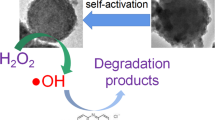
Abstract
In this paper, magnetic MnFe2O4/mSiO2 nanocomposites were successfully synthesized, and the activation performance of the materials for persulfate was evaluated by the degradation efficiency of perfluorooctanoic acid. The structure of the catalyst was proved to be a core–shell structure by several characterization methods. The mesoporous silicon coating can effectively avoid the agglomeration of MnFe2O4 and at the same time increase the contact area with the reactants. A comparison of different catalyst addition conditions demonstrates that MnFe2O4/mSiO2 can effectively activate the persulfate. The optimal reaction conditions were investigated by several key influencing factors. It was experimentally demonstrated that about 90% of PFOA (10 mg·L−1) could be decomposed under the conditions of 0.4 g·L−1 MnFe2O4/mSiO2 and PS, pH 5.68, and 25 °C within 4 h; the defluorination rate reached 58.33%. In addition, the cyclability and stability tests demonstrated that MnFe2O4/mSiO2 is a stable material that can be recycled. Furthermore, XPS characterization and radical scavenging experiments demonstrated that sulfate radicals (SO4·−) and hydroxyl radicals (OH) play a major role in the reaction of MnFe2O4/mSiO2 activated PS. Subsequently, the degradation products were detected by high-performance liquid chromatography tandem triple quadrupole mass spectrometry, indicating that the degradation of PFOA is a gradual process of defluorination and decarbonization in the presence of free radicals. Finally, the metal leaching rate is tested to prove that the material meets environmental requirements while reacting efficiently. In conclusion, this study shows that MnFe2O4/mSiO2 is an easily recoverable and highly efficient and stable material that has great potential for PS activation to treat organic pollutants in water.











Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this article and its supplementary information files.
References
Askeland M, Clarke BO, Cheema SA, Mendez A, Gasco G, Paz-Ferreiro J (2020) Biochar sorption of PFOS, PFOA, PFHxS and PFHxA in two soils with contrasting texture. Chemosphere 249:126072. https://doi.org/10.1016/j.chemosphere.2020.126072
Berger D, Georgescu D, Bajenaru L, Zanfir A, Stănică N, Matei C (2017) Properties of mesostructured silica coated CoFe2O4 versus Fe3O4-silica composites. J Alloy Compd 708:278–284. https://doi.org/10.1016/j.jallcom.2017.02.268
Cao J, Lai L, Lai B, Yao G, Chen X, Song L (2019) Degradation of tetracycline by peroxymonosulfate activated with zero-valent iron: performance, intermediates, toxicity and mechanism. Chem Eng J 364:45–56. https://doi.org/10.1016/j.cej.2019.01.113
Cerro-Gálvez E, Roscales JL, Jiménez B, Sala MM, Dachs J, Vila-Costa M (2020): Microbial responses to perfluoroalkyl substances and perfluorooctanesulfonate (PFOS) desulfurization in the Antarctic marine environment. Water Research 171 https://doi.org/10.1016/j.watres.2019.115434
Cheng M, Zeng G, Huang D, Lai C, Xu P, Zhang C, Liu Y (2016) Hydroxyl radicals based advanced oxidation processes (AOPs) for remediation of soils contaminated with organic compounds: a review. Chem Eng J 284:582–598. https://doi.org/10.1016/j.cej.2015.09.001
da Silva-Rackov CKO, Lawal WA, Nfodzo PA, Vianna MMGR, do Nascimento CAO, Choi H, (2016) Degradation of PFOA by hydrogen peroxide and persulfate activated by iron-modified diatomite. Appl Catal B 192:253–259. https://doi.org/10.1016/j.apcatb.2016.03.067
