Abstract
Stemonae Radix (dried roots of Stemona japonica (Bl.) Miq., Stemona sessilifolia (Miq.) Miq., and Stemona tuberosa Lour.), a widely used traditional Chinese medicine (TCM), is also known as “Baibu” (Chinese: 百部). Stemonae Radix has a wide range of pharmacological effects, mainly used to treat respiratory diseases and as an insecticide. In this review, with the help of both published materials (such as ancient and modern books, monographs on medicinal plants, and the pharmacopoeia of different countries) and electronic databases (such as PubMed, Web of Science, and Google Scholar), we have summarized the up-to-date research progress in botany, traditional application, phytochemistry, pharmacology, toxicity, and quality control, and discussed the research prospects and development direction of Stemonae Radix and its origin plants. Up to now, more than 170 compounds were isolated and identified from the three official species of Stemonae Radix, mainly including Stemona alkaloids, Stemona stilbenoids and other components. This medicine has also exhibited pharmacological activities, including antitussive, antiviral, insecticidal, antitumor, antioxidant, and therapeutic effects on lung diseases. However, pharmacological reports most often attribute activities to various extracts, rather than specific compounds. This hampers the establishment of a connection between chemical constituents and pharmacological activities, resulting in a difficulty to identify the major active compounds. Therefore, in the future, it is necessary to design experiments to deeply understand the development and clinical use of Stemonae Radix.
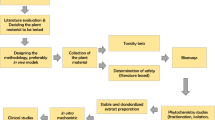
Graphic abstract










Similar content being viewed by others
Abbreviations
- TCM:
-
Traditional Chinese medicine
- Ref.:
-
Reference
- S. japonica :
-
Stemona japonica (Bl.) Miq.
- S. sessilifolia :
-
Stemona sessilifolia (Miq.) Miq.
- S. tuberosa :
-
Stemona tuberosa Lour.
- NTS:
-
Neotuberostemonine
- PSN:
-
Protostemonine
- LPS:
-
Lipopolysaccharide
- i.p.:
-
Intraperitoneal
- i.g.:
-
Intragastric
- p.o.:
-
Oral administration
- AChE:
-
Acetylcholinesterase
- 6-OHDA:
-
6-Hydroxydopamine
- PD:
-
Parkinson’s disease
- MDR:
-
Multidrug-resistant
- DPPH:
-
Diphenyl-P-Picrylhydrazyl
- BLM:
-
Bleomycin
- HIF-1α:
-
Hypoxia inducible factor-1α
- TGF-β:
-
Transforming growth factor-β
- FGF2:
-
Fibroblast growth factor 2
- α-SMA:
-
α-Smooth muscle actin
- TRAF6:
-
Tumor necrosis factor receptor-associated factor 6
- NF-κB:
-
Nuclear factor kappa B
- MAPK:
-
Mitogen-activated protein kinase
- AKT:
-
Protein kinase B
- iNOS:
-
Inducible nitric oxide synthase
References
Adams M, Pacher T, Greger H, Bauer R (2005) Inhibition of leukotriene biosynthesis by stilbenoids from Stemona species. J Nat Prod 68:83–85. https://doi.org/10.1021/np0497043
Bardají N, Sánchez-Izquierdo F, Alibés R, Font J, Busqué F, Figueredo M (2012) Flexible approach to Stemona alkaloids: total syntheses of (−)-stemospironine and three new diastereoisomeric analogs. Org Lett 14:4854–4857. https://doi.org/10.1021/ol302185j
Brem B, Seger C, Pacher T, Hofer O, Greger H (2002) Feeding deterrence and contact toxicity of Stemona alkaloids a source of potent natural insecticides. J Agric Food Chem 50:6383–6388
