Abstract
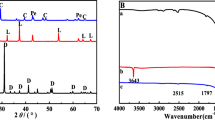
In this study, a hydrotalcite (HT MgAl) was synthesized by coprecipitation at a constant pH. Its mixed oxide (HTC MgAl) was obtained for the removal of triclosan (TCS). The two materials were characterized by Scanning Electron Microscopy (SEM), X-ray Dispersive Spectroscopy (EDS), X-Ray Diffraction (XRD), and Fourier Transform Infrared Spectroscopy (FTIR). Measurements of N2 physisorption and Point of Zero Charge (pHPZC) were also analyzed. TCS removal was investigated in terms of pH, adsorption kinetics, adsorption isotherms, and temperature. The HT MgAl material presented the typical morphology and crystalline structure of a hydrotalcite composed of C, O, Al, and Mg. This material is classified as a mesoporous material with a specific surface area of 114.92 m2/g and a pHPZC of 8.82. On the other hand, HTC MgAl material is a MgAl oxide with a surface area of 140 m2/g and a pHPZC of 11.06. Adsorption results showed that the highest adsorption capacity was obtained at pH 9.0 and at an equilibrium time of 24 h in both materials. The experimental kinetic and isotherm data were well-adjusted to the pseudo-first order and Freundlich models, respectively. This indicates a physisorption mechanism on heterogeneous materials. Therefore, the adsorption of TCS into the HT MgAl material was by way of ion exchange. On the other hand, the adsorption of TCS into the HTC MgAl material was by way of ion exchange and TCS intercalation by the reconstruction of the material. The TCS adsorption process was exothermic, and not spontaneous; hence, randomness was reduced.






Similar content being viewed by others
Availability of Data and Materials
All data generated or analyzed during this study are included in this published and supplementary information files.
Code Availability
Not applicable.
References
A Alshishani M Saaid C Basheer B Saad 2019 High performance liquid chromatographic determination of triclosan, triclocarban and methyl-triclosan in wastewater using mini-bar micro-solid phase extraction Microchemical Journal 147 339 348 https://doi.org/10.1016/j.microc.2019.03.044
Alvarez-García, S., Ramírez-García, J. J., Granados-Correa, F., & Sánchez-Meza, J. C. (2018). Determination of kinetic, isotherm, and thermodynamic parameters of the methamidophos adsorption onto cationic surfactant-modified zeolitic materials. Water, air, and soil pollution, 229(11). doi: https://doi.org/10.1007/s11270-018-3995-7
Alvarez-García, Sonia, Macedo-Miranda, G., Martínez-Gallegos, S., Ordoñez-Regíl, E., López-Castillo, J., & Aguirre-Miranda, E. Removal of triclosan by CTAB-modified zeolite-rich tuff from aqueous solutions. MRS advances, 1–8. (2020). doi: https://doi.org/10.1557/adv.2020.394
O Aschenbrenner P McGuire S Alsamaq J Wang S Supasitmongkol B Al-Duri 2011 Adsorption of carbon dioxide on hydrotalcite-like compounds of different compositions Chemical Engineering Research and Design 89 9 1711 1721 https://doi.org/10.1016/j.cherd.2010.09.019
S Bellú L Sala J González S García M Frascaroli 2010 Thermodynamic and dynamic of chromium biosorption by pectic and lignocellulocic biowastes Journal of Water Resource and Protection 02 10 888 897 https://doi.org/10.4236/jwarp.2010.210106
A Calderón M Meraz A Tomasini 2019 Pharmaceuticals present in urban and hospital wastewaters in mexico city Journal of Water Chemistry and Technology 41 2 105 112 https://doi.org/10.3103/s1063455x19020073
Dávila-Estrada, M., Ramírez-García, J. J., Solache-Ríos, M. J., & Gallegos-Pérez, J. L. (2018). Kinetic and equilibrium sorption studies of ceftriaxone and paracetamol by surfactant-modified zeolite. Water, air, and soil pollution, 229(4). https://doi.org/10.1007/s11270-018-3783-4
Dhillon, G. S., Kaur, S., Pulicharla, R., Brar, S. K., Cledón, M., Verma, M., & Surampalli, R. Y. (2015, May 22). Triclosan: Current status, occurrence, environmental risks, and bioaccumulation potential. International journal of environmental research and public health. MDPI AG. https://doi.org/10.3390/ijerph120505657
