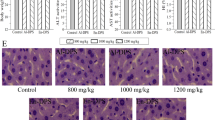
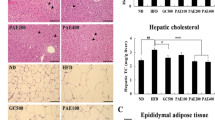
Abstract
The aim of this study was to investigate the functional mechanism of Wuniuzao dark tea polysaccharide (WDTP) that protect against hyperlipidemia in mice induced by high-fat diet. WDTP was extracted by hot water, isolated and purified by DEAE-52 chromatography and characterized by high-performance liquid chromatograph (HPLC), Fourier transform infrared spectroscopy (FT-IR) and scanning electron microscope (SEM). Different doses (200 or 800 mg/kg/day) of WDTP were orally administered to mice induced by high-fat diet to evaluate the mechanism of WDTP regulating lipid metabolism. And these results showed that average molecular weight of WDTP was nearly 63,869 Da. And WDTP intervention significantly reduced body weight, lipid accumulation, and modulated blood lipid levels. The mechanism of WDTP ameliorating lipid metabolism was associated with regulating the expression of lipid metabolism-related genes and serum exosomes miR-19b-3p, and modulating the community structure of gut microbiota in mice.






Similar content being viewed by others
Data Availability
Raw sequence data from all 16 S rRNA sequencing experiments in this study have been deposited in the GSA (https://ngdc.cncb.ac.cn/gsa/browse/) under accession number CRA012322.
References
Rahman MS, Woollard K (2017) Atherosclerosis. Adv Exp Med Biol 1003:121–144. https://doi.org/10.1007/978-3-319-57613-8-7
Sun J, Zhou W, Gu T et al (2018) A retrospective study on association between obesity and cardiovascular risk diseases with aging in Chinese adults. Sci Rep 8(1):5806. https://doi.org/10.1038/s41598-018-24161-0
Chen G, Yuan Q, Saeeduddin M et al (2016) Recent advances in tea polysaccharides: extraction, purification, physicochemical characterization and bioactivities. Carbohydr Polym 153:663–678. https://doi.org/10.1016/j.carbpol.2016.08.022
Harvey AL, Edrada-Ebel R, Quinn RJ (2015) The re-emergence of natural products for drug discovery in the genomics era. Nat Rev Drug Discov 14(2):111–129. https://doi.org/10.1038/nrd4510
Jin M, Huang Q, Zhao K et al (2013) Biological activities and potential health benefit effects of polysaccharides isolated from Lycium barbarum L. Int J Biol Macromol 54:16–23. https://doi.org/10.1016/j.ijbiomac.2012.11.023
Deng X, Li X, Luo S et al (2017) Antitumor activity of Lycium barbarum polysaccharides with different molecular weights: an in vitro and in vivo study. Food Nutr Res 61(1):1399770. https://doi.org/10.1080/16546628.2017.1399770
Wang Y, Li Y, Liu Y et al (2015) Extraction, characterization and antioxidant activities of Se-enriched tea polysaccharides. Int J Biol Macromol 77:76–84. https://doi.org/10.1016/j.ijbiomac.2015.02.052
Nie C, Zhu P, Ma S et al (2018) Purification, characterization and immunomodulatory activity of polysaccharides from stem lettuce. Carbohydr Polym 188:236–242. https://doi.org/10.1016/j.carbpol.2018.02.009
Xiong X, Wenjian Y, Gangliang H et al (2023) Ultrasonic-assisted extraction, characteristics and activity of Ipomoea batatas polysaccharide. Ultrason Sonochem 96:106420. https://doi.org/10.1016/j.ultsonch.2023.106420
Mao Y, Wei B, Teng J et al (2017) Polysaccharides from Chinese Liupao dark tea and their protective effect against hyperlipidemia. Int J Food Sci Technol 53(3):599–607. https://doi.org/10.1111/ijfs.13633
Xu Y, Zhang M, Wu T et al (2015) The anti-obesity effect of green tea polysaccharides, polyphenols and caffeine in rats fed with a high-fat diet. Food Funct 6(1):297–304. https://doi.org/10.1039/c4fo00970c
Bougarne N, Weyers B, Desmet SJ et al (2018) Molecular actions of PPARα in lipid metabolism and inflammation. Endocr Rev 39(5):760–802. https://doi.org/10.1210/er.2018-00064
