Abstract
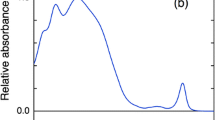
Carotenoids (Cars) exhibit two functions in photosynthesis, light-harvesting and photoprotective functions, which are performed through the excited states of Cars. Therefore, increasing our knowledge on excitation relaxation dynamics of Cars is important for understanding of the functions of Cars. In light-harvesting complexes, there exist Cars functioning by converting the π-conjugation number in response to light conditions. It is well known that some microalgae have a mechanism controlling the conjugation number of Cars, called as the diadinoxanthin cycle; diadinoxanthin (10 conjugations) is accumulated under low light, whereas diatoxanthin (11 conjugations) appears under high light. However, the excitation relaxation dynamics of these two Cars have not been clarified. In the present study, we investigated excitation relaxation dynamics of diadinoxanthin and diatoxanthin in relation to their functions, by the ultrafast fluorescence spectroscopy. After an excitation to the S2 state, the intramolecular vibrational redistribution occurs, followed by the internal conversion to the S1 state. The S2 lifetimes were analyzed to be 175 fs, 155 fs, and 140 fs in diethyl ether, ethanol, and acetone, respectively, for diadinoxanthin, and 155 fs, 135 fs, and 125 fs in diethyl ether, ethanol, and acetone, respectively for diatoxanthin. By converting diadinoxanthin to diatoxanthin, the absorption spectra shift to longer wavelengths by 5–7 nm, and lifetimes of S2 and S1 states decrease by 11–13% and 52%, respectively. Differences in levels and lifetimes of excited states between diadinoxanthin and diatoxanthin are small; therefore, it is suggested that changes in the energy level of chlorophyll a are necessary to efficiently control the functions of the diadinoxanthin cycle.





Similar content being viewed by others
References
Akimoto S, Mimuro M (2005) Excitation relaxation dynamics of carotenoids probed by ultrafast fluorescence spectroscopy. In: Wada N, Mimuro M (eds) Recent progress of bio/chemiluminescence and fluorescence analysis in photosynthesis. Research Signpost, Trivandrum, pp 213–234
Akimoto S, Yamazaki I, Takaichi S, Mimuro M (2000) Excitation relaxation dynamics of linear carotenoids. J Lumin 87–89:797–799
Akimoto S, Yokono M, Ohmae M, Yamazaki I, Tanaka A, Higuchi M, Tsuchiya T, Miyashita H, Mimuro M (2005) Ultrafast excitation relaxation dynamics of lutein in solution and in the light-harvesting complexes II isolated from Arabidopsis thaliana. J Phys Chem B 109:12612–12619
Akimoto S, Teshigahara A, Yokono M, Mimuro M, Nagao R, Tomo T (2014) Excitation relaxation dynamics and energy transfer in fucoxanthin–chlorophyll a/c–protein complexes, probed by time-resolved fluorescence. Biochim Biophys Acta Bioenerg 1837:1514–1521
Akimoto S, Ueno Y, Yokono M, Shen J-R, Nagao R (2020) Adaptation of light-harvesting and energy-transfer processes of a diatom Chaetoceros gracilis to different light qualities. Photosynth Res 146:87–93
Beddard GS, Davidson RS, Trethewey KR (1977) Quenching of chlorophyll fluorescence by β-carotene. Nature 267:373–374
Britton G (1995) UV/Visible spectroscopy. In: Britton G, Liaaen-Jensen S, Pfanser H (eds) Carotenoids, Vol 1B: Spectroscopy. Birkhaüser Verlag, Basel, pp 13–62
Britton G, Liaaen-Jensen S, Pfanser H (2004) Carotenoids handbook. Birkhaüser Verlag, Basel
Buchecker R, Noack K (1995) Circular dichroism. In: Britton G, Liaaen-Jensen S, Pfanser H (eds) Carotenoids. Spectroscopy, vol 1B. Birkhaüser Verlag, Basel, pp 63–116
DeCoster B, Christensen RL, Gebhard R, Lugtenburg J, Farhoosh R, Frank HA (1992) Low-lying electronic states of carotenoids. Biochim Biophys Acta Bioenerg 1102:107–114
Demmig-Adams B (1990) Carotenoids and photoprotection: a role for the xanthophyll cycle zeaxanthin. Biochim Biophys Acta Bioenerg 1020:1–24