J Ding M Su C Wu K Lin 2015 Transformation of triclosan to 2,8-dichlorodibenzo-p-dioxin by iron and manganese oxides under near dry conditions Chemosphere 133 41 46 https://doi.org/10.1016/j.chemosphere.2015.03.055
R Elmoubarki FZ Mahjoubi A Elhalil H Tounsadi M Abdennouri M Sadiq 2017 Ni/Fe and Mg/Fe layered double hydroxides and their calcined derivatives: Preparation, characterization and application on textile dyes removal Journal of Materials Research and Technology 6 3 271 283 https://doi.org/10.1016/j.jmrt.2016.09.007
J Fu Z Chen M Wang S Liu J Zhang J Zhang 2015 Adsorption of methylene blue by a high-efficiency adsorbent (polydopamine microspheres): Kinetics, isotherm, thermodynamics and mechanism analysis Chemical Engineering Journal 259 53 61 https://doi.org/10.1016/j.cej.2014.07.101
V Geissen H Mol E Klumpp G Umlauf M Nadal M Ploeg van der 2015 Emerging pollutants in the environment: A challenge for water resource management International Soil and Water Conservation Research 3 1 57 65 https://doi.org/10.1016/j.iswcr.2015.03.002
F Granados Correa J Serrano Gómez 2006 Kinetic and thermodynamic parameters of 99Mo sorption on thermally treated hydrotalcite Journal of Radioanalytical and Nuclear Chemistry 268 1 95 101 https://doi.org/10.1556/jrnc.268.2006.1.15
Halden, R. U., Lindeman, A. E., Aiello, A. E., Andrews, D., Arnold, W. A., Fair, P., et al. (2017). The florence statement on triclosan and triclocarban. Environmental health perspectives, 125(6). https://doi.org/10.1289/EHP1788
YS Ho G McKay 1999 Pseudo-second order model for sorption processes Process Biochemistry 34 5 451 465 https://doi.org/10.1016/S0032-9592(98)00112-5
Iturbe-García, J. L., Bonifacio Martínezs, J., Granados Correa, F., & López-Muñoz, B. E. (2019). Behavior of a hydrotalcite type material obtained from MgAl alloy for CO2 adsorption. Applied clay science, 183. doi: https://doi.org/10.1016/j.clay.2019.105296
Jiang, N., Shang, R., Heijman, S. G. J., & Rietveld, L. C. (2020). Adsorption of triclosan, trichlorophenol and phenol by high-silica zeolites: Adsorption efficiencies and mechanisms. Separation and purification technology, 235. doi: https://doi.org/10.1016/j.seppur.2019.116152
H Kaur G Hippargi GR Pophali A Bansiwal 2019 Biomimetic lipophilic activated carbon for enhanced removal of triclosan from water Journal of Colloid and Interface Science 535 111 121 https://doi.org/10.1016/j.jcis.2018.09.093
Khalkhali, M., Zhu, X., Shi, Y., Liu, Q., Choi, P., & Zhang, H. (2020). Structure and CO2 physisorption capacity of hydrotalcite-derived oxide. Journal of CO2 utilization, 36, 64–75. https://doi.org/10.1016/j.jcou.2019.10.019
Khatem, R., Ojeda Miguel, R., & Bakhti, A. (2015). Use of synthetic clay for removal of diclofenac anti-inflammatory. Eurasian J Soil Sci (Vol. 2015).
RF Lehutso AP Daso JO Okonkwo 2017 Occurrence and environmental levels of triclosan and triclocarban in selected wastewater treatment plants in Gauteng Province South Africa. Emerging Contaminants 3 3 107 114 https://doi.org/10.1016/j.emcon.2017.07.001
JG López-Castillo G Macedo-Miranda S Martínez-Gallegos E Ordoñez- Regíl S Álvarez-García J Illescas- Martínez 2020 Fluoride removal using a MgFe hydrotalcite and a MgFe oxide MRS Advances 5 62 3239 3246 https://doi.org/10.1557/adv.2020.420
MJD Low 1960 Kinetics of chemisorption of gases on solids Chemical Reviews 60 3 267 312 https://doi.org/10.1021/cr60205a003
N Lozano CP Rice M Ramirez A Torrents 2010 Fate of triclosan in agricultural soils after biosolid applications Chemosphere 78 6 760 766 https://doi.org/10.1016/j.chemosphere.2009.10.043
Mbarki, F., & Kesraoui, A. (2018). Kinetic, Thermodynamic, and adsorption behavior of cationic and anionic dyes onto corn stigmata : Nonlinear and stochastic analyses.