Deng J, Feng S, Ma X, Tan C, Wang H, Zhou S, Zhang T, Li J (2016) Heterogeneous degradation of Orange II with peroxymonosulfate activated by ordered mesoporous MnFe2O4. Sep Purif Technol 167:181–189. https://doi.org/10.1016/j.seppur.2016.04.035
Du J, Bao J, Liu Y, Kim SH, Dionysiou DD (2019): Facile preparation of porous Mn/Fe3O4 cubes as peroxymonosulfate activating catalyst for effective bisphenol A degradation. Chemical Engineering Journal 376 https://doi.org/10.1016/j.cej.2018.05.177
Fan J, Qin H, Jiang S (2019) Mn-doped g-C3N4 composite to activate peroxymonosulfate for acetaminophen degradation: the role of superoxide anion and singlet oxygen. Chem Eng J 359:723–732. https://doi.org/10.1016/j.cej.2018.11.165
Fu HC, Ma SL, Zhao P, Xu SJ, Zhan SH (2019) Activation of peroxymonosulfate by graphitized hierarchical porous biochar and MnFe2O4 magnetic nanoarchitecture for organic pollutants degradation: structure dependence and mechanism. Chem Eng J 360:157–170. https://doi.org/10.1016/j.cej.2018.11.207
Ghauch A, Baalbaki A, Amasha M, El Asmar R, Tantawi O (2017) Contribution of persulfate in UV-254 nm activated systems for complete degradation of chloramphenicol antibiotic in water. Chem Eng J 317:1012–1025. https://doi.org/10.1016/j.cej.2017.02.133
Gomez-Ruiz B, Gómez-Lavín S, Diban N, Boiteux V, Colin A, Dauchy X, Urtiaga A (2017) Efficient electrochemical degradation of poly- and perfluoroalkyl substances (PFASs) from the effluents of an industrial wastewater treatment plant. Chem Eng J 322:196–204. https://doi.org/10.1016/j.cej.2017.04.040
Gomez-Ruiz B, Ribao P, Diban N, Rivero MJ, Ortiz I, Urtiaga A (2018) Photocatalytic degradation and mineralization of perfluorooctanoic acid (PFOA) using a composite TiO2-rGO catalyst. J Hazard Mater 344:950–957. https://doi.org/10.1016/j.jhazmat.2017.11.048
Hao P, Hu M, Xing R, Zhou W (2020): Synergistic degradation of methylparaben on CuFe2O4-rGO composite by persulfate activation. Journal of Alloys and Compounds 82 310.1016/j.jallcom.2020.153757
Huang KC, Zhao Z, Hoag GE, Dahmani A, Block PA (2005) Degradation of volatile organic compounds with thermally activated persulfate oxidation. Chemosphere 61:551–560. https://doi.org/10.1016/j.chemosphere.2005.02.032
Huang S, Jaffe PR (2019) Defluorination of perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) by Acidimicrobium sp. strain A6. Environ Sci Technol 53:11410–11419. https://doi.org/10.1021/acs.est.9b04047
James Wood R, Sidnell T, Ross I, McDonough J, Lee J, Bussemaker MJ (2020) Ultrasonic degradation of perfluorooctane sulfonic acid (PFOS) correlated with sonochemical and sonoluminescence characterisation. Ultrason Sonochem 68:105196. https://doi.org/10.1016/j.ultsonch.2020.105196
Kharisov BI, Dias HVR, Kharissova OV (2019) Mini-review: ferrite nanoparticles in the catalysis. Arab J Chem 12:1234–1246. https://doi.org/10.1016/j.arabjc.2014.10.049
Kurtan U, Amir M, Yıldız A, Baykal A (2016) Synthesis of magnetically recyclable MnFe 2 O 4 @SiO 2 @Ag nanocatalyst: Its high catalytic performances for azo dyes and nitro compounds reduction. Appl Surf Sci 376:16–25. https://doi.org/10.1016/j.apsusc.2016.02.120
Li J, Xu M, Yao G, Lai B (2018) Enhancement of the degradation of atrazine through CoFe2O4 activated peroxymonosulfate (PMS) process: kinetic, degradation intermediates, and toxicity evaluation. Chem Eng J 348:1012–1024. https://doi.org/10.1016/j.cej.2018.05.032
Li N, Fu F, Lu J, Ding Z, Tang B, Pang J (2017a) Facile preparation of magnetic mesoporous MnFe2O4@SiO2-CTAB composites for Cr(VI) adsorption and reduction. Environ Pollut 220:1376–1385. https://doi.org/10.1016/j.envpol.2016.10.097
