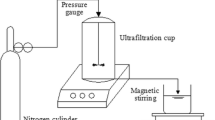
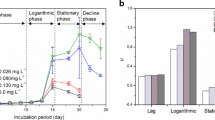
Abstract
This paper focuses on the characterization of extracellular polymeric substances (EPS), which are composed of soluble EPS (SL-EPS), loosely bound EPS (LB-EPS), and tightly bound EPS (TB-EPS) produced by Microcystis aeruginosa under the stress of linoleic acid (LA) and LA sustained-release microspheres. Three-dimensional excitation-emission matrix (3D-EEM) fluorescence spectroscopy and Fourier transform infrared (FTIR) spectrometry were used to characterize three forms of EPS while the content of polysaccharide and protein was tested, respectively. The results showed that the highest inhibitor rate (IR) occurred when M. aeruginosa were exposed to LA sustained-release microspheres of 0.3 g L−1. The 3D-EEM contour demonstrated that tryptophan and protein-like substances were detected in all three EPS fractions, whereas humic acid-like substance was only distributed in SL-EPS, and aromatic proteins merely existed in LB-EPS and TB-EPS. The infrared spectrum showed that functional groups in three EPS fractions had no obvious change in all experimental groups. Polysaccharide (1120–1270 cm−1, C–O–C and C–O stretching vibration) and protein (1384–1670 cm−1, C–N and N–H stretching) were detected in three forms of EPS.

ᅟ








Similar content being viewed by others
References
Ahmad S, Reynolds D (1999) Monitoring of water quality using fluorescence technique: prospect of on-line process control. Water Res 33:2069–2074
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cai SW, Singh BR (1999) Identification of β-turn and random coil amide III infrared bands for secondary structure estimation of proteins. Biophys Chem 80:7–20
Chabaliná LD, Pastor MR, Rico DP (2013) Characterization of soluble and bound EPS obtained from 2 submerged membrane bioreactors by 3D-EEM and HPSEC. Talanta 115:706–712
Chang XW, Eigemann F, Hilt S (2012) Do macrophytes support harmful cyanobacteria? Interactions with a green alga reverse the inhibiting effects of macrophyte allelochemicals on Microcystis aeruginosa. Harmful Algae 19:76–84
Chen W, Westerhoff P, Leenheer JA, Booksh K (2003) Fluorescence excitation-emission matrix regional integration to quantify spectra for dissolved organic matter. Environ Sci Technol 37:5701–5710
Chen YP, Zhang P, Guo JS, Fang F, Gao X, Li C (2013) Functional groups characteristics of EPS in biofilm growing on different carriers. Chemosphere 92:633–638
Chiou YT, Hsieh ML, Yeh HH (2010) Effect of algal extracellular polymer substances on UF membrane fouling. Desalination 250:648–652
Cogan N, Keener JP (2004) The role of the biofilm matrix in structural development. Math Med Biol 21:147–166
De Philippis R, Vincenzini M (1998) Exocellular polysaccharides from cyanobacteria and their possible applications. FEMS Microbiol Rev 22:151–175
Dixon MB, Richard Y, Ho L, Chow CW, O’Neill BK, Newcombe G (2011) A coagulation–powdered activated carbon–ultrafiltration–multiple barrier approach for removing toxins from two Australian cyanobacterial blooms. J Hazard Mater 186:1553–1559
Dubois M, Gilles KA, Hamilton JK, Rebers P, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Fang JY, Yang X, Ma J, Shang C, Zhao Q (2010) Characterization of algal organic matter and formation of DBPs from chlor(am)ination. Water Res 44:5897–5906
Gallardo-Williams MT, Geiger CL, Pidala JA, Martin DF (2002) Essential fatty acids and phenolic acids from extracts and leachates of southern cattail (Typha domingensis P.) Phytochemistry 59:305–308
Gross EM (2003) Allelopathy of aquatic autotrophs. Crit Rev Plant Sci 22:313–339
Guibaud G, Comte S, Bordas F, Dupuy S, Baudu M (2005) Comparison of the complexation potential of extracellular polymeric substances (EPS), extracted from activated sludges and produced by pure bacteria strains, for cadmium, lead and nickel. Chemosphere 59:629–638
Hong Y, Hu HY (2009) Research and application of aquatic plant allelopathy inhibition on algae. Chin Sci Bull 54:287–293
Hung MT, Liu JC (2006) Microfiltration for separation of green algae from water. Colloids Surf B Biointerfaces 51:157–164
Ismaili MM, Belin C, Lamotte M, Texier H (1998) Distribution et caractérisation par fluorescence de la matière organique dissoute dans les eaux de la Manche centrale. Oceanol Acta 21:645–654
Joshi PM, Juwarkar AA (2009) In vivo studies to elucidate the role of extracellular polymeric substances from Azotobacter in immobilization of heavy metals. Environ Sci Technol 43:5884–5889
Laspidou CS, Rittmann BE (2002) A unified theory for extracellular polymeric substances, soluble microbial products, and active and inert biomass. Water Res 36:2711–2720
Li L, Gao NY, Deng Y, Yao JJ, Zhang KJ (2012) Characterization of intracellular & extracellular algae organic matters (AOM) of Microcystic aeruginosa and formation of AOM-associated disinfection byproducts and odor & taste compounds. Water Res 46:1233–1240
