. In this model, the 10 metals, diabetes, hypertension, as well as the 8 factors that were mentioned in the covariates of method section were included as covariates when assessing the effect on the level of SUA, SCR, or eGFR. *(**) represents P<0.05(0.01) (PNG 98 kb)

Fig. S5
The univariate relationship between each metal and SUA(a-b), SCR(c-d), or eGFR (e-f) level by using BKMR for sensitivity analysis. The univariate relationship between each metal exposure and the SUA (a), SCR (c), or eGFR (e), where levels of all other metals are fixed to the 50th quantile. The shaded part was 95% CI for the curve. The estimated effect of single metal and relative 95% CI for the level of SUA (b), SCR (d), or eGFR(f) when levels of other metals were fixed at various quantiles. In the model of BKMR for sensitivity analysis, diabetes, and hypertension were included as covariates (PNG 565 kb)

Fig. S6
Joint effect and 95%CI of the 11 metals on level of SUA (a), SCR (b), or eGFR (c) when all the metals at particular percentiles were compared to their 50th percentile in the sensitivity analysis (PNG 127 kb)

Fig. S7
Bi-variate exposure-response functions of the metal mixture and level of SUA (a), SCR (b), or eGFR (c) in the sensitivity analysis. This figure shows the association of each pair of the 11 metals with levels of SUA, SCR, or eGFR when all other heavy metals are fixed at their 50th percentile (PNG 1165 kb)
ESM 8
(DOCX 17 kb)
ESM 9
(DOCX 16 kb)
ESM 10
(DOCX 18 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Du, G., Song, X., Zhou, F. et al. Association between multiple metal(loid)s exposure and renal function: a cross-sectional study from southeastern China. Environ Sci Pollut Res 30, 94552–94564 (2023). https://doi.org/10.1007/s11356-023-29001-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-29001-x