Li Q, Chen Z, Wang H, Yang H, Wen T, Wang S, Hu B, Wang X (2021) Removal of organic compounds by nanoscale zero-valent iron and its composites. Sci Total Environ 792:148546. https://doi.org/10.1016/j.scitotenv.2021.148546
Li S, Zhang G, Zhang W, Zheng H, Zhu W, Sun N, Zheng Y, Wang P (2017b) Microwave enhanced Fenton-like process for degradation of perfluorooctanoic acid (PFOA) using Pb-BiFeO3/rGO as heterogeneous catalyst. Chem Eng J 326:756–764. https://doi.org/10.1016/j.cej.2017.06.037
Li Y, Baghi R, Filip J, Islam S, Hope-weeks L, Yan W (2019) Activation of peroxydisulfate by ferrite materials for phenol degradation. ACS Sustainable Chemistry & Engineering 7:8099–8108. https://doi.org/10.1021/acssuschemeng.8b05257
Liang C, Bruell CJ, Marley MC, Sperry KL (2004) Persulfate oxidation for in situ remediation of TCE. I. Activated by ferrous ion with and without a persulfate-thiosulfate redox couple. Chemosphere 55:1213–1223. https://doi.org/10.1016/j.chemosphere.2004.01.029
Lindstrom AB, Strynar MJ, Libelo EL (2011) Polyfluorinated compounds: past, present, and future. Environ Sci Technol 45:7954–7961. https://doi.org/10.1021/es2011622
Liu H, Bruton TA, Li W, Buren JV, Prasse C, Doyle FM, Sedlak DL (2016) Oxidation of benzene by persulfate in the presence of Fe(III)- and Mn(IV)-containing oxides: stoichiometric efficiency and transformation products. Environ Sci Technol 50:890–898. https://doi.org/10.1021/acs.est.5b04815
Liu H, Yao J, Wang L, Wang X, Qu R, Wang Z (2019) Effective degradation of fenitrothion by zero-valent iron powder (Fe0) activated persulfate in aqueous solution: kinetic study and product identification. Chem Eng J 358:1479–1488. https://doi.org/10.1016/j.cej.2018.10.153
Liu Z, Chen G, Hu F, Li X (2020) Synthesis of mesoporous magnetic MnFe2O4@CS-SiO2 microsphere and its adsorption performance of Zn(2+) and MB studies. J Environ Manage 263:110377. https://doi.org/10.1016/j.jenvman.2020.110377
Ma Q, Zhang H, Zhang X, Li B, Guo R, Cheng Q, Cheng X (2019) Synthesis of magnetic CuO/MnFe2O4 nanocomposite and its high activity for degradation of levofloxacin by activation of persulfate. Chem Eng J 360:848–860. https://doi.org/10.1016/j.cej.2018.12.036
Miklos DB, Remy C, Jekel M, Linden KG, Drewes JE, Hubner U (2018) Evaluation of advanced oxidation processes for water and wastewater treatment - a critical review. Water Res 139:118–131. https://doi.org/10.1016/j.watres.2018.03.042
Ren Y, Zhang H, An H, Zhao Y, Feng J, Xue L, Luan T, Fan Z (2018) Catalytic ozonation of di-n-butyl phthalate degradation using manganese ferrite/reduced graphene oxide nanofiber as catalyst in the water. J Colloid Interface Sci 526:347–355. https://doi.org/10.1016/j.jcis.2018.04.073
Ribeiro AR, Nunes OC, Pereira MF, Silva AM (2015) An overview on the advanced oxidation processes applied for the treatment of water pollutants defined in the recently launched Directive 2013/39/EU. Environ Int 75:33–51. https://doi.org/10.1016/j.envint.2014.10.027
Sahoo B, Sahu SK, Nayak S, Dhara D, Pramanik P (2012): Fabrication of magnetic mesoporous manganese ferrite nanocomposites as efficient catalyst for degradation of dye pollutants. Catalysis Science & Technology 2 https://doi.org/10.1039/c2cy20026k
Sahoo B, Devi KS, Dutta S, Maiti TK, Pramanik P, Dhara D (2014) Biocompatible mesoporous silica-coated superparamagnetic manganese ferrite nanoparticles for targeted drug delivery and MR imaging applications. J Colloid Interface Sci 431:31–41. https://doi.org/10.1016/j.jcis.2014.06.003