Enriquez MM, LaFountain AM, Budarz J, Fuciman M, Gibson GN, Frank HA (2010) Direct determination of the excited state energies of xanthophylls diadinoxanthin and diatoxanthin from Phaeodactylum tricornutum. Chem Phys Lett 493:353–357
Förster T (1948) Intermolecular energy migration and fluorescence. Ann Phys 2:55–75
Frank HA, Cogdell RJ (1996) Carotenoids in photosynthesis. Photochem Photobiol 63:257–264
Frank HA, Farhoosh R, Gebhard R, Lugtenburg J, Gosztola D, Wasielewski MR (1993) The dynamics of the S1 states of carotenoids. Chem Phys Lett 207:88–92
Frank HA, Cua A, Chynwat V, Young A, Gosztola D, Wasielewski MR (1994) Photophysics of the carotenoids associated with the xanthophyll cycle in photosynthesis. Photosynth Res 41:389–395
Frank HA, Cua A, Chynwat V, Young A, Gosztola D, Wasielewski MR (1996) The lifetimes and energies of the first excited singlet states of diadinoxanthin and diatoxanthin: the role of these molecules in excess energy dissipation in algae. Biochim Biophys Acta Bioenerg 1277:243–252
Goodwin TW, Britton G (1988) Distribution and analysis of carotenoids. In: Goodwin TW (ed) Plant pigments. Academic, London, pp 61–132
Hamada F, Murakami A, Akimoto S (2015) Comparative analysis of ultrafast excitation energy-transfer pathways in three strains of divinyl chlorophyll a/b-containing cyanobacterium, Prochlorococcus marinus. J Phys Chem B 119:15993–15600
Herbstová M, Bína D, Koník P, Gardian Z, Vaćha F, Litvín R (2015) Molecular basis of chromatic adaptation in pennate diatom Phaeodactylum tricornutum. Biochim Biophys Acta Bioenerg 1847:534–543
Herbstová M, Bína D, Kaňa R, Vaćha F, Litvín R (2017) Red-light phenotype in a marine diatom involves a specialized oligomeric red-shifted antenna and altered cell morphology. Sci Rep 7:11976
Holt NE, Zigmantas D, Valkunas L, Li X-P, Niyogi KK, Fleming GR (2005) Carotenoid cation formation and the regulation of photosynthetic light harvesting. Science 307:433–436
Horton P, Ruban AV, Young AJ (1999) Regulation of the structure and function of the light harvesting complexes of photosystem II by the xanthophyll cycle. In: Frank HA, Young AJ, Britton G, Cogdell RJ (eds) The photochemistry of carotenoids. Kluwer Academic Publishers, Dordrecht, pp 271–291
Kohler BE (1995) Electronic structure of carotenoids. In: Britton G, Liaaen-Jensen S, Pfanser H (eds) Carotenoids. Spectroscopy, vol 1B. Birkhaüser Verlag, Basel, pp 1–12
Koyama Y, Kuki M, Anderson PO, Gillbro T (1996) Single excited states and the light-harvesting function of carotenoids in bacterial photosynthesis. Photochem Photobiol 63:243–256
Kuczynska P, Jemiola-Rzeminska M, Strzalka K (2015) Photosynthetic pigments in diatoms. Mar Drugs 13:5847–5881
Larkum AWD (2003) Light-harvesting systems in algae. In: Larkum AWD, Douglas SE, Raven JA (eds) Photosynthesis in algae. Kluwer Academic Publishers, Dordrecht, pp 277–304
MacPherson AN, Gillbro T (1998) Solvent dependence of the ultrafast S2–S1 internal conversion rate of β-carotene. J Phys Chem A 102:5049–5058
MacPherson AN, Hiller RG (2003) Light-harvesting systems in chlorophyll c-containing algae. In: Green R, Parson WW (eds) Light-harvesting antennas in photosynthesis. Kluwer Academic Publishers, Dordrecht, pp 323–352
Mimuro M, Akimoto S (2003) Carotenoids of light harvesting systems: energy transfer processes from fucoxanthin and peridinin to chlorophyll. In: Larkum AWD, Douglas SE, Raven JA (eds) Photosynthesis in algae. Kluwer Academic Publishers, Dordrecht, pp 335–349
Nagae H, Kakitani T, Katoh T, Mimuro M (1993) Calculation of the excitation transfer matrix elements between the S2 or S1 state of carotenoid and the S2 or S1 state of bacteriochlorophyll. J Chem Phys 98:8012–8023
