Abstract
Key Message
Plasma membrane-localized AtAVT6D importing aspartic acid can be targeted to develop plants with enhanced osmotic and nitrogen-starvation tolerance. AtAVT6D promoter can be exploited as a stress-inducible promoter for genetic improvements to raise stress-resilient crops.
Abstract
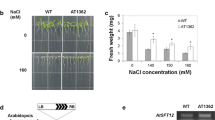
The AtAVT6 family of amino acid transporters in Arabidopsis thaliana has been predicted to export amino acids like aspartate and glutamate. However, the functional characterization of these amino acid transporters in plants remains unexplored. The present study investigates the expression patterns of AtAVT6 genes in different tissues and under various abiotic stress conditions using quantitative Real-time PCR. The expression analysis demonstrated that the member AtAVT6D was significantly induced in response to phytohormone ABA and stresses like osmotic and drought. The tissue-specific expression analysis showed that AtAVT6D was strongly expressed in the siliques. Taking together these results, we can speculate that AtAVT6D might play a vital role in silique development and abiotic stress tolerance. Further, subcellular localization study showed AtAVT6D was localized to the plasma membrane. The heterologous expression of AtAVT6D in yeast cells conferred significant tolerance to nitrogen-deficient and osmotic stress conditions. The Xenopus oocyte studies revealed that AtAVT6D is involved in the uptake of Aspartic acid. While overexpression of AtAVT6D resulted in smaller siliques in Arabidopsis thaliana. Additionally, transient expression studies were performed with the full-length AtAVT6D promoter and its deletion constructs to study the effect of ACGT-N24-ACGT motifs on the reporter gene expression in response to abiotic stresses and ABA treatment. The fluorometric GUS analyses revealed that the promoter deletion construct-2 (Pro.C2) possessing a single copy of ACGT-N24-ACGT motif directed the strongest GUS expression under all the abiotic conditions tested. These results suggest that Pro.C2 can be used as a stress-inducible promoter to drive a significant transgene expression.







Similar content being viewed by others
Data availability
Enquiries about data availability should be directed to the authors.
References
Amir R, Galili G, Cohen H (2018) The metabolic roles of free amino acids during seed development. Plant Sci 275:11–18. https://doi.org/10.1016/j.plantsci.2018.06.011
Angelovici R, Fait A, Zhu X, Szymanski J, Feldmesser E, Fernie AR, Galili G (2009) Deciphering transcriptional and metabolic networks associated with lysine metabolism during Arabidopsis seed development. Plant Physiol 151:2058–2072. https://doi.org/10.1104/PP.109.145631
Angelovici R, Fait A, Fernie AR, Galili G (2011) A seed high-lysine trait is negatively associated with the TCA cycle and slows down Arabidopsis seed germination. New Phytol 189:148–159. https://doi.org/10.1111/J.1469-8137.2010.03478.X
Balazadeh S, Schildhauer J, Araújo WL, Munné-Bosch S, Fernie AR, Proost S, Humbeck K, Mueller-Roeber B (2014) Reversal of senescence by N resupply to N-starved Arabidopsis thaliana: transcriptomic and metabolomic consequences. J Exp Bot 65:3975–3992. https://doi.org/10.1093/jxb/eru119
Besnard J, Sonawala U, Maharjan B, Collakova E, Finlayson SA, Pilot G, McDowell J, Okumoto S (2021) Increased expression of UMAMIT amino acid transporters results in activation of salicylic acid dependent stress response. Front Plant Sci 11:2214. https://doi.org/10.3389/FPLS.2020.606386/BIBTEX
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Chahomchuen T, Sekito T, Hondo K, Nishimoto S, Sugahara T, Kakinuma Y (2008) Characterization of the vacuolar transporters for amino acid recycling in yeast autophagy. Interdiscip Stud Environ Chem Biol Responses Chem Pollut 1:251–261
Chahomchuen T, Hondo K, Ohsaki M, Sekito T, Kakinuma Y (2009) Evidence for Avt6 as a vacuolar exporter of acidic amino acids in Saccharomyces cerevisiae cells. J Gen Appl Microbiol 55:409–417. https://doi.org/10.2323/jgam.55.409
Chen L, Bush DR (1997) LHT1, a lysine- and histidine-specific amino acid transporter in Arabidopsis. Plant Physiol 115:1127–1134. https://doi.org/10.1104/pp.115.3.1127
