Abstract
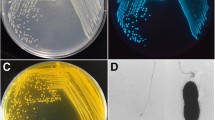
Salmonella enteritidis is one of the most important foodborne pathogens that cause numerous outbreaks worldwide. Some strains of Salmonella have become progressively resistant to antibiotics, so they could represent a critical threat to public health and have led to the use of alternative therapeutic approaches like phage therapy. In this study, a lytic phage, vB_SenS_TUMS_E4 (E4), was isolated from poultry effluent and characterized to evaluate its potential and efficacy for bio-controlling S. enteritidis in foods. Transmission electron microscopy revealed that E4 has a siphovirus morphotype, with an isometric head and non-contractile tail. Determining the host range showed that this phage can effectively infect different motile as well as non-motile Salmonella enterica serovars. The biological characteristics of E4 showed that it has a short latent period of about 15 min and a large burst size of 287 PFU/cell, and is also significantly stable in a broad range of pHs and temperatures. The E4 whole genome contains 43,018 bp and encodes 60 coding sequences (CDSs) but no tRNA genes. Bioinformatics analysis revealed that the genome of E4 lacks any genes related to lysogeny behavior, antibiotic resistance, toxins, or virulence factors. The efficacy of phage E4 as a bio-control agent was assessed in various foodstuffs inoculated with S. enteritidis at 4°C and 25°C, and the resulting data indicated that it could eradicate S. enteritidis after a very short time of 15 min. The findings of the present study showed that E4 is a hopeful candidate as a bio-control agent against S. enteritidis and has the potential to be used in various foodstuffs.








Similar content being viewed by others
Data availability statement
Our data set can be found in online repositories. Repository name/repositories and access number (s) can be found below: https://www.ncbi.nlm.nih.gov/nuccore/MZ955866.1/.
References
Ackermann Hans-W (2009) ‘Phage classification and characterization.‘ in, Bacteriophages (Springer). https://doi.org/10.1007/978-1-60327-164-6_13
Adriaenssens E, Rodney Brister J (2017) ‘How to name and classify your phage: an informal guide’, Viruses, 9: 70. https://doi.org/10.3390/v9040070
Amarillas L, Rubí-Rangel L Cristobal Chaidez, Arturo González-Robles, Luis Lightbourn-Rojas, and Josefina León-Félix (2017) ‘isolation and characterization of phiLLS, a novel phage with potential biocontrol agent against multidrug-resistant Escherichia coli’,Front. Microbiol., 8:1355. https://doi.org/10.3389/fmicb.2017.01355
Brewer ML, Lori Freitag Diachin PM, Knupp T, Leurent, Darryl JM (2003)”The Mesquite Mesh Quality Improvement Toolkit.“ In IMR
Caflisch KM, Gina A, Suh, Patel R (2019) Biological challenges of phage therapy and proposed solutions: a literature review. Expert Rev anti-infective therapy 17:1011–1041. https://doi.org/10.1080/14787210.2019.1694905
Casjens S, Winn-Stapley DA, Gilcrease EB, Morona R, Kühlewein C, Chua JEH, Manning PA, Inwood W, Alvin JC (2004) The chromosome of Shigella flexneri bacteriophage Sf6: complete nucleotide sequence, genetic mosaicism, and DNA packaging. J Mol Biol 339:379–394. https://doi.org/10.1016/j.jmb.2004.03.068
Chan PP, Todd M, Lowe (2019) ‘tRNAscan-SE: searching for tRNA genes in genomic sequences.‘ in, Gene prediction (Springer). https://doi.org/10.1007/978-1-4939-9173-0_1
Clokie MRJ, Kropinski A (2009) ‘Methods and protocols, volume 1: Isolation, characterization, and interactions.‘ in, Bacteriophages. https://doi.org/10.1007/978-1-60327-164-6