Nagao R, Ishii A, Tada O, Suzuki T, Dohmae N, Okumura A, Iwai M, Takahashi T, Kashino Y, Enami I (2007) Isolation and characterization of oxygen-evolving thylakoid membranes and Photosystem II particles from a marine diatom Chaetoceros gracilis. Biochim Biophys Acta Bioenerg 1767:1353–1362
Nagao R, Yokono M, Akimoto S (2013) Tomo, T (2013) High excitation energy quenching in fucoxanthin chlorophyll a/c-binding protein complexes from the diatom Chaetoceros gracilis. J Phys Chem B 117:6888–6895
Nagao R, Kagatani K, Ueno Y, Shen J-R, Akimoto S (2019a) Ultrafast excitation energy dynamics in a diatom photosystem I–antenna complex: a femtosecond fluorescence upconversion study. J Phys Chem B 123:2673–2678
Nagao R, Ueno Y, Yokono M, Shen J-R, Akimoto S (2019b) Effects of excess light energy on excitation-energy dynamics in a pennate diatom Phaeodactylum tricornutum. Photosynth Res 141:355–365
Nagao R, Kato K, Kumazawa M, Ifuku K, Yokono M, Suzuki T, Dohmae N, Akita F, Akimoto S, Miyazaki N, Shen J-R (2022) Structural basis for different types of hetero-tetrameric light-harvesting complexes in a diatom PSII-FCPII supercomplex. Nat Commun 13:1764
Niedzwieddzki DM, Sullivan JO, Plívka T, Brirge RR, Frank HA (2006) Femtosecond time-resolved transient absorption spectro-scopy of xanthophylls. J Phys Chem B 110:22872–22885
N’soukpoé-Kossi CN, Leblanc RM (1987) Absorption and photoacoustic spectroscopies of lutein and zeaxanthin Langmuir-Blodgett films in connection with the Haidinger’s brushes. Can J Chem 66:1459–1466
Oka K, Ueno Y, Yokono M, Shen J-R, Nagao R (2020) Adaptation of light-harvesting and energy-transfer processes of a diatom Phaeodactylum tricornutum to different light qualities. Photosynth Res 146:227–234
Scheer H (2003) The pigments. In: Green R, Parson WW (eds) Light-harvesting antennas in photosynthesis. Kluwer Academic Publishers, Dordrecht, pp 29–81
Shedbalkar VB, Rebeiz CA (1992) Chloroplast biogenesis: determination of the molar extinction coefficients of divinyl chlorophyll a and b and their pheophytins. Anal Biochem 207:261–266
Stransky H, Hager A (1970) The carotenoid pattern and the occurrence of the light induced xanthophyll cycle in various classes of algae. Arch Microbiol 73:315–323
Takaichi S (2020) Carotenoids in phototrophic microalgae: distributions and biosynthesis. In: Jacob-Lopes E, Queiroz MI, Zepka LQ (eds) Pigments from microalgae handbook. Springer, Cham, pp 19–42
Tanaka K, Iida S, Takaichi S, Mimuro M, Murakami A, Akimoto S (2016) Excitation relaxation dynamics and energy transfer in pigment–protein complexes of a dinoflagellate, revealed by ultrafast fluorescence spectroscopy. Photosynth Res 130:183–191
Ueno Y, Nagao R, Shen J-R, Akimoto S (2019) Spectral properties and excitation relaxation of novel fucoxanthin chlorophyll a/c-binding protein complexes. J Phys Chem Lett 10:5148–5152
Yamano Y, Chary MV, Wada A (2012) Stereoselective total synthesis of the acetylenic carotenoids alloxanthin and triophaxanthin. Org Biomol Chem 10:4103–4108
Yamano Y, Eno K, Hikita Y, Kurimoto H, Wada A (2015) Stereocontrolled first total synthesis of salmoxanthin and deoxysalmoxanthin. Curr Org Synt 12:180–188
Young A (1993) Carotenoids in pigment–protein complexes. In: Young A, Britton G (eds) Carotenoids in photosynthesis. Chapman & Hall, London, pp 72–95
Acknowledgements
This work was supported by the Grants-in-Aid for Scientific Research from Japan Society for the Promotion of Science JP17H06433 (to J.-R. S.) and JP16H06553 (to S. A.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kagatani, K., Nagao, R., Shen, JR. et al. Excitation relaxation dynamics of carotenoids constituting the diadinoxanthin cycle. Photosynth Res 154, 13–19 (2022). https://doi.org/10.1007/s11120-022-00944-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-022-00944-5