Abstract
Purpose
To make a systematic review and meta-analysis of studies evaluating the effect of cabergoline (CBG) in the treatment of non-functioning pituitary adenomas (NFPAs).
Methods
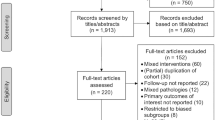
The primary outcome was tumor shrinkage, using as cut-off a reduction of at least 20% of the NFPA size from baseline. The secondary outcomes were prevention of tumor progression, clinically required additional interventions and adverse events (AE). Search strategies were applied to MEDLINE, EMBASE, LILACS and CENTRAL. Independent reviewers assessed the study eligibility, extracted data, and evaluated risk of bias. Random meta-analysis for the proportion of tumor shrinkage, prevention of tumor progression, clinically required additional interventions and frequency of AE were conducted.
Results
Five studies were included. The meta-analysis of proportion was 19% for tumor shrinkage (95% CI 8–38%, 4 studies, 108 participants), 50% for prevention of tumor progression (95% CI 35–64%, 5 studies, 187 participants), 14% for clinically required additional interventions (95% CI 6–30%, 4 studies, 128 participants) and 2% for adverse events (95% CI 1–6%, 3 studies, 157 participants).
Conclusions
Effect of CBG to promote tumor shrinkage in NFPAs was low, while prevention of tumor progression after surgery was seen in half of the cases, with a low frequency of adverse events.
Systematic review registration
PROSPERO CRD42020206778.


Similar content being viewed by others
References
Molitch ME (2008) Nonfunctioning pituitary tumors and pituitary incidentalomas. Endocrinol Metab Clin North Am 37(1):151–171
Mercado M, Melgar V, Salame L, Cuenca D (2017) Clinically non-functioning pituitary adenomas: Pathogenic, diagnostic and therapeutic aspects. Endocrinologia, Diabetes y Nutricion 64(7):384–395
AlMalki MH, Ahmad MM, Brema I, AlDahmani KM, Pervez N, Al-Dandan S et al (2020) Contemporary management of clinically non-functioning pituitary adenomas: a clinical review. Clin Med Insights 13:1–13
Farrell CJ, Garzon-Muvdi T, Fastenberg JH, Nyquist GG, Rabinowitz MR, Rosen MR et al (2019) Management of nonfunctioning recurrent pituitary adenomas. Neurosurg Clin N Am 30(4):473–482
Chanson P, Dormoy A, Dekkers OM (2019) Use of radiotherapy after pituitary surgery for non-functioning pituitary adenomas. Eur J Endocrinol 181(1):D1–D13
Tampourlou M, Karapanou O, Vassiliadi DA, Tsagarakis S (2019) Medical therapy for non-functioning pituitary tumors-a critical approach. Hormones 18(2):117–126
Pivonello R, Matrone C, Filippella M, Cavallo LM, Di Somma C, Cappabianca P et al (2004) Dopamine receptor expression and function in clinically nonfunctioning pituitary tumors: comparison with the effectiveness of cabergoline treatment. J Clin Endocrinol Metab 89(4):1674–1683
Lin S, Zhang A, Zhang X, Wu ZB (2020) Treatment of pituitary and other tumours with cabergoline: new mechanisms and potential broader applications. Neuroendocrinology 110(6):477–488
Auriemma RS, Grasso LF, Pivonello R, Colao A (2016) The safety of treatments for prolactinomas. Expert Opin Drug Saf 15(4):503–512
Greenman Y, Bronstein MD (2021) Cabergoline should be attempted in progressing non-functioning pituitary macroadenoma. Eur J Endocrinol 185(4):D11-d20
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6(7):e1000097
Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A (2016) Rayyan-a web and mobile app for systematic reviews. Syst Rev 5(1):210
Munn Z, Barker TH, Moola S, Tufanaru C, Stern C, McArthur A et al (2020) Methodological quality of case series studies: an introduction to the JBI critical appraisal tool. JBI Evid Synth 18(10):2127–2133
Nyaga VN, Arbyn M, Aerts M (2014) Metaprop: a Stata command to perform meta-analysis of binomial data. Arch Public Health 72(1):39
National Library of Medicine (U.S.). (2014). Dopamine agonist treatment of non-functioning pituitary adenomas. Identifier NCT02288962. https://clinicaltrials.gov/show/NCT02288962 (2014). Accessed 20 Nov 2020