Singh G, Chandra S (2018) Electrochemical performance of MnFe 2 O 4 nano-ferrites synthesized using thermal decomposition method. Int J Hydrogen Energy 43:4058–4066. https://doi.org/10.1016/j.ijhydene.2017.08.181
Slowing II, Vivero-Escoto JL, Wu CW, Lin VS (2008) Mesoporous silica nanoparticles as controlled release drug delivery and gene transfection carriers. Adv Drug Deliv Rev 60:1278–1288. https://doi.org/10.1016/j.addr.2008.03.012
Tan C, Gao N, Fu D, Deng J, Deng L (2017) Efficient degradation of paracetamol with nanoscaled magnetic CoFe 2 O 4 and MnFe 2 O 4 as a heterogeneous catalyst of peroxymonosulfate. Sep Purif Technol 175:47–57. https://doi.org/10.1016/j.seppur.2016.11.016
Trojanowicz M, Bojanowska-Czajka A, Bartosiewicz I, Kulisa K (2018) Advanced oxidation/reduction processes treatment for aqueous perfluorooctanoate (PFOA) and perfluorooctanesulfonate (PFOS) – a review of recent advances. Chem Eng J 336:170–199. https://doi.org/10.1016/j.cej.2017.10.153
Wacławek S, Lutze HV, Grübel K, Padil VVT, Černík M, Dionysiou DD (2017) Chemistry of persulfates in water and wastewater treatment: a review. Chem Eng J 330:44–62. https://doi.org/10.1016/j.cej.2017.07.132
Wang J, Liu L, Chou S, Liu H, Wang J (2017a) A 3D porous nitrogen-doped carbon-nanofiber-supported palladium composite as an efficient catalytic cathode for lithium–oxygen batteries. Journal of Materials Chemistry A 5:1462–1471. https://doi.org/10.1039/c6ta07050g
Wang J, Wang S (2018) Activation of persulfate (PS) and peroxymonosulfate (PMS) and application for the degradation of emerging contaminants. Chem Eng J 334:1502–1517. https://doi.org/10.1016/j.cej.2017.11.059
Wang L, Kong D, Ji Y, Lu J, Yin X, Zhou Q (2018) Formation of halogenated disinfection byproducts during the degradation of chlorophenols by peroxymonosulfate oxidation in the presence of bromide. Chem Eng J 343:235–243. https://doi.org/10.1016/j.cej.2018.03.006
Wang S, Yang Q, Chen F, Sun J, Luo K, Yao F, Wang X, Wang D, Li X, Zeng G (2017b) Photocatalytic degradation of perfluorooctanoic acid and perfluorooctane sulfonate in water: a critical review. Chem Eng J 328:927–942. https://doi.org/10.1016/j.cej.2017.07.076
Wang S, Liu Y, Wang J (2020): Iron and sulfur co-doped graphite carbon nitride (FeOy/S-g-C3N4) for activating peroxymonosulfate to enhance sulfamethoxazole degradation. Chemical Engineering Journal 382 https://doi.org/10.1016/j.cej.2019.122836
Wang Y, Zhao H, Li M, Fan J, Zhao G (2014) Magnetic ordered mesoporous copper ferrite as a heterogeneous Fenton catalyst for the degradation of imidacloprid. Appl Catal B 147:534–545. https://doi.org/10.1016/j.apcatb.2013.09.017
Wu D, Li X, Zhang J, Chen W, Lu P, Tang Y, Li L (2018) Efficient PFOA degradation by persulfate-assisted photocatalytic ozonation. Sep Purif Technol 207:255–261. https://doi.org/10.1016/j.seppur.2018.06.059
Xu M, Li J, Yan Y, Zhao X, Yan J, Zhang Y, Lai B, Chen X, Song L (2019) Catalytic degradation of sulfamethoxazole through peroxymonosulfate activated with expanded graphite loaded CoFe2O4 particles. Chem Eng J 369:403–413. https://doi.org/10.1016/j.cej.2019.03.075
Yamashita N, Kannan K, Taniyasu S, Horii Y, Petrick G, Gamo T (2005) A global survey of perfluorinated acids in oceans. Mar Pollut Bull 51:658–668. https://doi.org/10.1016/j.marpolbul.2005.04.026
Yang L, He L, Xue J, Ma Y, Xie Z, Wu L, Huang M, Zhang Z (2020) Persulfate-based degradation of perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) in aqueous solution: review on influences, mechanisms and prospective. J Hazard Mater 393:122405. https://doi.org/10.1016/j.jhazmat.2020.122405