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743. https://doi.org/10.1046/J.1365-313X.1998.00343.X
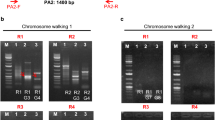
Dhatterwal P, Mehrotra S, Mehrotra R (2017) Optimization of PCR conditions for amplifying an AT-rich amino acid transporter promoter sequence with high number of tandem repeats from Arabidopsis thaliana. BMC Res Notes 10:638. https://doi.org/10.1186/s13104-017-2982-1
Dhatterwal P, Mehrotra S, Miller AJ, Mehrotra R (2021) Promoter profiling of Arabidopsis amino acid transporters: clues for improving crops. Plant Mol Biol 107:451–475. https://doi.org/10.1007/S11103-021-01193-1
Frankard V, Ghislain M, Jacobs M (1992) Two feedback-insensitive enzymes of the aspartate pathway in Nicotiana sylvestris. Plant Physiol 99:1285–1293. https://doi.org/10.1104/PP.99.4.1285
Freeman J, Sparks CA, West J, Shewry PR, Jones HD (2011) Temporal and spatial control of transgene expression using a heat-inducible promoter in transgenic wheat. Plant Biotechnol J 9:788–796. https://doi.org/10.1111/J.1467-7652.2011.00588.X
Frommer WB, Ninnemann O (1995) Heterologous expression of genes in bacterial, fungal, animal, and plant cells. Annu Rev Plant Physiol Plant Mol Biol 46:419–444. https://doi.org/10.1146/annurev.pp.46.060195.002223
Fujiki Y, Teshima H, Kashiwao S, Kawano-Kawada M, Ohsumi Y, Kakinuma Y, Sekito T (2017) Functional identification of AtAVT3, a family of vacuolar amino acid transporters, in Arabidopsis. FEBS Lett 591:5–15. https://doi.org/10.1002/1873-3468.12507
Galili G (2011) The aspartate-family pathway of plants: Linking production of essential amino acids with energy and stress regulation. Plant Signal Behav 6:192–195. https://doi.org/10.4161/psb.6.2.14425
Galili G, Amir R (2013) Fortifying plants with the essential amino acids lysine and methionine to improve nutritional quality. Plant Biotechnol J 11:211–222. https://doi.org/10.1111/pbi.12025
Galili G, Avin-Wittenberg T, Angelovici R, Fernie AR (2014) The role of photosynthesis and amino acid metabolism in the energy status during seed development. Front Plant Sci 5:447. https://doi.org/10.3389/fpls.2014.00447
Gietz RD, Woods RA (2002) Transformation of yeast by lithium acetate/single-stranded carrier DNA/polyethylene glycol method. Methods Enzymol 350:87–96. https://doi.org/10.1016/S0076-6879(02)50957-5
Grunennvaldt RL, Degenhardt-Goldbach J, Gerhardt IR, Quoirin M (2015) Promoters used in genetic transformation of plants. Res J Biol Sci 10:1–9. https://doi.org/10.3923/rjbsci.2015.1.9
Helm CV, De Francisco A, Gaziola SA, Fornazier RF, Pompeu GB, Azevedo RA (2004) Hull-less barley varieties: storage proteins and amino acid distribution in relation to nutritional quality. Food Biotechnol 18:327–341. https://doi.org/10.1081/FBT-200040531
Hildebrandt TM, Nunes Nesi A, Araújo WL, Braun HP (2015) Amino acid catabolism in plants. Mol Plant 8:1563–1579. https://doi.org/10.1016/j.molp.2015.09.005
Hirota T, Izumi M, Wada S, Makino A, Ishida H (2018) Vacuolar protein degradation via autophagy provides substrates to amino acid catabolic pathways as an adaptive response to sugar starvation in Arabidopsis thaliana. Plant Cell Physiol 59:1363–1376. https://doi.org/10.1093/pcp/pcy005
Huang A, Coutu C, Harrington M, Rozwadowski K, Hegedus DD (2021) Engineering a feedback inhibition-insensitive plant dihydrodipicolinate synthase to increase lysine content in Camelina sativa seeds. Transgenic Res. https://doi.org/10.1007/S11248-021-00291-6
Jefferson RA (1987) Assaying chimeric genes in plants: The GUS gene fusion system. Plant Mol Biol Report 5:387–405. https://doi.org/10.1007/BF02667740
Jia M, Wu H, Clay KL, Jung R, Larkins BA, Gibbon BC (2013) Identification and characterization of lysine-rich proteins and starch biosynthesis genes in the opaque2 mutant by transcriptional and proteomic analysis. BMC Plant Biol 13:60. https://doi.org/10.1186/1471-2229-13-60
Kirma M, Araújo WL, Fernie AR, Galili G (2012) The multifaceted role of aspartate-family amino acids in plant metabolism. J Exp Bot 63:4995–5001. https://doi.org/10.1093/jxb/ers119
Lee YH, Foster J, Chen J, Voll LM, Weber APM, Tegeder M (2007) AAP1 transports uncharged amino acids into roots of Arabidopsis. Plant J 50:305–319. https://doi.org/10.1111/J.1365-313X.2007.03045.X
