Abstract
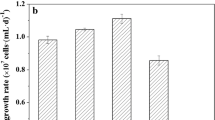
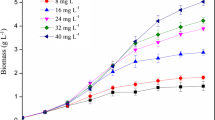
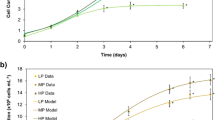
Intracellular phosphorus (P) accumulation can improve microalgal growth and lipid synthesis. However, large excess of P causes cell poisoning. This study utilized a P-fed-batch strategy to investigate its potential to improve the utilization of the excessive P, while avoiding toxic side effects. This strategy contributed to a more complete utilization of the intracellularly stored P, which enhanced the microalgae biomass by 10−15% by upregulating the brassinosteroid growth hormone gene at a P-fed-batch frequency of 2−8. Furthermore, the lipid content increased by 4−16% via upregulation of lipid synthesis-related genes. As a result, the P-fed-batch strategy significantly increased the lipid production by 13−19%. The content of saturated fatty acid increased by ~ 100%, implying improved combustibility and oxidative stability. This is the first study of this P-fed-batch strategy and provides a new concept for the complete utilization of excessive P.





Similar content being viewed by others
References
Basri EM, Maznah WOW (2017) Differential growth and biochemical composition of photoautotrophic and heterotrophic Isochrysis maritima: evaluation for use as aquaculture feed. J Appl Phycol 29:1159–1170. https://doi.org/10.1007/s10811-017-1054-1
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917. https://doi.org/10.1139/o59-099
Brown N, Shilton A (2014) Luxury uptake of phosphorus by microalgae in waste stabilisation ponds: current understanding and future direction. Rev Environ Sci Biotechnol 13:321–328. https://doi.org/10.1007/s11157-014-9337-3
Bumbak F, Cook S, Zachleder V, Hauser S, Kovar K (2011) Best practices in heterotrophic high-cell-density microalgal processes: achievements, potential and possible limitations. Appl Microbiol Biotechnol 91:31–46. https://doi.org/10.1007/s00253-011-3311-6
Cheirsilp B, Torpee S (2012) Enhanced growth and lipid production of microalgae under mixotrophic culture condition: effect of light intensity, glucose concentration and fed-batch cultivation. Bioresour Technol 110:510–516. https://doi.org/10.1016/j.biortech.2012.01.125
Chu FF, Chu PN, Cai PJ, Li WW, Lam PKS, Zeng RJ (2013) Phosphorus plays an important role in enhancing biodiesel productivity of Chlorella vulgaris under nitrogen deficiency. Bioresour Technol 134:341–346. https://doi.org/10.1016/j.biortech.2013.01.131
Chu FF, Shen XF, Lam PKS, Zeng RJ (2014) Optimization of CO2 concentration and light intensity for biodiesel production by Chlorella vulgaris FACHB-1072 under nitrogen deficiency with phosphorus luxury uptake. J Appl Phycol 26:1631–1638. https://doi.org/10.1007/s10811-013-0200-7
Cristobal Garcia-Canedo J, Cristiani-Urbina E, Flores-Ortiz CM, Ponce-Noyola T, Esparza-Garcia F, Olivia Canizares-Villanueva R (2016) Batch and fed-batch culture of Scenedesmus incrassatulus: effect over biomass, carotenoid profile and concentration, photosynthetic efficiency and non-photochemical quenching. Algal Res 13:41–52. https://doi.org/10.1016/j.algal.2015.11.013
Falkowski P (2000) Rationalizing elemental ratios in unicellular algae. J Phycol 36:3–6. https://doi.org/10.1046/j.1529-8817.2000.99161.x
Fu L, Cui X, Li Y, Xu L, Zhang C, Xiong R, Zhou D, Crittenden JC (2017) Excessive phosphorus enhances Chlorella regularis lipid production under nitrogen starvation stress during glucose heterotrophic cultivation. Chem Eng J 330:566–572. https://doi.org/10.1016/j.cej.2017.07.182
Goncalves EC, Wilkie AC, Kirst M, Rathinasabapathi B (2016) Metabolic regulation of triacylglycerol accumulation in the green algae: identification of potential targets for engineering to improve oil yield. Plant Biotechnol J 14:1649–1660. https://doi.org/10.1111/pbi.12523