Yang S, Wang P, Yang X, Shan L, Zhang W, Shao X, Niu R (2010) Degradation efficiencies of azo dye acid orange 7 by the interaction of heat, UV and anions with common oxidants: persulfate, peroxymonosulfate and hydrogen peroxide. J Hazard Mater 179:552–558. https://doi.org/10.1016/j.jhazmat.2010.03.039
Yao Y, Cai Y, Lu F, Wei F, Wang X, Wang S (2014) Magnetic recoverable MnFe(2)O(4) and MnFe(2)O(4)-graphene hybrid as heterogeneous catalysts of peroxymonosulfate activation for efficient degradation of aqueous organic pollutants. J Hazard Mater 270:61–70. https://doi.org/10.1016/j.jhazmat.2014.01.027
Yin P, Hu Z, Song X, Liu J, Lin N (2016): Activated persulfate oxidation of perfluorooctanoic acid (PFOA) in groundwater under acidic conditions. Int J Environ Res Public Health 13 https://doi.org/10.3390/ijerph13060602
Yu S, Pang H, Huang S, Tang H, Wang S, Qiu M, Chen Z, Yang H, Song G, Fu D, Hu B, Wang X (2021) Recent advances in metal-organic framework membranes for water treatment: a review. Sci Total Environ 800:149662. https://doi.org/10.1016/j.scitotenv.2021.149662
Zhang R, Sun P, Boyer TH, Zhao L, Huang CH (2015) Degradation of pharmaceuticals and metabolite in synthetic human urine by UV, UV/H2O2, and UV/PDS. Environ Sci Technol 49:3056–3066. https://doi.org/10.1021/es504799n
Zhang S, Xu W, Zeng M, Li J, Li J, Xu J, Wang X (2013): Superior adsorption capacity of hierarchical iron oxide@magnesium silicate magnetic nanorods for fast removal of organic pollutants from aqueous solution. Journal of Materials Chemistry A 1 https://doi.org/10.1039/c3ta12767b
Zhang Y, Moores A, Liu J, Ghoshal S (2019) New insights into the degradation mechanism of perfluorooctanoic acid by persulfate from density functional theory and experimental data. Environ Sci Technol 53:8672–8681. https://doi.org/10.1021/acs.est.9b00797
Zhang Y, Zhang BT, Teng Y, Zhao J, Sun X (2021) Heterogeneous activation of persulfate by carbon nanofiber supported Fe3O4@carbon composites for efficient ibuprofen degradation. J Hazard Mater 401:123428. https://doi.org/10.1016/j.jhazmat.2020.123428
Zhou X, Luo C, Luo M, Wang Q, Wang J, Liao Z, Chen Z, Chen Z (2020): Understanding the synergetic effect from foreign metals in bimetallic oxides for PMS activation: a common strategy to increase the stoichiometric efficiency of oxidants. Chemical Engineering Journal 381 https://doi.org/10.1016/j.cej.2019.122587
Zhou Z, Liu X, Sun K, Lin C, Ma J, He M, Ouyang W (2019) Persulfate-based advanced oxidation processes (AOPs) for organic-contaminated soil remediation: a review. Chem Eng J 372:836–851. https://doi.org/10.1016/j.cej.2019.04.213
Funding
This work was financially supported by the National Key Research and Development Project (No. 2020YFC1808203), National Natural Science Foundation of China (Nos. 11975147, 2075148, and 41773121), and Science and Technology Commission of Shanghai Municipality (No. 20010500300).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Pengfei Ji, Feng Zhu, Jiamin Zhou, and Jintao Ma. The first draft of the manuscript was written by Pengfei Ji, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Ricardo A. Torres-Palma
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ji, P., Zhu, F., Zhou, J. et al. Synthesis of superparamagnetic MnFe2O4/mSiO2 nanomaterial for degradation of perfluorooctanoic acid by activated persulfate. Environ Sci Pollut Res 29, 37071–37083 (2022). https://doi.org/10.1007/s11356-021-17782-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-17782-y