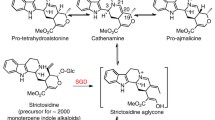
Abstract
The alkaloids are a major class of nitrogen-containing plant specialized metabolites that function as both toxic defense compounds and useful pharmaceuticals. Understanding the biosynthesis of these molecules offers a glimpse into the impressive diversity of metabolic chemistry found within plants and also enables the metabolic engineering of valuable alkaloids in other biological systems. Within the last decade, there have been major advances in our knowledge of alkaloid metabolism within multiple plant lineages, and this has allowed for the complete, or near complete elucidation of several alkaloid biosynthetic pathways. This work has been instrumental for establishing the chemical logic by which alkaloids are synthesized in plants and has also highlighted enzymatic functions that were not previously recognized or identified within plant metabolism. In this review, we highlight notable examples of unexpected or otherwise noteworthy enzyme-catalyzed reactions that have recently been identified within alkaloid biosynthesis. Our goal is to emphasize two major areas: (1) non-canonical reactions catalyzed by enzyme families with otherwise well-known roles in alkaloid biosynthesis, and (2) enzyme families that have emerged as having a broader role in alkaloid metabolism than previously appreciated. Beyond compiling these examples, we also discuss mechanistic details of novel enzyme classes that may be useful for future identification and study of non-canonical enzyme activity in alkaloid biosynthesis. Collectively, the recent discovery of several previously unidentified enzyme activities within the production of alkaloids indicates that there is still much to uncover about plant specialized metabolism. Thus, the continued study of alkaloids will not only provide us with the underlying enzymes necessary to engineer production of valuable molecules with medicinal potential, but will also yield foundational insight on the chemistry that is possible in the plant kingdom.







Similar content being viewed by others
References
Agarwal V, Miles ZD, Winter JM et al (2017) Enzymatic halogenation and dehalogenation reactions: pervasive and mechanistically diverse. Chem Rev 117:5619–5674. https://doi.org/10.1021/acs.chemrev.6b00571
Anarat-Cappillino G, Sattely ES (2014) The chemical logic of plant natural product biosynthesis. Curr Opin Plant Biol 19:51–58. https://doi.org/10.1016/j.pbi.2014.03.007
Araji S, Grammer TA, Gertzen R et al (2014) Novel roles for the polyphenol oxidase enzyme in secondary metabolism and the regulation of cell death in walnut. Plant Physiol 164:1191–1203. https://doi.org/10.1104/pp.113.228593
Austin MB, Noel JP (2003) The chalcone synthase superfamily of type III polyketide synthases. Nat Prod Rep 20:79–110. https://doi.org/10.1039/b100917f
Battersby AR (1996) The marvels of biosynthesis: tracking nature’s pathways. Bioorg Med Chem 4:937–964. https://doi.org/10.1016/0968-0896(96)00102-2
Bedewitz MA, Jones AD, D’Auria JC, Barry CS (2018) Tropinone synthesis via an atypical polyketide synthase and P450-mediated cyclization. Nat Commun 9:5281. https://doi.org/10.1038/s41467-018-07671-3
Bhattacharyya B, Panda D, Gupta S, Banerjee M (2008) Anti-mitotic activity of colchicine and the structural basis for its interaction with tubulin. Med Res Rev 28:155–183. https://doi.org/10.1002/med.20097