Yang D, Hu C, Deng X et al (2019) Therapeutic effect of Chitooligosaccharide tablets on lipids in High-Fat diets Induced hyperlipidemic rats. Molecules 24(3):514. https://doi.org/10.3390/molecules24030514
Zhou S, You H, Qiu S et al (2022) A new perspective on NAFLD: focusing on the crosstalk between peroxisome proliferator-activated receptor alpha (PPARα) and farnesoid X receptor (FXR). Biomed Pharmacother 154:113577. https://doi.org/10.1016/j.biopha.2022.113577
Villarroya F, Iglesias R, Giralt M (2007) PPARs in the control of uncoupling proteins gene expression. PPAR Res 2007(74364). https://doi.org/10.1155/2007/74364
Schoeler M, Caesar R (2019) Dietary lipids, gut microbiota and lipid metabolism. Rev Endocr Metab Disord 20(4):461–472. https://doi.org/10.1007/s11154-019-09512-0
Patterson E, Ryan PM, Cryan JF et al (2016) Gut microbiota, obesity and diabetes. Postgrad Med J 92(1087):286–300. https://doi.org/10.1136/postgradmedj-2015-133285
Yao Y, Yan L, Chen H et al (2020) Cyclocarya paliurus polysaccharides alleviate type 2 diabetic symptoms by modulating gut microbiota and short-chain fatty acids. Phytomedicine 77:153268. https://doi.org/10.1016/j.phymed.2020.153268
Ley RE, Turnbaugh PJ, Klein S et al (2006) Microbial ecology: human gut microbes associated with obesity. Nature 444(7122):1022–1023. https://doi.org/10.1038/4441022a
Turnbaugh PJ, Hamady M, Yatsunenko T et al (2009) A core gut microbiome in obese and lean twins. Nature 457(7228):480–484. https://doi.org/10.1038/nature07540
Zeng Q, Li D, He Y et al (2019) Discrepant gut microbiota markers for the classification of obesity-related metabolic abnormalities. Sci Rep 9(1):13424. https://doi.org/10.1038/s41598-019-49462-w
Li J, Yuan H, Zhao Z et al (2022) The mitigative effect of isorhamnetin against type 2 diabetes via gut microbiota regulation in mice. Front Nutr 9:1070908. https://doi.org/10.3389/fnut.2022.1070908
Thomou T, Mori MA, Dreyfuss JM et al (2017) Adipose-derived circulating miRNAs regulate gene expression in other tissues. Nature 542(7642):450–455. https://doi.org/10.1038/nature21365
Yu X, Li M, Ling ZM et al (2022) The role of MicroRNAs in Hyperlipidemia: from pathogenesis to therapeutical application. Mediators Inflamm 2022:3101900. https://doi.org/10.1155/2022/3101900
Acknowledgements
We are grateful to thank Luting Ye, Guangmei Li, Shengnan Zhu, Weijia Xu, Zhaowen Chen, Simin Ren, Min Cheng and Jingwu Song (College of Life Sciences, China Jiliang University) for generously providing rats administration. Financial assistance from the National Natural Science Foundation of China and the Major Science and Technology Projects in Zhejiang Province are sincerely thanked.
Funding
This work was supported financially by the National Natural Science Foundation of China (31100499 and 31672394), the Major Science and Technology Projects in Zhejiang Province (2020C02045) and Zhejiang Science and Technology Commissioner Team Project.
Author information
Authors and Affiliations
Contributions
X. Z., Z. D. and J. G. conceived the project. X. Z. and Z. D. analyzed the data. X. Z. and Z. D. wrote the manuscript. X. Z., Z. D., W. L., J. Z., F. G. and J. G. provided critical discussion, editing and final approval of the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
The animal experiments protocol was approved according to the agreement of Laboratory Animal Ethics Committee from China Jiliang University (2022-003), and all animal procedures were performed in accordance with the institutional and national guidelines.
Conflict of Interest
All authors declare that they have known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zou, X., Dong, Z., Liu, W. et al. Modulation Mechanism of Wuniuzao Dark Tea Polysaccharide on Lipid Metabolism in Hyperlipidemic Mice Induced by High-Fat Diet. Plant Foods Hum Nutr 79, 173–181 (2024). https://doi.org/10.1007/s11130-024-01145-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11130-024-01145-2