Brem B et al (2004) Antioxidant dehydrotocopherols as a new chemical character of Stemona species. Phytochemistry 65:2719–2729. https://doi.org/10.1016/j.phytochem.2004.08.023
Changying Z, Hongzheng F, Haimin L, Jun L, Wenhan L (1999) New alkaloids from the roots of Stemona japonica Miq. J Chin Pharm Sci 8:185–190
Chen X, Ju C, Xia L, Wei X, Jia T (2012) The difference in antitussive and expectorant activity between the different polar franctions of crude and processed Stemona tuberose. Chin J Exp Tradit Med Formulae 18:146–149
Chen X, Wei X, Deng S, Jia T (2013) Effect of raw instead of processed Baibu on chemical constituents in prescription. Guangzhou Chem Ind 41:136–138
Cheng D, Guo J, Chu TT, Röder E (1988) A study of Stemona alkaloids, III. Application of 2D-NMR spectroscopy in the structure determination of stemoninine. J Nat Prod 51:202–211
Chung H, Hon P, Lin G, But PP, Dong H (2003) Antitussive activity of Stemona alkaloids from Stemona tuberosa. Planta Med 69:914–920. https://doi.org/10.1055/s-2003-45100
Cui G, Huang R, He Q (2011) Stemonae Radix and honey Stemonae Radix. Cap Med 18:47
Dong W, Sun H, Zhang A, Meng X, Wang X, Wang W (2018) Preliminary study on serum pharmacochemistry of anti-mycoplasma pneumoniae elution fraction in Stemona tuberosa Lour. extract. Chin J Hosp Pharm 38:1387–1393
Gao J, Zhang X (2005) Experimental observation on germicidal efficacy of a plant compound disinfectant. Chin J Disinfect 22:305–306
Goldstein DM, Wipf P (1996) Studies toward the synthesis of Stemona alkaloids; a short synthesis of the tricyclic core of tuberostemonines. Tetrahedron Lett 37:739–742
Greger H (2012) The diversity of Stemona stilbenoids as a result of storage and fungal infection. J Nat Prod 75:2261–2268. https://doi.org/10.1021/np300690c
Guo A, Jin L, Deng Z, Cai S, Guo S, Lin W (2008) New Stemona alkaloids from the roots of Stemona sessilifolia. Chem Biodivers 5:598–605
Han L, Ma Y, An L, Zhang Q, Wang C, Zhao Q (2016) Non-alkaloids extract from Stemona sessilifolia enhances the activity of chemotherapeutic agents through P-glycoprotein-mediated multidrug-resistant cancer cells. Nat Prod Lett 10:1–4
Han Lin W, Hongzheng F (1999) Three new alkaloids from the roots of Stemona tuberosa Lour. J Chin Pharm Sci 8:1–7
Han Lin W, Ma L, Shen Cai M, Barnes RA (1994) Two minor alkaloids from roots of Stemona tuberosa. Phytochemistry 36:1333–1335. https://doi.org/10.1016/S0031-9422(00)89662-4
Hanbo Z, Ping Z (2007) Cultivation techniques of Baibu special. Econ Anim Plants 10:42. https://doi.org/10.3969/j.issn.1001-4713.2007.03.030
Hitotsuyanagi Y, Hikita M, Oda T, Kakuta D, Fukaya H, Takeya K (2006) Structures of four new alkaloids from Stemona sessilifolia. Tetrahedron 63:1008–1013
Hitotsuyanagi Y, Hikita M, Nakada K (2007) Sessilifoliamide I, a new alkaloid from Stemona sessilifolia. Heterocycles 71:2035–2040
Hitotsuyanagi Y, Takeda E, Fukaya H, Takeya K (2008) Sessilifoliamine A and sessilifoliamide J: new alkaloids from Stemona sessilifolia. Tetrahedron Lett 49:7376–7379
Hitotsuyanagi Y, Hikita M, Uemura G, Fukaya H, Takeya K (2010a) Structures of stemoxazolidinones A–F, alkaloids from Stemona sessilifolia. Tetrahedron 67:455–461
Hitotsuyanagi Y, Uemura G, Takeya K (2010b) Sessilifoliamides K and L: new alkaloids from Stemona sessilifolia. Tetrahedron Lett 51:5694–5696
Hu JP, Yang DH, Lin WH, Cai SQ (2009) Alkaloids from the roots of Stemona tuberosa. Helv Chim Acta 92:2125–2133