Greenman Y, Tordjman K, Osher E, Veshchev I, Shenkerman G, Reider G II et al (2005) Postoperative treatment of clinically nonfunctioning pituitary adenomas with dopamine agonists decreases tumour remnant growth. Clin Endocrinol 63(1):39–44
Vieira Neto L, Wildemberg LE, Moraes AB, Colli LM, Kasuki L, Marques NV et al (2015) Dopamine receptor subtype 2 expression profile in nonfunctioning pituitary adenomas and in vivo response to cabergoline therapy. Clin Endocrinol 82(5):739–746
de Herder WW, Reijs AE, Feelders RA, van Aken MO, Krenning EP, Tanghe HL et al (2006) Dopamine agonist therapy of clinically non-functioning pituitary macroadenomas. Is there a role for 123I-epidepride dopamine D2 receptor imaging? Eur J Endocrinol 155(5):717–23
Robenshtok E, Benbassat CA, Hirsch D, Tzvetov G, Cohen ZR, Iraqi HM et al (2014) Clinical course and outcome of nonfunctioning pituitary adenomas in the elderly compared with younger age groups. Endocr Pract 20(2):159–164
Gsponer J, De Tribolet N, Déruaz JP, Janzer R, Uské A, Mirimanoff RO et al (1999) Diagnosis, treatment, and outcome of pituitary tumors and other abnormal intrasellar masses. Retrospective analysis of 353 patients. Medicine 78(4):236–69
Gasser RW, Mueller-Holzner E, Skrabal F, Finkenstedt G, Mayr U, Tabarelli M et al (1987) Macroprolactinomas and functionless pituitary tumours. Immunostaining and effect of dopamine agonist therapy. Acta Endocrinol 116(2):253–9
Colao A, Ferone D, Lastoria S, Cerbone G, Di Sarno A, Di Somma C et al (2000) Hormone levels and tumour size response to quinagolide and cabergoline in patients with prolactin-secreting and clinically non-functioning pituitary adenomas: predictive value of pituitary scintigraphy with 123I-methoxybenzamide. Clin Endocrinol 52(4):437–445
Batista RL, Musolino NRC, Cescato VAS, da Silva GO, Medeiros RSS, Herkenhoff CGB et al (2019) Cabergoline in the management of residual nonfunctioning pituitary adenoma: a single-center, open-label, 2-year randomized clinical trial. Am J Clin Oncol 42(2):221–227
Greenman Y, Cooper O, Yaish I, Robenshtok E, Sagiv N, Jonas-Kimchi T et al (2016) Treatment of clinically nonfunctioning pituitary adenomas with dopamine agonists. Eur J Endocrinol 175(1):63–72
Garcia EC, Naves LA, Silva AO, De Castro LF, Casulari LA, Azevedo MF (2013) Short-term treatment with cabergoline can lead to tumor shrinkage in patients with nonfunctioning pituitary adenomas. Pituitary 16(2):189–194
Lohmann T, Trantakis C, Biesold M, Prothmann S, Guenzel S, Schober R et al (2001) Minor tumour shrinkage in nonfunctioning pituitary adenomas by long-term treatment with the dopamine agonist cabergoline. Pituitary 4(3):173–178
Vargas G, Gonzalez B, Ramirez C, Ferreira A, Espinosa E, Mendoza V et al (2015) Clinical characteristics and treatment outcome of 485 patients with nonfunctioning pituitary macroadenomas. Int J Endocrinol
Greenman Y (2017) Management of endocrine disease: Present and future perspectives for medical therapy of nonfunctioning pituitary adenomas. Eur J Endocrinol 177(3):R113–R124
Levy MJ, Robertson IJ, Khalk N, Vitello S, Reddy N, Bhake R et al (2018) Long-term follow-up of a large prospective cohort of patients with nonfunctioning pituitary adenomas: the outcome of a conservative management policy. Clin Endocrinol 89(3):354–359
Tampourlou M, Ntali G, Ahmed S, Arlt W, Ayuk J, Byrne JV et al (2017) Outcome of nonfunctioning pituitary adenomas that regrow after primary treatment: a study from two large UK centers. J Clin Endocrinol Metab 102(6):1889–1897
Chen Y, Wang CD, Su ZP, Chen YX, Cai L, Zhuge QC et al (2012) Natural history of postoperative nonfunctioning pituitary adenomas: a systematic review and meta-analysis. Neuroendocrinology 96(4):333–342
Dekkers OM, Pereira AM, Romijn JA (2008) Treatment and follow-up of clinically nonfunctioning pituitary macroadenomas. J Clin Endocrinol Metab 93(10):3717–3726
Minniti G, Flickinger J, Tolu B, Paolini S (2018) Management of nonfunctioning pituitary tumors: radiotherapy. Pituitary 21(2):154–161
Tsang RW, Laperriere NJ, Simpson WJ, Brierley J, Panzarella T, Smyth HS (1993) Glioma arising after radiation therapy for pituitary adenoma. A report of four patients and estimation of risk. Cancer 72(7):2227–33