Bontpart T, Cheynier V, Ageorges A, Terrier N (2015) BAHD or SCPL acyltransferase? What a dilemma for acylation in the world of plant phenolic compounds. New Phytol 208:695–707. https://doi.org/10.1111/nph.13498
Bulling S, Schicker K, Zhang YW et al (2012) The mechanistic basis for noncompetitive ibogaine inhibition of serotonin and dopamine transporters. J Biol Chem 287:18524–18534. https://doi.org/10.1074/jbc.M112.343681
Bunsupa S, Katayama K, Ikeura E et al (2012) Lysine decarboxylase catalyzes the first step of quinolizidine alkaloid biosynthesis and coevolved with alkaloid production in Leguminosae. Plant Cell 24:1202–1216. https://doi.org/10.1105/tpc.112.095885
Bunsupa S, Hanada K, Maruyama A et al (2016) Molecular evolution and functional characterization of a bifunctional decarboxylase involved in Lycopodium alkaloid biosynthesis. Plant Physiol 171:2432–2444. https://doi.org/10.1104/pp.16.00639
Bunsupa S, Yamazaki M, Saito K (2017) Lysine-derived alkaloids: overview and update on biosynthesis and medicinal applications with emphasis on quinolizidine alkaloids. Mini-Rev Med Chem 17:1002–1012. https://doi.org/10.2174/1389557516666160506151213
Caputi L, Franke J, Farrow SC et al (2018) Missing enzymes in the biosynthesis of the anticancer drug vinblastine in Madagascar periwinkle. Science 360:1235–1239. https://doi.org/10.1126/science.aat4100
Caputi L, Franke J, Bussey K et al (2020) Structural basis of cycloaddition in biosynthesis of iboga and aspidosperma alkaloids. Nat Chem Biol 16:383–386. https://doi.org/10.1038/s41589-019-0460-x
Chang WC, Guo Y, Wang C et al (2014) Mechanism of the C5 stereoinversion reaction in the biosynthesis of carbapenem antibiotics. Science 343:1140–1144. https://doi.org/10.1126/science.1248000
Chavez BG, Srinivasan P, Glockzin K et al (2022) Elucidation of tropane alkaloid biosynthesis in Erythroxylum coca using a microbial pathway discovery platform. Proc Natl Acad Sci USA 119:e2215372119. https://doi.org/10.1073/pnas.2215372119
Chen X, Hagel JM, Chang L et al (2018a) A pathogenesis-related 10 protein catalyzes the final step in thebaine biosynthesis. Nat Chem Biol 14:738–743. https://doi.org/10.1038/s41589-018-0059-7
Chen YC, Holmes EC, Rajniak J et al (2018b) N-Hydroxy-pipecolic acid is a mobile metabolite that induces systemic disease resistance in Arabidopsis. Proc Natl Acad Sci USA 115:E4920–E4929. https://doi.org/10.1073/pnas.1805291115
Cona A, Rea G, Angelini R et al (2006) Functions of amine oxidases in plant development and defence. Trends Plant Sci 11:80–88. https://doi.org/10.1016/j.tplants.2005.12.009
Courdavault V, O’Connor SE, Jensen MK, Papon N (2021) Metabolic engineering for plant natural products biosynthesis: New procedures, concrete achievements and remaining limits. Nat Prod Rep 38:2145–2153. https://doi.org/10.1039/d0np00092b
D’Auria JC (2006) Acyltransferases in plants: a good time to be BAHD. Curr Opin Plant Biol 9:331–340. https://doi.org/10.1016/j.pbi.2006.03.016
Dastmalchi M, Chen X, Hagel JM et al (2019) Neopinone isomerase is involved in codeine and morphine biosynthesis in opium poppy. Nat Chem Biol 15:384–390. https://doi.org/10.1038/s41589-019-0247-0
De La Peña R, Hodgson H, Liu JC-T et al (2023) Complex scaffold remodeling in plant triterpene biosynthesis. Science 379:361–368. https://doi.org/10.1126/science.adf1017
Dewick PM (2009) Alkaloids. Medicinal natural products medicinal natural products: a biosynthetic approach, 3rd edn. Wiley, Chichester, pp 311–417