Liang G, He H, Yu D (2012) Identification of nitrogen starvation-responsive microRNAs in Arabidopsis thaliana. PLoS ONE 7:e48951. https://doi.org/10.1371/journal.pone.0048951
Lim PO, Kim HJ, Gil Nam H (2007) Leaf senescence. Annu Rev Plant Biol 58:115–136. https://doi.org/10.1146/annurev.arplant.57.032905.105316
Liu JH, Peng T, Dai W (2014) Critical cis-acting elements and interacting transcription factors: key players associated with abiotic stress responses in plants. Plant Mol Biol Report 32:303–317. https://doi.org/10.1007/s11105-013-0667-z
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Locascio A, Andrés-Colás N, Mulet JM, Yenush L (2019) Saccharomyces cerevisiae as a tool to investigate plant potassium and sodium transporters. Int J Mol Sci 20:2133. https://doi.org/10.3390/ijms20092133
Lu K, Wu B, Wang J, Zhu W, Nie H, Qian J, Huang W, Fang Z (2018) Blocking amino acid transporter OsAAP3 improves grain yield by promoting outgrowth buds and increasing tiller number in rice. Plant Biotechnol J 16:1710–1722. https://doi.org/10.1111/pbi.12907
Mehrotra R, Mehrotra S (2010) Promoter activation by ACGT in response to salicylic and abscisic acids is differentially regulated by the spacing between two copies of the motif. J Plant Physiol 167:1214–1218. https://doi.org/10.1016/j.jplph.2010.04.005
Mehrotra R, Kiran K, Chaturvedi CP, Ansari SA, Lodhi N, Sawant S, Tuli R (2005) Effect of copy number and spacing of the ACGT and GT cis elements on transient expression of minimal promoter in plants. J Genet 84:183–187. https://doi.org/10.1007/BF02715844
Mehrotra R, Yadav A, Bhalothia P, Karan R, Mehrotra S (2012) Evidence for directed evolution of larger size motif in Arabidopsis thaliana genome. Sci World J. https://doi.org/10.1100/2012/983528
Mehrotra R, Sethi S, Zutshi I, Bhalothia P, Mehrotra S (2013) Patterns and evolution of ACGT repeat cis-element landscape across four plant genomes. BMC Genomics 14:203. https://doi.org/10.1186/1471-2164-14-203
Miller AJ, Zhou JJ (2000) Xenopus oocytes as an expression system for plant transporters. Biochim Biophys Acta Biomembr 1465:343–358. https://doi.org/10.1016/S0005-2736(00)00148-6
Potenza C, Aleman L, Sengupta-Gopalan C (2004) Targeting transgene expression in research, agricultural, and environmental applications: promoters used in plant transformation. Vitr Cell Dev Biol Plant 40:1–22. https://doi.org/10.1079/IVP2003477
Rentsch D, Hirner B, Schmelzer E, Frommer WB (1996) Salt stress-induced proline transporters and salt stress-repressed broad specificity amino acid permeases identified by suppression of a yeast amino acid permease-targeting mutant. Plant Cell 8:1437–1446. https://doi.org/10.1105/tpc.8.8.1437
Rushton PJ, Reinstädler A, Lipka V, Lippok B, Somssich IE (2002) Synthetic plant promoters containing defined regulatory elements provide novel insights into pathogen-and wound-induced signaling. Plant Cell 14:749–762. https://doi.org/10.1105/tpc.010412
Russnak R, Konczal D, McIntire SL (2001) A family of yeast proteins mediating bidirectional vacuolar amino acid transport. J Biol Chem 276:23849–23857. https://doi.org/10.1074/jbc.M008028200
Sah SK, Reddy KR, Li J (2016) Abscisic acid and abiotic stress tolerance in crop plants. Front Plant Sci 7:571. https://doi.org/10.3389/fpls.2016.00571
Schroeder JI, Delhaize E, Frommer WB, Lou GM, Harrison MJ, Herrera-Estrella L, Horie T, Kochian LV, Munns R, Nishizawa NK, Tsay YF, Sanders D (2013) Using membrane transporters to improve crops for sustainable food production. Nature 497:60–66. https://doi.org/10.1038/nature11909
Seifi HS, Van BJ, Angenon G, Höfte M (2013) Glutamate metabolism in plant disease and defense: friend or foe? MPMI 26:475–485. https://doi.org/10.1094/MPMI-07-12-0176-CR
Seki M, Ishida J, Narusaka M, Fujita M, Nanjo T, Umezawa T, Kamiya A, Nakajima M, Enju A, Sakurai T, Satou M (2002) Monitoring the expression pattern of around 7,000 Arabidopsis genes under ABA treatments using a full-length cDNA microarray. Funct Integr Genom 2:282–291. https://doi.org/10.1007/S10142-002-0070-6
Shaul O, Galili G (1993) Concerted regulation of lysine and threonine synthesis in tobacco plants expressing bacterial feedback-insensitive aspartate kinase and dihydrodipicolinate synthase. Plant Mol Biol 23:759–768. https://doi.org/10.1007/BF00021531