Dann, Turner Matthew E., Wand Yves, Briers Rob, Lavigne J. Mark, Sutton Darren M., Reynolds (2017) Characterisation and genome sequence of the lytic Acinetobacter baumannii bacteriophage vB_AbaS_Loki PLOS ONE 12(2):e0172303. https://doi.org/10.1371/journal.pone.0172303
Fong K, LaBossiere B, Andrea IM, Switt P, Delaquis L, Goodridge, Roger C, Levesque MD, Danyluk, Wang S (2017) Characterization of four novel bacteriophages isolated from British Columbia for control of non-typhoidal Salmonella in vitro and on sprouting alfalfa seeds. Front Microbiol 8:2193. https://doi.org/10.3389/fmicb.2017.02193
Gomez-Garcia J, Segundo-Arizmendi AChavez-CarbajalN, Miriam G, Baron-Pichardo SE, Mendoza-Elvira E, Hernandez-Baltazar AP, Hynes, and Oscar Torres-Angeles (2021) ‘Efficacy of Salmonella Bacteriophage S1 DeliveredReleased by Alginate Beads in a Chicken Model of Infection’, Viruses, 13: 1932. https://doi.org/10.3390/v13101932
Grant, Jason R, Adriano S, Arantes, Stothard P (2012) Comparing thousands of circular genomes using the CGView Comparison Tool. BMC genom 13:1–8. https://doi.org/10.1186/1471-2164-13-202
Górski A, Międzybrodzki R Grzegorz Węgrzyn, Ewa Jończyk-Matysiak, Jan Borysowski, and Beata Weber‐Dąbrowska (2020) ‘phage therapy: current status and perspectives’,Med. Res. Rev., 40:459–63. https://doi.org/10.1002/med.21593
Hany, Anany Andrea I. Moreno, Switt Niall, De Lappe Hans-Wolfgang, Ackermann Darren M., Reynolds Andrew M., Kropinski Martin, Wiedmann Mansel W., Griffiths Denise, Tremblay Sylvain, Moineau John H. E., Nash Dann, Turner (2015) A proposed new bacteriophage subfamily: “Jerseyvirinae” Archives of Virology 160(4):1021–1033. https://doi.org/10.1007/s00705-015-2344-z
Islam MS, Mizan YHuMFurkanurR, Nime TYanI, Zhou Y, Li J (2020) ‘Characterization of salmonella phage LPST153 that effectively targets most prevalent salmonella serovars’, Microorganisms, 8: 1089. https://doi.org/10.3390/microorganisms8071089
Jurczak-Kurek A, Gąsior T, Bożena Nejman-Faleńczyk, Bloch S, Dydecka A, Topka G, Malwina Richert (2016) Agnieszka Necel, Magdalena Jakubowska-Deredas, Magdalena Narajczyk, and ‘Biodiversity of bacteriophages: morphological and biological properties of a large group of phages isolated from urban sewage’, Sci. Rep, 6: 1–17. https://doi.org/10.1038/srep34338
Kaliniene L, Noreika A, Kaupinis A, Valius M, Jurgelaitis E, Lazutka J, Meškienė R, Meškys R (2021) Analysis of a novel bacteriophage vB_AchrS_AchV4 highlights the diversity of Achromobacter Viruses. Viruses 13:374. https://doi.org/10.3390/v13030374
Katoh K, Daron M, Standley (2016) A simple method to control over-alignment in the MAFFT multiple sequence alignment program. Bioinformatics 32:1933–1942. https://doi.org/10.1093/bioinformatics/btw108
Khan Mirzaei, Mohammadali, Anders S, Nilsson (2015) Isolation of phages for phage therapy: a comparison of spot tests and efficiency of plating analyses for determination of host range and efficacy. PLoS ONE 10:e0118557. https://doi.org/10.1371/journal.pone.0118557
Kim ES, Bae HW, Cho YH. (2018). A pilin region affecting host range of the pseudomonas aeruginosa RNA phage, PP7. Front Microbiol, 2018 Feb 16;9:247. https://doi.org/10.3389/fmicb.2018.00247
Kim H, Joong JW, Jun SS, Giri SG, Kim SW, Kim J, Kwon SB, Lee C, Chi, and Se Chang Park (2020) ‘Bacteriophage cocktail for the prevention of multiple-antibiotic-resistantmono-phage-resistant Vibrio coralliilyticus infection in pacific oyster (Crassostrea gigas) larvae’, Pathogens, 9: 831. https://doi.org/10.3390/pathogens9100831
Kim S, Lee D-W, Jin J-S, Kim J (2020) Antimicrobial activity of LysSS, a novel phage endolysin, against Acinetobacter baumannii and Pseudomonas aeruginosa. J Glob Antimicrob Resist 22:32–39. https://doi.org/10.1016/j.jgar.2020.01.005