Dudley QM, Jo S, Guerrero DAS et al (2022) Reconstitution of monoterpene indole alkaloid biosynthesis in genome engineered Nicotiana benthamiana. Commun Biol 5:949. https://doi.org/10.1038/s42003-022-03904-w
Dührkop K, Fleischauer M, Ludwig M et al (2019) SIRIUS 4: a rapid tool for turning tandem mass spectra into metabolite structure information. Nat Methods 16:299–302. https://doi.org/10.1038/s41592-019-0344-8
Eggers R, Jammer A, Jha S et al (2021) The scope of flavin-dependent reactions and processes in the model plant Arabidopsis thaliana. Phytochemistry 189:112822. https://doi.org/10.1016/j.phytochem.2021.112822
Eliot AC, Kirsch JF (2004) Pyridoxal phosphate enzymes: mechanistic, structural, and evolutionary considerations. Annu Rev Biochem 73:383–415. https://doi.org/10.1146/annurev.biochem.73.011303.074021
Eng JGM, Shahsavarani M, Smith DP et al (2022) A Catharanthus roseus Fe(II)/α-ketoglutarate-dependent dioxygenase catalyzes a redox-neutral reaction responsible for vindolinine biosynthesis. Nat Commun 13:3335. https://doi.org/10.1038/s41467-022-31100-1
Facchini PJ, Morris JS (2019) Molecular origins of functional diversity in benzylisoquinoline alkaloid methyltransferases. Front Plant Sci 10:1058. https://doi.org/10.3389/fpls.2019.01058
Fallon TR, Čalounová T, Mokrejš M et al (2023) transXpress: a Snakemake pipeline for streamlined de novo transcriptome assembly and annotation. BMC Bioinform 24:133. https://doi.org/10.1186/s12859-023-05254-8
Farrow SC, Facchini PJ (2014) Functional diversity of 2-oxoglutarate/Fe(II)-dependent dioxygenases in plant metabolism. Front Plant Sci 5:524. https://doi.org/10.3389/fpls.2014.00524
Farrow SC, Hagel JM, Beaudoin GAW et al (2015) Stereochemical inversion of (S)-reticuline by a cytochrome P450 fusion in opium poppy. Nat Chem Biol 11:728–732. https://doi.org/10.1038/nchembio.1879
Farrow SC, Kamileen MO, Caputi L et al (2019) Biosynthesis of an anti-addiction agent from the iboga plant. J Am Chem Soc 141:12979–12983. https://doi.org/10.1021/jacs.9b05999
Finefield JM, Sherman DH, Kreitman M, Williams RM (2012) Enantiomeric natural products: occurrence and biogenesis. Angew Chem Int Ed 51:4802–4836. https://doi.org/10.1002/anie.201107204
Florean M, Luck K, Hong B et al (2023) Reinventing metabolic pathways: independent evolution of benzoxazinoids in flowering plants. Proc Natl Acad Sci USA 120:e2307981120. https://doi.org/10.1073/pnas.2307981120
Galanie S, Thodey K, Trenchard IJ et al (2015) Complete biosynthesis of opioids in yeast. Science 349:1095–1100. https://doi.org/10.1126/science.aac9373
Gao L, Su C, Du X et al (2020) FAD-dependent enzyme-catalysed intermolecular [4+2] cycloaddition in natural product biosynthesis. Nat Chem 12:620–628. https://doi.org/10.1038/s41557-020-0467-7
Gollwitzer J, Lenz R, Hampp N, Zenk MH (1993) The transformation of neopinone to codeinone in morphine biosynthesis proceeds non-enzymatically. Tetrahedron Lett 34:5703–5706. https://doi.org/10.1016/S0040-4039(00)73838-X
Gribble GW (2003) The diversity of naturally produced organohalogens. Chemosphere 52:289–297. https://doi.org/10.1016/S0045-6535(03)00207-8
Haas BJ, Papanicolaou A, Yassour M et al (2013) De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat Protoc 8:1494–1512. https://doi.org/10.1038/nprot.2013.084
Hagel JM, Facchini PJ (2018) Expanding the roles for 2-oxoglutarate-dependent oxygenases in plant metabolism. Nat Prod Rep 35:721–734. https://doi.org/10.1039/c7np00060j