Shinozaki K, Yamaguchi-Shinozaki K (2003) Molecular mechanisms of plant responses and tolerance of drought and cold stress. Plant biotechnology 2002 and beyond. Springer, Dordrecht, pp 30–37
Wan J, Tanaka K, Zhang XC, Son GH, Brechenmacher L, Nguyen THN, Stacey G (2012) LYK4, a lysin motif receptor-like kinase, is important for chitin signaling and plant innate immunity in Arabidopsis. Plant Physiol 160:396–406. https://doi.org/10.1104/pp.112.201699
Wang W, Vinocur B, Altman A (2003) Plant responses to drought, salinity and extreme temperatures: towards genetic engineering for stress tolerance. Planta 218:1–14. https://doi.org/10.1007/s00425-003-1105-5
Wu S-J, Wang L-C, Yeh C-H, Lu C-A, Wu S-J (2010) Isolation and characterization of the Arabidopsis heat-intolerant 2 (hit2) mutant reveal the essential role of the nuclear export receptor EXPORTIN1A (XPO1A) in plant heat tolerance. New Phytol 186:833–842. https://doi.org/10.1111/J.1469-8137.2010.03225.X
Xia T, Xiao D, Liu D, Chai W, Gong Q, Wang NN (2012) Heterologous expression of ATG8c from soybean confers tolerance to nitrogen deficiency and increases yield in Arabidopsis. PLoS ONE 7:e37217. https://doi.org/10.1371/journal.pone.0037217
Yang H, Postel S, Kemmerling B, Ludewig U (2014) Altered growth and improved resistance of Arabidopsis against Pseudomonas syringae by overexpression of the basic amino acid transporter AtCAT1. Plant Cell Environ 37:1404–1414. https://doi.org/10.1111/pce.12244
Yang QQ, Zhao DS, Zhang CQ, Wu HY, Li QF, Gu MH, Sun SSM, Liu QQ (2018) A connection between lysine and serotonin metabolism in rice endosperm. Plant Physiol 176:1965–1980. https://doi.org/10.1104/PP.17.01283
Yang G, Wei Q, Huang H, Xia J (2020) Amino acid transporters in plant cells: a brief review. Plants 9:1–17. https://doi.org/10.3390/plants9080967
Yu S, Pratelli R, Denbow C, Pilot G (2015) Suppressor mutations in the Glutamine Dumper1 protein dissociate disturbance in amino acid transport from other characteristics of the Gdu1D phenotype. Front Plant Sci 6:593. https://doi.org/10.3389/FPLS.2015.00593/BIBTEX
Zhang R, Zhu J, Cao HZ, Xie XL, Huang JJ, Chen XH, Luo ZY (2013) Isolation and characterization of LHT-type plant amino acid transporter gene from Panax ginseng Meyer. J Ginseng Res 37:361–370. https://doi.org/10.5142/jgr.2013.37.361
Acknowledgements
The authors are grateful to BITS Pilani, K. K. Birla Goa Campus, Goa, India for providing infrastructural and logistic support. We thank Dr. Yi Chen for Xenopus oocyte experiment. PD is thankful to BITS Pilani, CSIR and EMBO for financial assistance. This study was supported by SERB Project EMR/2016/002470 sanctioned by the government of India to SM and RM. AJM was funded by the UK Biotechnology and Biological Sciences Research Council (BBSRC) Institute Strategic Program Grants ‘Molecules from Nature’ (BB/P012523/1) and ‘Plant Health’ (BB/P012574/1) and the John Innes Foundation.
Funding
The details of funding are mentioned in the Acknowledgements section as-<br>This study was supported by SERB Project EMR/2016/002470 sanctioned by the government of India to SM and RM. AJM was funded by the UK Biotechnology and Biological Sciences Research Council (BBSRC) Institute Strategic Program Grants ‘Molecules from Nature’ (BB/P012523/1) and ‘Plant Health’ (BB/P012574/1) and the John Innes Foundation.
Author information
Authors and Affiliations
Contributions
PD planned, designed, performed experiments, analyzed the data, and wrote the manuscript. AM supervised with the yeast and Xenopus studies. RA helped with the promoter studies. RM, and SM supervised the planning, designing of the experiments, writing of the manuscript and gave final approval for publication. All authors read and approved the manuscript. RM agrees to serve as the author responsible for contact and ensures communication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dhatterwal, P., Mehrotra, S., Miller, A.J. et al. Effect of ACGT motif in spatiotemporal regulation of AtAVT6D, which improves tolerance to osmotic stress and nitrogen-starvation. Plant Mol Biol 109, 67–82 (2022). https://doi.org/10.1007/s11103-022-01256-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-022-01256-x