Irie H, Masaki N, Ohno K, Osaki K, Taga T, Uyeo S (1970) The crystal structure of a new alkaloid, stemofoline, from Stemona japonica. J Chem Soc D Chem Commun 17:1066
Jangmi Y, Ki Yong L, Byoungduck P (2019) Neotuberostemonine inhibits osteoclastogenesis via blockade of NF-κB pathway. Biochimie 157:81–91
Jiang D, Wang G (2003) Study on the bacteriostatic activity of 22 Chinese herbal medicines. Liao Ning High Vocat Tech Inst J 005(140–141):146
Jiang R, Hon P, But PP, Chung H, Lin G, Ye W, Mak TCW (2002) Isolation and stereochemistry of two new alkaloids from Stemona tuberosa. Tetrahedron 58:6705–6712. https://doi.org/10.1016/S0040-4020(02)00678-6
Jiang R et al (2006a) Alkaloids and chemical diversity of Stemona tuberosa. J Nat Prod 69:749–754. https://doi.org/10.1021/np050539g
Jiang R, Hon P, Xu Y, Chan Y, Xu H, Shaw P, But PP (2006b) Isolation and chemotaxonomic significance of tuberostemospironine-type alkaloids from Stemona tuberosa. Phytochemistry 67:52–57. https://doi.org/10.1016/j.phytochem.2005.10.004
Jiang D, Wu J, Liu H, Guan H (2011a) Advances in alkaloids constituents and bioactivities of Stemona. Med Mater J Anhui Agric Sci 39:19097–19099
Jiang D, Yang X, Liu H, Guan H (2011b) Development and validation of a high-performance liquid chromatographic method for the determination of stemoninine in rat plasma after administration of Stemona tuberosa extracts. Biomed Chromatogr 25:498–502. https://doi.org/10.1002/bmc.1474
Jin L (2015) Screening for anti-inflammat ion component in Radix Stemonae. Chin Tradit Med 13:140–142
Jun W, Yu G, Chaofeng Z, Mian Z (2012) Non-alkaloid Constituents from Stemona tuberosa. Pharmaceut Clin Res 20:193–195
Junsong L, Tong Z, Yiqiong P, Tao W, Jiansheng T (2011) A study on the source of Radix Stemonae in the first Chinese Pharmacopoeia. J Chin Med Mater 34:1700–1703
Kakuta D, Hitotsuyanagi Y, Matsuura N, Fukaya H, Takeya K (2003) Structures of new alkaloids sessilifoliamides A–D from Stemona sessilifolia. Tetrahedron 59:7779–7786
Kaltenegger E et al (2003) Insecticidal pyrido [1, 2-a] azepine alkaloids and related derivatives from Stemona species. Phytochemistry 63:803–816
Khamko VA, Quang DN, Dien PH (2013) Three new phenanthrenes, a new stilbenoid isolated from the roots of Stemona tuberosa Lour. and their cytotoxicity. Nat Prod Res 27:2328–2332. https://doi.org/10.1080/14786419.2013.832677
Kil Y, Park J, Han A, Woo HA, Seo E (2015) A new 9,10-dihydrophenanthrene and cell proliferative 3,4-δ-dehydrotocopherols from Stemona tuberosa. Molecules 20:5965–5974
Kostecki K, Engelmeier D, Pacher T, Hofer O, Vajrodaya S, Greger H (2004) Dihydrophenanthrenes and other antifungal stilbenoids from Stemona cf. pierrei. Phytochemistry 65:99–106
Lai D, Yang Z, Xue W, Sheng J, Shi Y, Yao X (2013) Isolation, characterization and acetylcholinesterase inhibitory activity of alkaloids from roots of Stemona sessilifolia. Fitoterapia 89:257–264. https://doi.org/10.1016/j.fitote.2013.06.010
Lee KY, Sung SH, Kim YC (2006) Neuroprotective bibenzyl glycosides of Stemona tuberosa roots. J Nat Prod 69:679–681. https://doi.org/10.1021/np0504154
Lei Z, Qian-dong Q, Xiao-lin Z, De-kui Z (2009) The Research Advancement on the Genus Stemona in China. North Hortic 4:105–108
Leung PH, Zhang L, Zuo Z, Lin G (2006) Intestinal absorption of Stemona alkaloids in a Caco-2 cell model. Planta Med 72:211–216. https://doi.org/10.1055/s-2005-916195