Mishra, G., Dash, B., & Pandey, S. (2018, March 1). Layered double hydroxides: A brief review from fundamentals to application as evolving biomaterials. Applied clay science. Elsevier Ltd. https://doi.org/10.1016/j.clay.2017.12.021
D Mitrogiannis G Markou A Çelekli H Bozkurt 2015 Biosorption of methylene blue onto Arthrospira platensis biomass: Kinetic, equilibrium and thermodynamic studies Journal of Environmental Chemical Engineering 3 2 670 680 https://doi.org/10.1016/j.jece.2015.02.008
LK Ong FE Soetaredjo A Kurniawan A Ayucitra JC Liu S Ismadji 2014 Investigation on the montmorillonite adsorption of biocidal compounds incorporating thermodynamical-based multicomponent adsorption isotherm Chemical Engineering Journal 241 9 18 https://doi.org/10.1016/j.cej.2013.12.001
FJ Peng CG Pan M Zhang NS Zhang R Windfeld D Salvito 2017 Occurrence and ecological risk assessment of emerging organic chemicals in urban rivers: Guangzhou as a case study in China Science of the Total Environment 589 46 55 https://doi.org/10.1016/j.scitotenv.2017.02.200
Quan, B., Li, X., Zhang, H., Zhang, C., Ming, Y., Huang, Y., et al. (2019, December 15). Technology and principle of removing triclosan from aqueous media: A review. Chemical engineering journal. Elsevier B.V. https://doi.org/10.1016/j.cej.2019.122185
V Rives 2002 Characterization of layered double hydroxides and their decomposition products Materials Chemistry and Physics 75 1–3 19 25 https://doi.org/10.1016/S0254-0584(02)00024-X
M Rosset LW Sfreddo GEN Hidalgo OW Perez-Lopez LA Féris 2019 Adsorbents derived from hydrotalcites for the removal of diclofenac in wastewater Applied Clay Science 175 150 158 https://doi.org/10.1016/j.clay.2019.04.014
L Santamaría F Devred EM Gaigneaux MA Vicente SA Korili A Gil 2020 Effect of the surface properties of Me2+/Al layered double hydroxides synthesized from aluminum saline slag wastes on the adsorption removal of drugs Microporous and Mesoporous Materials 309 August 110560 https://doi.org/10.1016/j.micromeso.2020.110560
Santamaría, L., Vicente, M. A., Korili, S. A., & Gil, A. (2020). Progress in the removal of pharmaceutical compounds from aqueous solution using layered double hydroxides as adsorbents: A review. Journal of environmental chemical engineering, 8(6). https://doi.org/10.1016/j.jece.2020.104577
MN Sepehr TJ Al-Musawi E Ghahramani H Kazemian M Zarrabi 2017 Adsorption performance of magnesium/aluminum layered double hydroxide nanoparticles for metronidazole from aqueous solution Arabian Journal of Chemistry 10 5 611 623 https://doi.org/10.1016/j.arabjc.2016.07.003
AA Sharipova SB Aidarova NY Bekturganova A Tleuova M Kerimkulova O Yessimova 2017 Triclosan adsorption from model system by mineral sorbent diatomite Colloids and Surfaces a: Physicochemical and Engineering Aspects 532 97 101 https://doi.org/10.1016/j.colsurfa.2017.06.012
Z Xu J Fan S Zheng F Ma D Yin 2009 On the adsorption of tetracycline by calcined magnesium-aluminum hydrotalcites Journal of Environmental Quality 38 3 1302 1310 https://doi.org/10.2134/jeq2008.0246
J Xu J Niu X Zhang J Liu G Cao X Kong 2015 Sorption of triclosan on electrospun fibrous membranes: Effects of pH and dissolved organic matter Emerging Contaminants 1 1 25 32 https://doi.org/10.1016/j.emcon.2015.05.002
JTC Yokoyama AL Cazetta KC Bedin L Spessato JM Fonseca PS Carraro 2019 Stevia residue as new precursor of CO 2 -activated carbon: Optimization of preparation condition and adsorption study of triclosan Ecotoxicology and Environmental Safety 172 403 410 https://doi.org/10.1016/j.ecoenv.2019.01.096
Yu, M., Du, N., & Hou, W. (2021). Model prediction of the point of zero net charge of layered double hydroxides and clay minerals. Colloids and surfaces A: physicochemical and engineering aspects, 611. doi: https://doi.org/10.1016/j.colsurfa.2020.125860
Funding
The authors are grateful for the financial support from the National Technological Institute of Mexico (TecNM), the LIIA Instituto Tecnológico de Toluca (ITTol), and the CONACYT postdoctoral scholarship No. 003632 awarded to Sonia Alvarez García CVU 553982.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Macedo-Miranda, G., Martínez-Gallegos, S., Ordoñez-Regíl, E. et al. Triclosan Removal on a MgAl Hydrotalcite and its Calcined Product. Water Air Soil Pollut 233, 47 (2022). https://doi.org/10.1007/s11270-022-05521-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-022-05521-z