Liu A, Ahn IS, Mansfield C, Lion LW, Shuler ML, Ghiorse WC (2001) Phenanthrene desorption from soil in the presence of bacterial extracellular polymer: observations and model predictions of dynamic behavior. Water Res 35:835–843
Mounier S, Patel N, Quilici L, Benaim J, Benamou C (1999) Fluorescence 3D de la matière organique dissoute du fleuve amazone:(Three-dimensional fluorescence of the dissolved organic carbon in the Amazon river). Water Res 33:1523–1533
Neu T, Lawrence J (2009) Extracellular polymeric substances in microbial biofilms. In: Microbial glycobiology: structures, relevance and applications. Elsevier, San Diego, pp 735–758
Ni LX, Jie XT, Wang PF, Li SY, Hu SZ, Li YP, Li Y, Acharya K (2015a) Characterization of unsaturated fatty acid sustained-release microspheres for long-term algal inhibition. Chemosphere 120:383–390
Ni LX, Jie XT, Wang PF, Li SY, Wang GX, Li YP, Li Y, Acharya K (2015b) Effect of linoleic acid sustained-release microspheres on Microcystis aeruginosa antioxidant enzymes activity and microcystins production and release. Chemosphere 121:110–116
Paerl HW, Paul VJ (2012) Climate change: links to global expansion of harmful cyanobacteria. Water Res 46:1349–1363
Parlanti E, Wörz K, Geoffroy L, Lamotte M (2000) Dissolved organic matter fluorescence spectroscopy as a tool to estimate biological activity in a coastal zone submitted to anthropogenic inputs. Org Geochem 31:1765–1781
Pereira S, Zille A, Micheletti E, Moradas-Ferreira P, De Philippis R, Tamagnini P (2009) Complexity of cyanobacterial exopolysaccharides: composition, structures, inducing factors and putative genes involved in their biosynthesis and assembly. FEMS Microbiol Rev 33:917–941
Qu FS, Liang H, Wang ZZ, Wang H, Yu HR, Li GB (2012) Ultrafiltration membrane fouling by extracellular organic matters (EOM) of Microcystis aeruginosa in stationary phase: influences of interfacial characteristics of foulants and fouling mechanisms. Water Res 46:1490–1500
Reynolds D, Ahmad S (1997) Rapid and direct determination of wastewater BOD values using a fluorescence technique. Water Res 31:2012–2018
Sheng GP, Yu HQ, Li XY (2010) Extracellular polymeric substances (EPS) of microbial aggregates in biological wastewater treatment systems: a review. Biotechnol Adv 28:882–894
Stadnik MJ, Freitas MB (2014) Algal polysaccharides as source of plant resistance inducers. Trop Plant Pathol 39:111–118
Sun WH, Yu ZW, Yu SW (1989) The harness of an eutrophic water body by water-hyacinth. Acta scientiae circumstantiae/Huanjing Kexue Xuebao Beijing 9:188–195
Tønnesen HH, Karlsen J (2002) Alginate in drug delivery systems. Drug Dev Ind Pharm 28:621–630
Wingender J, Neu TR, Flemming H-C (2012) Microbial extracellular polymeric substances: characterization, structure and function. Springer Science & Business Media
Xiao R, Zheng Y (2016) Overview of microalgal extracellular polymeric substances (EPS) and their applications. Biotechnol Adv 34:1225–1244
Xu YF, Yang J, Ou MM, Wang YL, Jia JP (2007) Study of Microcystis aeruginosa inhibition by electrochemical method. Biochem Eng J 36:215–220
Xu HC, He PJ, Wang GZ, Yu GH, Shao LM (2010) Enhanced storage stability of aerobic granules seeded with pellets. Bioresour Technol 101:8031–8037
Xu HC, Cai HY, Yu GH, Jiang HL (2013) Insights into extracellular polymeric substances of cyanobacterium Microcystis aeruginosa using fractionation procedure and parallel factor analysis. Water Res 47:2005–2014
You GX, Hou J, Xu Y, Wang C, Wang PF, Miao LZ, Ao YH, Li Y, Lv BW (2015) Effects of CeO2 nanoparticles on production and physicochemical characteristics of extracellular polymeric substances in biofilms in sequencing batch biofilm reactor. Bioresour Technol 194:91–98
Zhang TT, Chen CP, He M, Wu AP, Nie LW (2007) Allelopathic effects of several higher aquatic plants on algae. J Biol 24:32–36
Zhu L, Qi HY, Kong Y, Yu YW, Xu XY (2012) Component analysis of extracellular polymeric substances (EPS) during aerobic sludge granulation using FTIR and 3D-EEM technologies. Bioresour Technol 124:455–459
Acknowledgements
This work was supported jointly by the Key Program of the National Natural Science Foundation of China (No. 91647206), the National Science Fund for Creative Research Groups of China (No. 51421006), the Program for Changjiang Scholars and Innovative Research Team at Hohai University (No. IRT13061), the National Natural Science Foundation (Grant No. 51579073, 41373111), the Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), and the Top-notch Academic Programs Project of Jiangsu Higher Education Institutions (TAPP).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible editor: Vitor Manuel Oliveira Vasconcelos
Rights and permissions
About this article
Cite this article
Ni, L., Li, D., Rong, S. et al. Characterization of extracellular polymeric substance (EPS) fractions produced by Microcystis aeruginosa under the stress of linoleic acid sustained-release microspheres. Environ Sci Pollut Res 24, 21091–21102 (2017). https://doi.org/10.1007/s11356-017-9540-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-9540-1