Li Z (2008) History of processing of Baibu decoction pieces. China foreign medical treatment 11: 46–47
Li S et al (2007) Quality evaluation of Radix Stemonae through simultaneous quantification of bioactive alkaloids by high-performance liquid chromatography coupled with diode array and evaporative light scattering detectors. Biomed Chromatogr BMC 21:1088–1094
Lin W, Ye Y, Xu R (1992) Chemical studies on new Stemona alkaloids, IV. Studies on new alkaloids from Stemona tuberosa. J Nat Prod 55:571–576
Lin L, Zhong Q, Cheng T, Tang C, Ke C, Lin G, Ye Y (2006) Stemoninines from the roots of Stemona tuberosa. J Nat Prod 69:1051–1054. https://doi.org/10.1021/np0505317
Lin L et al (2008a) Croomine- and tuberostemonine-type alkaloids from roots of Stemona tuberosa and their antitussive activity. Tetrahedron 64:10155–10161. https://doi.org/10.1016/j.tet.2008.08.046
Lin L, Li KM, Tang C, Ke C, Rudd JA, Lin G, Ye Y (2008b) Antitussive stemoninine alkaloids from the roots of Stemona tuberosa. J Nat Prod 71:1107–1110
Lin L, Yang X, Tang C, Ke C, Zhang J, Ye Y (2008c) Antibacterial stilbenoids from the roots of Stemona tuberosa. Phytochemistry 69:457–463. https://doi.org/10.1016/j.phytochem.2007.07.012
Liqin Z (2015) Nonalkaloid Constituents from Stemona tuberosa. China Mod Med 22:91–94
Liu ZL, Goh SH, Ho SH (2017) Screening of Chinese medicinal herbs for bioactivity against Sitophilus zeamais Motschulsky and Tribolium castaneum (Herbst). J Stored Prod Res 43:290–296
Lu L, Ye W, Zhao S, Zhang X (2005) Chemical constituents of Stemona sessilifolia. J China Pharm Univ 36:408
Lv XM et al (2018) Neotuberostemonine inhibits the differentiation of lung fibroblasts into myofi broblasts in mice by regulating HIF-1α signaling. Acta Pharmacol Sin 9:1501–1512
National Pharmacopoeia Committee (2020) Pharmacopoeia of People’s Republic of China. China Medical Science and Technology Press, Beijing
Pacher T, Seger C, Engelmeier D, Vajrodaya S, Hofer O, Greger H (2002) Antifungal stilbenoids from Stemona collinsae. J Nat Prod 65:820–827
Pilli RA, de Oliveira MDCF (2000) Recent progress in the chemistry of the Stemona alkaloids. Nat Prod Rep 17:117–127
Pilli RA, Rosso GB, de Oliveira MDCF (2010) The chemistry of Stemona alkaloids: An update. Nat Prod Rep 27:1908. https://doi.org/10.1039/c005018k
Pudjiastuti P, Pyne SG, Sugiyanto LW (2012) Isolation of tuberospironine A, a novel croomine derivative from Stemona tuberosa Lour. Phytochem Lett 5:358–360
Qian F (2017) Protostemonine effectively attenuates lipopolysaccharide-induced acute lung injury in mice. Acta Pharmacol Sin 39:1–12
Qin GW, Xu RS (1998) Recent advances on bioactive natural products from Chinese medicinal plants. Med Res Rev 18:375–382
Rascol O, Payoux P, Ory F, Ferreira JJ, Brefel-Courbon C, Montastruc JL (2010) Limitations of current Parkinson’s disease therapy. Ann Neurol 53:S3–S15
Rinner B, Siegl V, Pürstner P, Efferth T, Brem B, Greger H, Pfragner R (2004) Activity of novel plant extracts against medullary thyroid carcinoma cells. Anticancer Res 24:495
Sakata K, AoKI K, Chang C, Sakurai A, Tamura S, Murakoshi S (1978) Stemospironine, a new insecticidal alkaloid of Stemona japonica Miq. Isolation, structural determination and activity. Agric Biol Chem 42:457–463
