Abstract
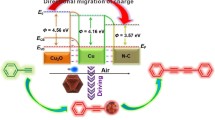
Phenols are one of the most important classes of raw materials in the chemical industry, and a variety of compounds are derived from them. The direct conversion of benzene to phenol by hydroxylation is an economical and environmentally friendly method among phenol syntheses, especially when such a reaction can be driven by solar energy. Herein, for the first, a CuO/Cu-MOF/GO photocatalyst which was facilely prepared from a single Cu-MOF/GO template by partial pyrolysis. Compared to completely pyrolysis, the CuO/Cu-MOF/GO got by partial pyrolysis exhibited significantly higher photocatalytic performance towards the hydroxylation of benzene to phenol. It was characterized in detail by SEM, XRD, FT-IR, UV–Vis-DRS, XPS, and N2 adsorption–desorption method. Its catalytic performance for direct hydroxylation of benzene to phenol has been investigated. The results showed that the conversion of benzene and the yield of phenol were 38.7% and 20.1% which were 4.1 times and 5.2 times higher than those using Cu-MOF/GO, respectively. The CuO/Cu-MOF/GO heterostructures showed remarkably enhanced photocatalytic activity toward the hydroxylation of benzene to phenol, which can be attributed to the band gap narrowing and enhanced photogenerated electron–hole separation ability. This work may provide useful guidance for the design of MOF-derived photocatalyst for the applications in the hydroxylation of aromatics.


















Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
References
Y. Ma, W. Zhang, C. Wang et al., J. Appl. Polym. Sci. 129(6), 3096 (2013)
J.Y. Shin, D.J. Jung, S.G. Lee, Acs Catal. 3(4), 525 (2013)
X. Gao, X. Jie, Catal. Lett. 111(3), 203 (2006)
I. Kuzniarska-biernacka, M.M. Raposo, R. Batista et al., Micropor. Mesopor. Mat. 227, 272 (2016)
S. Buttha, S. Youngme, J. Wittayakun et al., Mol. Catal. 461, 26 (2018)
L. Zeng, H. Liang, P. An et al., Appl. Catal. A Gen. 633, 118499 (2022)
X. Li, S. Li, W. Jia et al., New J. Chem. 46(6), 2908 (2022)
Z. Wang, H. Hojo, H. Einaga, Chem. Eng. J. 427(2), 131369 (2022)
L.K. Putri, W.J. Ong, W.S. Chang et al., Appl. Surf. Sci. 358, 2 (2015)
C. Zhu, J. Yun, Q. Wang et al., Adv. Theor. Simul. 1(4), 1800005 (2018)
S.S. Acharyya, S. Ghosh, R. Tiwari et al., ACS Catal. 5(5), 2850 (2015)
C. Wang, L. Hu, M. Wang et al., Chin. J. Catal. 37, 2003 (2016)
M. Padervand, B. Rhimi, C. Wang, J. Alloys Compd. 852, 156955 (2021)
E.A. Dawi, M. Padervand, S. Ghasemi et al., J. Water Process Eng. 54, 103979 (2023)
B. Abdollahi, D. Salari, M. Zarei, J. Environ. Eng. 10(2), 107144 (2022)
J.J. Liu, Z.W. Jiang, S.W. Hsu, ACS Appl. Mater. Interfaces 15(5), 6716 (2023)
S. Li, F. Wu, R. Lin et al., Chem. Eng. J. 429, 132217 (2022)
K. Kioka, N. Mizutani, N. Hosono et al., ACS Nano 16(4), 6771 (2022)
B. Abdollahi, A. Najafidoust, E.A. Asl et al., Arab. J. Chem. 14(12), 103444 (2021)
Z. Li, S. Qiu, G. Jiang et al., Asian J. Org. Chem. 7(1), 15 (2018)
P. Cui, Y. Xue, Spectrochim. Acta Part A 287(Pt 1), 122117 (2023)
G. Ramalingam, N. Perumal, A.K. Priya et al., Chemosphere 300, 134391 (2022)
J. He, M. Zhang, P. Ana et al., J. Mater. Chem. A 6(40), 19782 (2018)
X. Li, J. Chem. 2018, 1 (2018)
H. Pazoki, M. Anbia, Polyhedron 171, 108 (2019)
A. Ghorbanichoghamarani, H. Bastan, Z. Kakakhani et al., RSC Adv. 11, 14905 (2011)
M. Saraf, R. Rajak, M.M. Shaikh, J. Mater. Chem. A 4, 16432 (2016)
C. Li, T. Zhang, J. Zhao et al., ACS Appl. Mater. Interfaces 9(3), 2984 (2017)
X. Wang, Q. Wang, Q. Wang et al., Appl. Mater. Interfaces 6(14), 11573 (2014)
S. Sadeghi, M. Jafarzadeh, A.R. Abbasi et al., New J. Chem. 41(20), 12014 (2017)
E. Akbarzadeh, H.Z. Soheili, M. Hosseinifard et al., Mater. Res. Bull. 121(198), 110621 (2020)
M. Xu, Y. Zhu, J.Y. Ji et al., MATEC Web Conf. 26, 2002 (2015)
D.F. Mostafa, A.A. Farhad, R.E. Milad et al., J. Water Process Eng. 34, 101180 (2019)
M.D. Firouzjaei, A.A. Shamsabadi, G.M. Sharifian et al., Adv. Mater. Interfaces 5(11), 1701365 (2018)
M.L. Rahman, Z.J. Wong, M.S. Sarjadi et al., J. Appl. Polym. Sci. 138(2), 49671 (2021)
P. Arul, N.S.K. Gowthaman, S.A. John et al., ACS Omega 5(24), 14242 (2020)
C. Petit, J.B. Teresa, Adv. Mater. 21, 4753 (2009)
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No. 22005242), and the Shaanxi Provincial Natural Science Foundation (No. 2018JM2014).
Funding
Shaanxi Provincial Natural Science Foundation (No. 2018JM2014), National Natural Science Foundation of China (No. 22005242).
Author information
Authors and Affiliations
Contributions
ZM contributed to investigation, methodology, and writing of the original draft. JS contributed to synthesis, performance measurement, and writing of the original draft. WH contributed to methodology. ZL contributed to performance measurement.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interest or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Meng, Z., Shao, J., Han, W. et al. Facile synthesis of CuO/Cu-MOF/GO for efficient photocatalytic benzene hydroxylation to phenol. Res Chem Intermed 49, 5485–5504 (2023). https://doi.org/10.1007/s11164-023-05140-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-05140-1