Minniti G, Traish D, Ashley S, Gonsalves A, Brada M (2005) Risk of second brain tumor after conservative surgery and radiotherapy for pituitary adenoma: update after an additional 10 years. J Clin Endocrinol Metab 90(2):800–804
Bülow B, Hagmar L, Mikoczy Z, Nordström CH, Erfurth EM (1997) Increased cerebrovascular mortality in patients with hypopituitarism. Clin Endocrinol 46(1):75–81
Brada M, Ashley S, Ford D, Traish D, Burchell L, Rajan B (2002) Cerebrovascular mortality in patients with pituitary adenoma. Clin Endocrinol 57(6):713–717
de Bruin TW, Kwekkeboom DJ, Van’t Verlaat JW, Reubi JC, Krenning EP, Lamberts SW et al (1992) Clinically nonfunctioning pituitary adenoma and octreotide response to long term high dose treatment, and studies in vitro. J Clin Endocrinol Metab 75(5):1310–1317
Colao A, Filippella M, Di Somma C, Manzi S, Rota F, Pivonello R et al (2003) Somatostatin analogs in treatment of non-growth hormone-secreting pituitary adenomas. Endocrine 20(3):279–283
Fusco A, Giampietro A, Bianchi A, Cimino V, Lugli F, Piacentini S et al (2012) Treatment with octreotide LAR in clinically non-functioning pituitary adenoma: results from a case-control study. Pituitary 15(4):571–578
Halevy C, Whitelaw BC (2017) How effective is temozolomide for treating pituitary tumours and when should it be used? Pituitary 20(2):261–266
Colao A, Di Somma C, Pivonello R, Faggiano A, Lombardi G, Savastano S (2008) Medical therapy for clinically non-functioning pituitary adenomas. Endocr Relat Cancer 15(4):905–915
Stiles CE, Tetteh-Wayoe ET, Bestwick J, Steeds RP, Drake WM (2018) A meta-analysis of the prevalence of cardiac valvulopathy in hyperprolactinemic patients treated with Cabergoline. J Clin Endocrinol Metab. https://doi.org/10.1210/jc.2018-01071
Ilie MD, Raverot G (2020) Treatment options for gonadotroph tumors: current state and perspectives. J Clin Endocrinol Metab 105(10):e3507–e3518
Sato M, Tamura R, Tamura H et al (2019) Analysis of tumor angiogenesis and immune microenvironment in non-functional pituitary endocrine tumors. J Clin Med 8(5):695
Marques P, Barry S, Carlsen E et al (2019) Chemokines modulate the tumour microenvironment in pituitary neuroendocrine tumours. Acta Neuropathol Commun 7(1):172
Fachi MM, de Deus Bueno L, de Oliveira DC, da Silva LL, Bonetti AF (2021) Efficacy and safety in the treatment of hyperprolactinemia: a systematic review and network meta-analysis. J Clin Pharm Ther 46(6):1549–56
Budayr A, Tan TC, Lo JC, Zaroff JG, Tabada GH, Yang J et al (2020) Cardiac valvular abnormalities associated with use and cumulative exposure of cabergoline for hyperprolactinemia: the CATCH study. BMC Endocr Disord 20(1):25
Boguszewski CL, dos Santos CM, Sakamoto KS, Marini LC, de Souza AM, Azevedo M (2012) A comparison of cabergoline and bromocriptine on the risk of valvular heart disease in patients with prolactinomas. Pituitary 15(1):44–49
Stiles CE, Lloyd G, Bhattacharyya S, Steeds RP, Boomla K, Bestwick JP et al (2021) Incidence of cabergoline-associated valvulopathy in primary care patients with prolactinoma using hard cardiac endpoints. J Clin Endocrinol Metab 106(2):e711–e720
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
CLB and VSNN conceptualized and designed the study. VSNN and MSB developed the search strategies. MSB and IAF independently screened eligible studies and extracted data from included studies. MSB and IAF assessed in pairs and independently the risk of bias. VSNN and CLB performed the meta-analyses. CLB and VSNN supervised all the phases of this review and refereed any disagreement to avoid errors. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Competing interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Botelho, M.S., Franzini, Í.A., Nunes-Nogueira, V. et al. Treatment of non-functioning pituitary adenoma with cabergoline: a systematic review and meta-analysis. Pituitary 25, 810–818 (2022). https://doi.org/10.1007/s11102-022-01257-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-022-01257-5