Schinnerl J, Brem B, But PP, Vajrodaya S, Hofer O, Greger H (2007) Pyrrolo- and pyridoazepine alkaloids as chemical markers in Stemona species. Phytochemistry 68:1417–1427. https://doi.org/10.1016/j.phytochem.2007.03.002
Seger C, Mereiter K, Kaltenegger E, Pacher T, Greger H, Hofer O (2004) Two Pyrrolo[1,2-a]azepine type alkaloids from Stemona collinsae craib: structure elucidations, relationship to asparagamine A, and a new biogenetic concept of their formation. Chem Biodivers 1:265–279. https://doi.org/10.1002/cbdv.200490023
Stuppner H (2008) Nonaqueous capillary electrophoresis-electrospray ionization-ion trap-mass spectrometry analysis of pyrrolo- and pyrido[1,2-a]azepine alkaloids in Stemona. Electrophoresis 29:2079–2087
Tang C, Chen T, Velten R, Jeschke P, Ebbinghaus-Kintscher U, Geibel S, Ye Y (2008) Alkaloids from stems and leaves of Stemona japonica and their insecticidal activities. J Nat Prod 71:112–116. https://doi.org/10.1021/np070427k
Wang FP, Chen QH (2014) Stemona alkaloids: biosynthesis, classification, and biogenetic relationships. Nat Prod Commun 12:1809–1822
Wang W, Chen X (2016) Comparative study on the efficacy and toxicity of Baibu honey before and after roasting. Asia-Pac Tradit Med 12:8–9
Wang P, Liu AL, An Z, Li ZH, Du GH, Qin HL (2007a) Novel Alkaloids from the roots of Stemona sessilifolia. Chem Biodivers 4:523–530. https://doi.org/10.1002/cbdv.200790045
Wang P, Qin HL, Li ZH, Liu AL, Du GH (2007b) A new alkaloid from Stemona sessilifolia. Chin Chem Lett 18:152–154
Wang P, Liu A, Li Z, Du G, Qin H (2008) Stemoninine-type alkaloids from the roots of Stemona sessilifolia. J Asian Nat Prod Res 10:311–314. https://doi.org/10.1080/10286020701833511
Wang Z, Shen L, Zhang Z, Yang P, Tang J (2013) Research progress of Baibu in the treatment of pulmonary diseases. J Clin Pulm Med 18:520–522
Wang Z, Yang P, Shen L, Tang J (2015) Clinical study of BaiBu YangFei decocted extract in the treatment for COPD patients at stable stage. West J Tradit Chin Med 28:56–59
Wu Y et al (2019) Protostemonine alleviates heat-killed methicillin-resistant Staphylococcus aureus-induced acute lung injury through MAPK and NF-κB signaling pathways. Int Immunopharmacol 77:105964
Xiadong C, Guojun X (1991) Protostemotinine, a new alkaloid Stemona sessilifolia (Miq.) Miq. J China Pharm Univ 22:92
Xiang J et al (2016) Neotuberostemonine attenuates bleomycin-induced pulmonary fibrosis by suppressing the recruitment and activation of macrophages. Int Immunopharmacol 36:158–164
Xie W, Ding Q, Sun J, Zhang C, Zhang M, Xu X (2018) Protective effects of Baibu Tang on bleomycin-induced pulmonary fibrosis in mice. J China Pharm Univ 4:483–489
Xu YT (2012) Authentication and quality assessment of the antitussive Herb Baibu (Radix Stemonae). Adv Bot Res 62:1–33
Xu R (2000) Some bioactive natural products from Chinese medicinal plants. In: Studies in natural products chemistry, vol 21. Elsevier, pp 729–772
Xu R, Lu Y, Chu J, Iwashita T, Naoki H, Naya Y, Nakanishi K (1982) Studies on some new Stemona alkaloids: a diagnostically useful 1H NMR line-broadening effect. Tetrahedron 38:2667–2670
Xu Y et al (2006) Antitussive effects of Stemona tuberosa with different chemical profiles. J Ethnopharmacol 108:46–53. https://doi.org/10.1016/j.jep.2006.04.022
Xu Y, Shaw P, Jiang R, Hon P, Chan Y, But PP (2010) Antitussive and central respiratory depressant effects of Stemona tuberosa. J Ethnopharmacol 128:679–684. https://doi.org/10.1016/j.jep.2010.02.018