Kropinski AM, Amanda Mazzocco TE, Waddell E, Lingohr, Roger PJ (2009) Enumeration of bacteriophages by double agar overlay plaque assay.‘ in. Bacteriophages. Springer. https://doi.org/10.1007/978-1-60327-164-6_7
K., Shahin L., Zhang H., Bao A., Hedayatkhah A., Soleimani‐Delfan M., Komijani T., He M., Barazandeh M., Mansoorianfar M., Bouzari R., Wang (2021) An in‐vitro study on a novel six‐phage cocktail against multi‐drug resistant‐ESBL Shigella in aquatic environment Letters in Applied Microbiology 72(3) 231–237. https://doi.org/10.1111/lam.v72.310.1111/lam.13418
Kuźmińska-Bajor M, Śliwka P, Ugorski M, Korzeniowski P, Skaradzińska A, Kuczkowski M, Narajaczyk M, Wieliczko A, Rafał K (2021) Genomic and functional characterization of five novel Salmonella-targeting bacteriophages. Virol J 18:1–14. https://doi.org/10.1186/s12985-021-01655-4
Kwon J, Kim SG, Kim HJ, Giri SS, Kim SW, Lee SB, and Se Chang Park (2021) ‘Isolationcharacterization of Salmonella jumbo-phage pSal-SNUABM-04’, Viruses, 13: 27. https://doi.org/10.3390/v13010027
Li J, Li Y, Ding Y, Huang C, Zhang Yu, Wang J, Wang X (2021) Characterization of a novel Siphoviridae Salmonella bacteriophage T156 and its microencapsulation application in food matrix. Int Food Res J 140:110004. https://doi.org/10.1016/j.foodres.2020.110004
Loc-Carrillo, Catherine, Stephen T, Abedon (2011) ‘Pros and cons of phage therapy’, Bacteriophage, 1: 111 – 14. https://doi.org/10.4161/bact.1.2.14590
Lu M, Liu H, Lu H, Liu R, Xinchun, Liu (2020) Characterization and genome analysis of a novel salmonella phage vB_SenS_SE1. Curr Microbiol 77:1308–1315
Medalla F, Gu W, Mahon BE, Judd M, Folster J, Patricia M, Griffin, Robert M, Hoekstra (2017) Estimated incidence of antimicrobial drug–resistant nontyphoidal Salmonella infections, United States, 2004–2012. Emerg Infect Dis 23:29. https://doi.org/10.3201/eid2301.160771
Minh B, Quang HA, Schmidt O, Chernomor D, Schrempf MD, Woodhams AV, Haeseler, Lanfear R (2020) IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol Biol Evol 37:1530–1534. https://doi.org/10.1093/molbev/msaa015
Philipson CW, Logan J, Voegtly MR, Lueder, Kyle A, Long GK, Rice KG, Frey B, Biswas, Regina Z, Cer T, Hamilton, Kimberly A, Bishop-Lilly (2018) ‘Characterizing phage genomes for therapeutic applications’, Viruses, 10: 188. https://doi.org/10.3390/v10040188
Principi N, Silvestri E, Esposito S (2019) Advantages and limitations of bacteriophages for the treatment of bacterial infections. Front pharmacol 10:513. https://doi.org/10.3389/fphar.2019.00513
Rambaut A (2009) ‘FigTree v1. 3.1’, http://tree.bio.ed.ac.uk/software/figtree/.
Richter M, Rosselló-Móra R, Glöckner FO, Jörg, Peplies (2016) JSpeciesWS: a web server for prokaryotic species circumscription based on pairwise genome comparison. J Bioinform 32:929–931. https://doi.org/10.1093/bioinformatics/btv681
Ross A, Ward S, Hyman P (2016) More is better: selecting for broad host range bacteriophages. Front Microbiol 7:1352. https://doi.org/10.3389/fmicb.2016.01352
Russell, Daniel A (2018) Sequencing, assembling, and finishing complete bacteriophage genomes.‘ in. Bacteriophages. Springer. https://doi.org/10.1007/978-1-4939-7343-9_9
Shahin K, Barazandeh M, Zhang L, Hedayatkhah A, He T, Bao H, Mansoorianfar M, Pang M, Wang H, Wei R, Wang R (2021) Biodiversity of new lytic bacteriophages infecting Shigella spp. in freshwater environment. Front Microbiol, 12:619323. https://doi.org/10.3389/fmicb.2021.619323
Shahin, Khashayar, Mohadeseh Barazandeh, Lili Zhang, Abolghasem Hedayatkhah, Tao He, Hongduo Bao, Mojtaba Mansoorianfar, Maoda Pang, Heye Wang, and Ruicheng Wei (2021) 'Biodiversity of new lytic bacteriophages infecting Shigella spp. in freshwater environment’, Front Microbiol, 12:619323.