Hansen CC, Nelson DR, Møller BL, Werck-Reichhart D (2021) Plant cytochrome P450 plasticity and evolution. Mol Plant 14:1244–1265. https://doi.org/10.1016/j.molp.2021.06.028
Hartmann M, Zeier T, Bernsdorff F et al (2018) Flavin monooxygenase-generated N-hydroxypipecolic acid is a critical element of plant systemic immunity. Cell 173:456-469.e16. https://doi.org/10.1016/j.cell.2018.02.049
Herbert RB, Kattah AE, Knagg E (1990) The biosynthesis of the phenethylisoquinoline alkaloid colchicine. Early and intermediate stages. Tetrahedron 46:7119–7138. https://doi.org/10.1016/S0040-4020(01)87895-9
Hong B, Grzech D, Caputi L et al (2022) Biosynthesis of strychnine. Nature 607:617–622. https://doi.org/10.1038/s41586-022-04950-4
Huang J-P, Fang C, Ma X et al (2019) Tropane alkaloids biosynthesis involves an unusual type III polyketide synthase and non-enzymatic condensation. Nat Commun 10:4036. https://doi.org/10.1038/s41467-019-11987-z
Huang JP, Wang YJ, Tian T et al (2021) Tropane alkaloid biosynthesis: a centennial review. Nat Prod Rep 38:1634–1658. https://doi.org/10.1039/d0np00076k
Jacobowitz JR, Weng JK (2020) Exploring uncharted territories of plant specialized metabolism in the postgenomic era. Annu Rev Plant Biol 71:631–658. https://doi.org/10.1146/annurev-arplant-081519-035634
Kamileen MO, Demars MD, Hong B et al (2022) Recycling upstream redox enzymes expands the regioselectivity of cycloaddition in pseudo-aspidosperma alkaloid biosynthesis. J Am Chem Soc 144:19673–19679. https://doi.org/10.1021/jacs.2c08107
Katoh A, Shoji T, Hashimoto T (2007) Molecular cloning of N-methylputrescine oxidase from tobacco. Plant Cell Physiol 48:550–554. https://doi.org/10.1093/pcp/pcm018
Kawai Y, Ono E, Mizutani M (2014) Evolution and diversity of the 2-oxoglutarate-dependent dioxygenase superfamily in plants. Plant J 78:328–343. https://doi.org/10.1111/tpj.12479
Kim CY, Mitchell AJ, Glinkerman CM et al (2020) The chloroalkaloid (−)-acutumine is biosynthesized via a Fe(II)- and 2-oxoglutarate-dependent halogenase in Menispermaceae plants. Nat Commun 11:1867. https://doi.org/10.1038/s41467-020-15777-w
Kobayashi J, Kubota T (2009) The Daphniphyllum alkaloids. Nat Prod Rep 26:936–962
Kulma A, Szopa J (2007) Catecholamines are active compounds in plants. Plant Sci 172:433–440. https://doi.org/10.1016/j.plantsci.2006.10.013
Kwan BD, Seligmann B, Nguyen TD et al (2023) Leveraging synthetic biology and metabolic engineering to overcome obstacles in plant pathway elucidation. Curr Opin Plant Biol 71:102330. https://doi.org/10.1016/j.pbi.2022.102330
Lau W, Sattely ES (2015) Six enzymes from mayapple that complete the biosynthetic pathway to the etoposide aglycone. Science 349:1224–1228. https://doi.org/10.1126/science.aac7202
Lenz R, Zenk MH (1995) Acetyl coenzyme A:salutaridinol-7-O-acetyltransferase from Papaver somniferum plant cell cultures. J Biol Chem 270:31091–31096. https://doi.org/10.1074/jbc.270.52.31091
Lichman BR (2021) The scaffold-forming steps of plant alkaloid biosynthesis. Nat Prod Rep 38:103–129. https://doi.org/10.1039/d0np00031k
Lichman BR, Godden GT, Hamilton JP et al (2020) The evolutionary origins of the cat attractant nepetalactone in catnip. Sci Adv 6:eaba0721. https://doi.org/10.1126/sciadv.aba0721
Liu X, Zhu X, Wang H et al (2020) Discovery and modification of cytochrome P450 for plant natural products biosynthesis. Synth Syst Biotechnol 5:187–199. https://doi.org/10.1016/j.synbio.2020.06.008
Ma X, Gang DR (2004) The Lycopodium alkaloids. Nat Prod Rep 21:752–772. https://doi.org/10.1039/b409720n