Yang Y, Guo-Wei Q, Ren-Sheng X (1994) Alkaloids of Stemona japonica. Phytochemistry 37:1205–1208
Yang X, Tang C, Ye Y (2006) Stilbenoids from Stemona japonica. J Asian Nat Prod Res 8:47–53
Yang XZ, Lin LG, Tang CP, Liu YQ, Ye Y (2007a) Nonalkaloid constituents from Stemona japonica. Helv Chim Acta 90:318–325
Yang XZ, Tang CP, Ke CQ, Ye Y (2007b) Stilbenoids from Stemona sessilifolia. J Asian Nat Prod Res 9:261–266. https://doi.org/10.1080/10286020600604310
Yang XZ et al (2009) Alkaloids from roots of Stemona sessilifolia and their antitussive activities. Planta Med 75:174–177. https://doi.org/10.1055/s-0028-1088345
Ye Y, Xu RS (1992) Studies on new alkaloids of Stemona japonica. Chin Chem Lett 3:511–514
Zhang T, Zhang Y, Tao J (2007) Antibacterial constituents from Stemona sessilifolia. J Asian Nat Prod Res 9:479–485. https://doi.org/10.1080/10286020701189286
Zhang Y, Xu G, Zhang T (2008) Antifungal stilbenoids from Stemona japonica. J Asian Nat Prod Res 10:634–639. https://doi.org/10.1080/10286020802133555
Zhang Y, Feng N, Xiu Y, Tao J (2010a) Comparison of total alkaloids in Baibu honey before and after roasting. Chin Patent Drug 32:451–453
Zhang YZ, Tong Z, Tao JS (2010b) Study on pharmacokinetics of protostemonine in rats. Lishizhen Med Mater Med Res 21:1364–1365
Zhang R, Tian H, Wu Y, Sun X, Zhang J, Ma Z, Jiang R (2014) Isolation and chemotaxonomic significance of stenine- and stemoninine-type alkaloids from the roots of Stemona tuberosa. Chin Chem Lett 25:1252–1255
Zhao W, Qin G, Ye Y, Xu R, Le X (1995) Bibenzyls from Stemona tuberosa. Phytochemistry 38:711–713
Zhong Y, Weidong G, Ying G, Weimin L (2009) Quality research progress of Baibu. Chin J Ethnomed Ethnopharm 18:55–56
Zhou Y et al (2006) Analyses of Stemona alkaloids in Stemonass tuberosa by liquid chromatography/tandem mass spectrometry. Rapid Commun Mass Spectrom 20:1030–1038. https://doi.org/10.1002/rcm.2409
Zhou X, Leung PH, Li N, Ye Y, Zhang L, Zuo Z, Lin G (2009) Oral absorption and antitussive activity of tuberostemonine alkaloids from the roots of Stemona tuberosa. Planta Med 75:575–580. https://doi.org/10.1055/s-0029-1185363
Zraunig A, Pacher T, Brecker L, Greger H (2014) Phenylbenzofuran-type stilbenoids from Stemona species. Phytochem Lett 9:33–36
Acknowledgements
The work was supported by grants from the National Key Research and Development Project (No.2018YFC1707106), and the Key Research and Development Plan of Shandong Province in 2018 (No.2018CXGC1305). The authors would like to thank Enago (www.enago.cn) for the English language review.
Author information
Authors and Affiliations
Contributions
Lixia Wang and Hongwei Wu searched the literature, collected the data, and drafted the manuscript; Lixia Wang and Zhuju Wang contributed to analysis and manuscript preparation; Cong Liu, Tong Jiang, Xiaoyun Yang, Xiaoxu Chen helped checking the chemical structures and formula; Lixia Wang downloaded the documents and made classification; Liying Tang and Zhuju Wang contributed comments for version of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, L., Wu, H., Liu, C. et al. A review of the botany, traditional uses, phytochemistry and pharmacology of Stemonae Radix. Phytochem Rev 21, 835–862 (2022). https://doi.org/10.1007/s11101-021-09765-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11101-021-09765-1