Guarnieri MT, Nag A, Yang S, Pienkos PT (2013) Proteomic analysis of Chlorella vulgaris: potential targets for enhanced lipid accumulation. J Proteome 93:245–253. https://doi.org/10.1016/j.jprot.2013.05.025
Harold FM (1966) Inorganic polyphosphates in biology: structure, metabolism, and function. Bacteriol Rev 30:772–794
Hoekman SK, Broch A, Robbins C, Ceniceros E, Natarajan M (2012) Review of biodiesel composition, properties, and specifications. Renew Sust Energ Rev 16:143–169. https://doi.org/10.1016/j.rser.2011.07.143
Kamalanathan M, Chaisutyakorn P, Gleadow R, Beardall J (2018) A comparison of photoautotrophic, heterotrophic, and mixotrophic growth for biomass production by the green alga Scenedesmus sp (Chlorophyceae). Phycologia 57:309–317. https://doi.org/10.2216/17-82.1
Konishi T, Shinohara K, Yamada K, Sasaki Y (1996) Acetyl-CoA carboxylase in higher plants: most plants other than gramineae have both the prokaryotic and the eukaryotic forms of this enzyme. Plant Cell Physiol 37:117–122. https://doi.org/10.1093/oxfordjournals.pcp.a028920
Leyva LA, Bashan Y, de-Bashan LE (2015) Activity of acetyl-CoA carboxylase is not directly linked to accumulation of lipids when Chlorella vulgaris is co-immobilised with Azospirillum brasilense in alginate under autotrophic and heterotrophic conditions. Ann Microbiol 65:339-349. https://doi.org/10.1007/s13213-014-0866-3
Li Y, Yuan Z, Mu J, Chen D, Feng B (2013) Proteomic analysis of lipid accumulation in Chlorella protothecoides cells by heterotrophic N deprivation coupling cultivation. Energy Fuel 27:4031–4040. https://doi.org/10.1021/ef4000177
Li Y, Han F, Xu H, Mu J, Chen D, Feng B, Zeng H (2014) Potential lipid accumulation and growth characteristic of the green alga Chlorella with combination cultivation mode of nitrogen (N) and phosphorus (P). Bioresour Technol 174:24–32. https://doi.org/10.1016/j.biortech.2014.09.142
Li Q, Fu L, Wang Y, Zhou D, Rittmann BE (2018) Excessive phosphorus caused inhibition and cell damage during heterotrophic growth of Chlorella regularis. Bioresour Technol 268:266–270. https://doi.org/10.1016/j.biortech.2018.07.148
Li-Beisson Y, Beisson F, Riekhof W (2015) Metabolism of acyl-lipids in Chlamydomonas reinhardtii. Plant J 82:504–522. https://doi.org/10.1111/tpj.12787
Meza B, de- Bashan LE, Hernandez J-P, Bashan Y (2015) Accumulation of intra-cellular polyphosphate in Chlorella vulgaris cells is related to indole-3-acetic acid produced by Azospirillum brasilense. Res Microbiol 166:399–407. https://doi.org/10.1016/j.resmic.2015.03.001
Morales-Sanchez D, Tinoco-Valencia R, Kyndt J, Martinez A (2013) Heterotrophic growth of Neochloris oleoabundans using glucose as a carbon source. Biotechnol Biofuel 6:100. https://doi.org/10.1186/1754-6834-6-100
Moreno B, Urbina JA, Oldfield E, Bailey BN, Rodrigues CO, Docampo R (2000) 31P NMR spectroscopy of Trypanosoma brucei, Trypanosoma cruzi, and Leishmania major. Evidence for high levels of condensed inorganic phosphates. J Biol Chem 275:28356–28362. https://doi.org/10.1074/jbc.M003893200
Nishikawa K, Machida H, Yamakoshi Y, Ohtomo R, Saito K, Saito M, Tominaga N (2006) Polyphosphate metabolism in an acidophilic alga Chlamydomonas acidophila KT-1 (Chlorophyta) under phosphate stress. Plant Sci 170:307–313. https://doi.org/10.1016/j.plantsci.2005.08.025
Perez-Garcia O, Escalante FME, de- Bashan LE, Bashan Y (2011) Heterotrophic cultures of microalgae: metabolism and potential products. Water Res 45:11–36. https://doi.org/10.1016/j.watres.2010.08.037
Powell N, Shilton AN, Pratt S, Chisti Y (2008) Factors influencing luxury uptake of phosphorus by microalgae in waste stabilization ponds. Environ Sci Technol 42:5958–5962. https://doi.org/10.1021/es703118s