Majhi BB, Gélinas SE, Mérindol N et al (2023) Characterization of norbelladine synthase and noroxomaritidine/norcraugsodine reductase reveals a novel catalytic route for the biosynthesis of Amaryllidaceae alkaloids including the Alzheimer’s drug galanthamine. Front Plant Sci 14:1231809. https://doi.org/10.3389/fpls.2023.1231809
Mancinotti D, Frick KM, Geu-Flores F (2022) Biosynthesis of quinolizidine alkaloids in lupins: mechanistic considerations and prospects for pathway elucidation. Nat Prod Rep 39:1423–1437. https://doi.org/10.1039/d1np00069a
Matsuda Y, Wakimoto T, Mori T et al (2014) Complete biosynthetic pathway of anditomin: nature’s sophisticated synthetic route to a complex fungal meroterpenoid. J Am Chem Soc 136:15326–15336. https://doi.org/10.1021/ja508127q
McGaw BA, Woolley JG (1978) Stereochemistry of tropane alkaloid formation in Datura. Phytochemistry 17:257–259. https://doi.org/10.1016/S0031-9422(00)94158-X
Menéndez-Perdomo IM, Facchini PJ (2023) Elucidation of the (R)-enantiospecific benzylisoquinoline alkaloid biosynthetic pathways in sacred lotus (Nelumbo nucifera). Sci Rep 13:2955. https://doi.org/10.1038/s41598-023-29415-0
Mindrebo JT, Nartey CM, Seto Y et al (2016) Unveiling the functional diversity of the alpha/beta hydrolase superfamily in the plant kingdom. Curr Opin Struct Biol 41:256–257. https://doi.org/10.1016/j.sbi.2016.08.005
Mizutani M, Sato F (2011) Unusual P450 reactions in plant secondary metabolism. Arch Biochem Biophys 507:194–203. https://doi.org/10.1016/j.abb.2010.09.026
Morris JS, Caldo KMP, Liang S, Facchini PJ (2021) PR10/Bet v1-like proteins as novel contributors to plant biochemical diversity. ChemBioChem 22:264–287. https://doi.org/10.1002/cbic.202000354
Munro AW, Girvan HM, McLean KJ (2007) Cytochrome P450-redox partner fusion enzymes. Biochim Biophys Acta 1770:345–359. https://doi.org/10.1016/j.bbagen.2006.08.018
Nardini M, Dijkstra BW (1999) α/β Hydrolase fold enzymes: the family keeps growing. Curr Opin Struct Biol 9:732–737. https://doi.org/10.1016/S0959-440X(99)00037-8
Nelson D, Werck-Reichhart D (2011) A P450-centric view of plant evolution. Plant J 66:194–211. https://doi.org/10.1111/j.1365-313X.2011.04529.x
Nett RS, Sattely ES (2021) Total biosynthesis of the tubulin-binding alkaloid colchicine. J Am Chem Soc 143:19454–19465. https://doi.org/10.1021/jacs.1c08659
Nett RS, Lau W, Sattely ES (2020a) Discovery and engineering of colchicine alkaloid biosynthesis. Nature 584:148–153. https://doi.org/10.1038/s41586-020-2546-8
Nett RS, Nguyen H, Nagel R et al (2020b) Unraveling a tangled skein: evolutionary analysis of the bacterial gibberellin biosynthetic operon. mSphere 5:e00292-20. https://doi.org/10.1128/msphere.00292-20
Nett RS, Dho Y, Low Y-Y, Sattely ES (2021) A metabolic regulon reveals early and late acting enzymes in neuroactive Lycopodium alkaloid biosynthesis. Proc Natl Acad Sci 118:e2102949118. https://doi.org/10.1073/pnas.2102949118
Nettleship L, Slaytor M (1974) Limitations of feeding experiments in studying alkaloid biosynthesis in Peganum harmala callus cultures. Phytochemistry 13:735–742. https://doi.org/10.1016/S0031-9422(00)91406-7
Nguyen TD, Dang TTT (2021) Cytochrome P450 enzymes as key drivers of alkaloid chemical diversification in plants. Front Plant Sci 12:682181. https://doi.org/10.3389/fpls.2021.682181