Shahin K, Zhang L, Delfan AS, Komijani M, Hedayatkhah A, Bao H, Barazandeh M, Mansoorianfar M, Pang M, He T, Bouzari M. (2021). Effective control of Shigella contamination in different foods using a novel six-phage cocktail. LWT. 2021 Jun 1;144:111137. https://doi.org/10.1016/j.lwt.2021.111137
Shahrbabak S, Sabouri Z, Khodabandehlou AR, Shahverdi M, Skurnik H-W, Ackermann M, Varjosalo MT, Yazdi, Zargham Sepehrizadeh (2013) Isolation, characterization and complete genome sequence of PhaxI: a phage of Escherichia coli O157: H7. Microbiology 159:1629–1638. https://doi.org/10.1099/mic.0.063776-0
Shang Y, Sun Q, Chen H, Wu Q, Chen M, Yang S, Du M, Zha F, Ye Q, Zhang J (2021) Isolation and characterization of a Novel Salmonella Phage vB_SalP_TR2. Front Microbiol 12:1452. https://doi.org/10.3389/fmicb.2021.664810
Shen A (2021) and Andrew Millard ‘Phage Genome Annotation: Where to Begin and End’, PHAGE, 2: 183 – 93. https://doi.org/10.1089/phage.2021.0015
Smith, H Williams, MB Huggins (1980) ‘The association of the O18, K1 and H7 antigens and the CoIV plasmid of a strain of Escherichia coli with its virulence and immunogenicity’, Microbiology, 121: 387–400. https://doi.org/10.1099/00221287-121-2-387
Sullivan MJ, Nicola K, Petty, Scott AB (2011) Easyfig: a genome comparison visualizer. J Bioinform 27:1009–1010. https://doi.org/10.1093/bioinformatics/btr039
Sun Z, Wen H, Ma L, Chen Z (2022) Isolation, characterization and application of bacteriophage PSDA-2 against Salmonella Typhimurium in chilled mutton. PLoS ONE 17:e0262946. https://doi.org/10.1371/journal.pone.0262946
Torres-Acosta, Mario A, Viviana Clavijo C, Vaglio AF, González‐Barrios, Martha JVives‐Flórez, Marco, Rito‐Palomares (2019) Economic evaluation of the development of a phage therapy product for the control of Salmonella in poultry. Biotechnol Prog 35:e2852. https://doi.org/10.1002/btpr.2852
Van Twest, Rohan, Andrew M, Kropinski (2009) Bacteriophage enrichment from water and soil.‘ in, Bacteriophages. Springer. https://doi.org/10.1007/978-1-60327-164-6_2
Waseh S, Hanifi-Moghaddam P, Coleman R, Masotti M, Ryan S, Foss M, MacKenzie R, Henry M, Szymanski CM, Tanha J (2010) Orally administered P22 phage tailspike protein reduces Salmonella colonization in chickens: prospects of a novel therapy against bacterial infections. PLoS ONE 5:e13904. https://doi.org/10.1371/journal.pone.0013904
Xiong D, Song L, Pan Z, Xinan J (2018) Identification and discrimination of Salmonella enterica serovar gallinarum biovars pullorum and gallinarum based on a one-step multiplex PCR assay. Front Microbiol 9:1718. https://doi.org/10.3389/fmicb.2018.01718
Young R (2014) Phage lysis: three steps, three choices, one outcome. J Microbiol 52:243–258. https://doi.org/10.1007/s12275-014-4087-z
Acknowledgements
We thank Professor Zahraei Salehi of the Department of Microbiology, Faculty of Veterinary Medicine, Tehran University for donating strains of S. enteritidis ATCC 13076. We also appreciate the support of IKHC and ABRII in accessing clinical and environmental samples.
Funding
This work was funded by the Deputy of Research, Tehran University of Medical Sciences (research code 99-1-104-48232).
Author information
Authors and Affiliations
Contributions
Narges Torkashvand: Data curation; Formal analysis; Investigation; Methodology; Writing original draft; Software. Haniyeh Kamyab: Data curation; Formal analysis; Investigation; Methodology; Software. Ahmad Reza Shahverdi: Supervision, Project administration; Funding acquisition. Mohammad Reza Khoshayand: Supervision; Validation. Zargham Sepehrizadeh: Conceptualization, Supervision, Project administration, Funding acquisition, Writing - review & editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics approval and consent to participate
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Torkashvand, N., Kamyab, H., Shahverdi, A.R. et al. Isolation, characterization, and genome analysis of a broad host range Salmonella phage vB_SenS_TUMS_E4: a candidate bacteriophage for biocontrol. Vet Res Commun 47, 1493–1503 (2023). https://doi.org/10.1007/s11259-023-10105-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-023-10105-1