Sansawa H, Endo H (2004) Production of intracellular phytochemicals in Chlorella under heterotrophic conditions. J Biosci Bioeng 98:437–444. https://doi.org/10.1263/jbb.98.437
Schmidt RA, Wiebe MG, Eriksen NT (2005) Heterotrophic high cell-density fed-batch cultures of the phycocyanin-producing red alga Galdieria sulphuraria. Biotechnol Bioeng 90:77–84. https://doi.org/10.1002/bit.20417
Shen XF, Chu FF, Lam PKS, Zeng RJ (2015) Biosynthesis of high yield fatty acids from Chlorella vulgaris NIES-227 under nitrogen starvation stress during heterotrophic cultivation. Water Res 81:294–300. https://doi.org/10.1016/j.watres.2015.06.003
Sureshkumar P, Thomas J (2019) Strategic growth of limnic green microalgae with phycoremediation potential for enhanced production of biomass and biomolecules for sustainable environment. Environ Sci Pollut Res 26:34702–34712. https://doi.org/10.1007/s11356-018-4012-9
Tan Y, Lin J (2011) Biomass production and fatty acid profile of a Scenedesmus rubescens-like microalga. Bioresour Technol 102:10131–10135. https://doi.org/10.1016/j.biortech.2011.07.091
Wang Y, Yu J, Wang P, Deng S, Chang J, Ran Z (2018) Response of energy microalgae Chlamydomonas reinhardtii to nitrogen and phosphorus stress. Environ Sci Pollut Res 25:5762–5770. https://doi.org/10.1007/s11356-017-0931-0
Xiong W, Li X, Xiang J, Wu Q (2008) High-density fermentation of microalga Chlorella protothecoides in bioreactor for microbio-diesel production. Appl Microbiol Biotechnol 78:29–36. https://doi.org/10.1007/s00253-007-1285-1
Yu X, Zhao P, He C, Li J, Tang X, Zhou J, Huang Z (2012) Isolation of a novel strain of Monoraphidium sp and characterization of its potential application as biodiesel feedstock. Bioresour Technol 121:256–262. https://doi.org/10.1016/j.biortech.2012.07.002
Zhang XQ, Powles SB (2006) The molecular bases for resistance to acetyl co-enzyme A carboxylase (ACCase) inhibiting herbicides in two target-based resistant biotypes of annual ryegrass (Lolium rigidum). Planta 223:550–557. https://doi.org/10.1007/s00425-005-0095-x
Zhang Y, Adams IP, Ratledge C (2007) Malic enzyme: the controlling activity for lipid production? Overexpression of malic enzyme in Mucor circinelloides leads to a 2.5-fold increase in lipid accumulation. Microbiology-Sgm 153:2013–2025. https://doi.org/10.1099/mic.0.2006/002683-0
Zhou D, Zhang C, Fu L, Xu L, Cui X, Li Q, Crittenden JC (2017) Responses of the microalga Chlorophyta sp to bacterial quorum sensing molecules (N-acylhomoserine lactones): aromatic protein induced self-aggregation. Environ Sci Technol 51:3490–3498. https://doi.org/10.1021/acs.est.7b00355
Zhu S, Huang W, Xu J, Wang Z, Xu J, Yuan Z (2014) Metabolic changes of starch and lipid triggered by nitrogen starvation in the microalga Chlorella zofingiensis. Bioresour Technol 152:292–298. https://doi.org/10.1016/j.biortech.2013.10.092
Funding
This research was financially supported by the National Natural Science Foundation of China (51708095, 51578117, 51722803), Fundamental Research Funds for the Central Universities (2412017QD027, 2412018ZD013, 2412018ZD042), the Science and Technology Project of Jilin Province (20180520168JH), and China Postdoctoral Science Foundation (2017M611302, 2018T110241).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Ta Yeong Wu
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 7714 kb)
Rights and permissions
About this article
Cite this article
Fu, L., Yan, G., Li, Y. et al. Phosphorus supply via a fed-batch strategy improves lipid heterotrophic production of Chlorella regularis. Environ Sci Pollut Res 27, 31677–31685 (2020). https://doi.org/10.1007/s11356-020-09495-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-09495-5