Nguyen TAM, Grzech D, Chung K et al (2023) Discovery of a cytochrome P450 enzyme catalyzing the formation of spirooxindole alkaloid scaffold. Front Plant Sci 14:1125158. https://doi.org/10.3389/fpls.2023.1125158
O’Connor SE (2015) Engineering of secondary metabolism. Annu Rev Genet 49:71–94. https://doi.org/10.1146/annurev-genet-120213-092053
O’Connor SE, Maresh JJ (2006) Chemistry and biology of monoterpene indole alkaloid biosynthesis. Nat Prod Rep 23:532–547. https://doi.org/10.1039/b512615k
Ortiz de Montellano PR (2015) Substrate Oxidation by Cytochrome P450 Enzymes. In: Ortiz De Montellano PR (ed) Cytochrome P450: structure, mechanism, and biochemistry, Fourth Edi. Springer International Publishing, Switzerland, pp 111–176
Ozber N, Carr SC, Morris JS et al (2022) Alkaloid binding to opium poppy major latex proteins triggers structural modification and functional aggregation. Nat Commun 13:6768. https://doi.org/10.1038/s41467-022-34313-6
Panis F, Kampatsikas I, Bijelic A, Rompel A (2020) Conversion of walnut tyrosinase into a catechol oxidase by site directed mutagenesis. Sci Rep 10:1659. https://doi.org/10.1038/s41598-020-57671-x
Paul CE, Eggerichs D, Westphal AH et al (2021) Flavoprotein monooxygenases: versatile biocatalysts. Biotechnol Adv 51:107712. https://doi.org/10.1016/j.biotechadv.2021.107712
Qiao F, He Y, Zhang Y et al (2023) Elucidation of the 1-phenethylisoquinoline pathway from an endemic conifer Cephalotaxus hainanensis. Proc Natl Acad Sci USA 120:e2209339120. https://doi.org/10.1073/pnas.2209339120
Qu Y, Easson MEAM, Simionescu R et al (2018) Solution of the multistep pathway for assembly of corynanthean, strychnos, iboga, and aspidosperma monoterpenoid indole alkaloids from 19E-geissoschizine. Proc Natl Acad Sci 115:3180–3185. https://doi.org/10.1073/pnas.1719979115
Reed J, Orme A, El-Demerdash A et al (2023) Elucidation of the pathway for biosynthesis of saponin adjuvants from the soapbark tree. Science 379:1252–1264. https://doi.org/10.1126/science.adf3727
Samanani N, Facchini PJ (2002) Purification and characterization of norcoclaurine synthase: the first committed enzyme in benzylisoquinoline alkaloid biosynthesis in plants. J Biol Chem 277:33878–33883. https://doi.org/10.1074/jbc.M203051200
Samanani N, Liscombe DK, Facchini PJ (2004) Molecular cloning and characterization of norcoclaurine synthase, an catalyzing the first committed step in benzylisoquinoline alkaloid biosynthesis. Plant J 40:302–314. https://doi.org/10.1111/j.1365-313X.2004.02210.x
Schenck CA, Maeda HA (2018) Tyrosine biosynthesis, metabolism, and catabolism in plants. Phytochemistry 149:82–102. https://doi.org/10.1016/j.phytochem.2018.02.003
Singh A, Massicotte MA, Garand A et al (2018) Cloning and characterization of norbelladine synthase catalyzing the first committed reaction in Amaryllidaceae alkaloid biosynthesis. BMC Plant Biol 18:338. https://doi.org/10.1186/s12870-018-1570-4
Srinivasan P, Smolke CD (2020) Biosynthesis of medicinal tropane alkaloids in yeast. Nature 585:614–619. https://doi.org/10.1038/s41586-020-2650-9
Struck AW, Thompson ML, Wong LS, Micklefield J (2012) S-adenosyl-methionine-dependent methyltransferases: highly versatile enzymes in biocatalysis, biosynthesis and other biotechnological applications. ChemBioChem 13:2642–2655. https://doi.org/10.1002/cbic.201200556
Sullivan ML (2015) Beyond brown: polyphenol oxidases as enzymes of plant specialized metabolism. Front Plant Sci 5:783. https://doi.org/10.3389/fpls.2014.00783
Sun J, Morita H, Chen G et al (2012) Molecular cloning and characterization of copper amine oxidase from Huperzia serrata. Bioorg Med Chem Lett 22:5784–5790. https://doi.org/10.1016/j.bmcl.2012.07.102
Tian T, Wang YJ, Huang JP et al (2022) Catalytic innovation underlies independent recruitment of polyketide synthases in cocaine and hyoscyamine biosynthesis. Nat Commun 13:4994. https://doi.org/10.1038/s41467-022-32776-1
Torrens-Spence MP, Pluskal T, Li FS et al (2018) Complete pathway elucidation and heterologous reconstitution of Rhodiola salidroside biosynthesis. Mol Plant 11:205–217. https://doi.org/10.1016/j.molp.2017.12.007
Torrens-Spence MP, Chiang Y-C, Smith T et al (2020) Structural basis for divergent and convergent evolution of catalytic machineries in plant aromatic amino acid decarboxylase proteins. Proc Natl Acad Sci 117:10806–10817. https://doi.org/10.1073/pnas.1920097117
Vavricka CJ, Takahashi S, Watanabe N et al (2022) Machine learning discovery of missing links that mediate alternative branches to plant alkaloids. Nat Commun 13:1405. https://doi.org/10.1038/s41467-022-28883-8
Wang R, Han X, Xu S et al (2019) Cloning and characterization of a tyrosine decarboxylase involved in the biosynthesis of galanthamine in Lycoris aurea. PeerJ 7:e6729. https://doi.org/10.7717/peerj.6729
Wang J, Zhang Z-K, Jiang F-F et al (2020) Deciphering the biosynthetic mechanism of pelletierine in Lycopodium alkaloid biosynthesis. Org Lett 22:9725–8729. https://doi.org/10.1021/acs.orglett.0c03339
Wang YJ, Huang JP, Tian T et al (2022) Discovery and engineering of the cocaine biosynthetic pathway. J Am Chem Soc 144:22000–22007. https://doi.org/10.1021/jacs.2c09091
Wink M (2007) Ecological Roles of Alkaloids. In: Fattorusso E, Taglialatela-Scafati O (eds) Modern alkaloids: structure, isolation, synthesis, and biology. Wiley, New York, pp 1–24
Winzer T, Kern M, King AJ et al (2015) Morphinan biosynthesis in opium poppy requires a P450-oxidoreductase fusion protein. Science 349:309–312. https://doi.org/10.1126/science.aab1852
Wood CC, Petrie JR, Shrestha P et al (2009) A leaf-based assay using interchangeable design principles to rapidly assemble multistep recombinant pathways. Plant Biotechnol J 7:914–924. https://doi.org/10.1111/j.1467-7652.2009.00453.x
Yang T, Nagy I, Mancinotti D et al (2017) Transcript profiling of a bitter variety of narrow-leafed lupin to discover alkaloid biosynthetic genes. J Exp Bot 68:5527–5537. https://doi.org/10.1093/jxb/erx362
Zakaria MM, Stegemann T, Sievert C et al (2022) Insights into polyamine metabolism: homospermidine is double-oxidized in two discrete steps by a single copper-containing amine oxidase in pyrrolizidine alkaloid biosynthesis. Plant Cell 34:2364–2382. https://doi.org/10.1093/plcell/koac068
Zekiri F, Molitor C, Mauracher SG et al (2014) Purification and characterization of tyrosinase from walnut leaves (Juglans regia). Phytochemistry 101:5–15. https://doi.org/10.1016/j.phytochem.2014.02.010
Zhang J, Liu YQ, Fang J (2023) The biological activities of quinolizidine alkaloids. In: Alkaloids: chemistry and biology. Academic Press, London, pp 1–37
Acknowledgements
This work was supported by startup funds and NIH R35 GM150908 awarded to R.S.N.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Muro-Villanueva, F., Nett, R.S. Emerging functions within the enzyme families of plant alkaloid biosynthesis. Phytochem Rev (2023). https://doi.org/10.1007/s11101-023-09901-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11101-023-09901-z