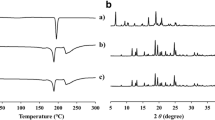
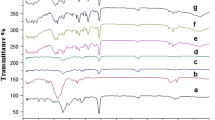
Nitrofurantoin is an old antibiotic currently prescribed to treat urinary tract infections. High dosing frequency causes fluctuations in the drug plasma concentration making less desirable prescription. This work was aimed to overcome these shortcomings by developing extended-release formulations. Tablets were prepared using nitrofurantoin and crosslinked poly(acrylic acid) and hydroxypropyl methylcellulose (HPMC) as hydrophilic polymer matrices. Modified tablets containing HPMC exhibited adequate swelling, erosion, and dissolution profiles and showed potential to decrease the frequency of administrations and reduce fluctuations in the drug plasma concentration. This would result in higher therapeutic effect and increases patient compliance by reducing adverse effects.



Similar content being viewed by others
References
Urinary Tract Infection. Centers for Disease Control and Prevention [Internet]. [cited 2018 Oct 10]. Available from: https://www.cdc.gov/antibiotic-use/community/for-patients/common-illnesses/uti.html
Medical Student Curriculum: Adult UTI [Internet]. 2020 [cited 2020 Aug 2]. Available from: https://www.auanet.org/education/auauniversity/for-medical-students/medical-students-curriculum/medical-student-curriculum/adult-uti
World Health Organization. Urinary Tract Infections [Internet]. [cited 2018 Nov 9]. Available from: www.who.int/gpsc/informationcentre/cauda-utieccmid.pdf
Foundation NK. Urinary Tract Infections [Internet]. [cited 2018 Dec 9]. Available from: https://www.kidney.org/sites/default/files/uti.pdf
R. M. Klevens, J. R. Edwards, C. L. Richards Jr., et al., Estimating Health Care-Associated Infections and Deaths in U. S. Hospitals, 2002. Public Health Rep [Internet]. 2007; 122(April):160–166. Available from: http://www.cdc.gov/HAI/pdfs/hai/infectionsdeaths.pdf
Centers for Disease Control and Prevention, National Center for Emerging and Zoonotic Infectious Diseases, Division of Healthcare Quality Promotion. Adult Treatment Recommendations. [Internet]. [cited 2018 Feb 10]. Available from: https://www.cdc.gov/antibiotic-use/community/for-hcp/outpatient-hcp/adult-treatment-rec.html
A. E. Muller, E. M. Verhaegh, S. Harbarth, et al., Clin. Microbiol. Infect., 23(6), 355–362 (2017).
B. G. Katzung and A. J. Trevor, Miscellaneous Antimicrobial Agents; Disinfectants, Antiseptics, and Sterilants, in: B. G. Katzung. Basic and Clinical Pharmacology, 14th edn, McGraw-Hill Education: New York, NY (2017).
A. Burke and M. D. Cunha, Med. Clin. North Am., 90(6), 1089 – 1107 (2006).
G. Vasvári, J. Kalmár, P. Veres, et al., Drug Discov. Today Technol., 27(xx), 71 – 80 (2018).
A. Burke and M. D. Cunha, Urology, 32(1), 67 – 71 (1988).
R. Gröning and G. Heun, Int. J. Pharm., 56(2), 111 – 116 (1989).
W. D. Mason, J. D. Conklin, and F. J. Hailey, Int. J. Pharm., 36(2–3), 105 – 111 (1987).
C. Maderuelo, A. Zarzuelo and J. M. Lanao, J. Control. Release, 154(1), 2 – 19 (2011).
B. M. Gibaldi and S. Feldman, J. Pharm. Sci., 56(10), 1238 – 1242 (1967).
Brasil. Ministério da Saúde-MS, Agência Nacional de Vigilância Sanitária – ANVISA. Farmacopeia Brasileira, 6a edição. [Internet]. 2019; 1. Available from: http://portal.anvisa.gov.br/
A. P. Cruz, C. D. Bertol, H. K. Stulzer, et al., Pharm. Chem. J., 42(7), (2008).
J. Siepmann and F. Siepmann, Int. J. Pharm., 364(2), 328 – 343 (2008).
P. Costa and J. M. S. Lobo, Eur. J. Pharm. Sci., 13, 123 – 133 (2001).
J. W. Moore and H. H. Flanner, Pharm. Technol., 20(6), 64 – 74 (1996).
FDA Guidance for Industry: Dissolution Testing of Immediate Release Solid Oral Dosage Forms [Internet]. Vol. 4, U. S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research. 1997 [cited 2017 Jun 8]. pp. 15 – 22. Available from: https://www.fda.gov/downloads/drugs/guidances/ucm070237.pdf
Brasil. Resolução RDC No. 31, de 11 de agosto de 2010 [Internet]. Agência Nacional de Vigilância Sanitária (Anvisa). 2010 [cited 2016 Jun 25]. Available from: http://portal.anvisa.gov.br/documents/33880/2568070/res0031_11_08_2010.pdf/5e157d15-d3d5-4bb9-98db-5667e4d9e0c8
N. H. Anderson, M. Bauer, N. Boussac, et al., J. Pharm. Biomed. Anal., 17(4–5), 811 – 822 (1998).
Brasil. Resolução Da Diretoria Colegiada - RDC No. 166, de 24 de julho de 2017 [Internet]. Ministério da Saúde – MS Agência Nacional de Vigilância Sanitária ANVISA. 2017 [cited 2017 Aug 2]. Available from: http://portal.anvisa.gov.br/documents/10181/2721567/RDC1662017COMP.pdf/d5fb92b3-6c6b-4130-8670-4e3263763401
International Conference on Harmonization. Validation of Analytical Procedures: Text and Methodology Q2 (R1). ICH Steer Committee, Switz. 2005; 17.
S. Dash, P. Murthy, L. Nath, and P. Chowdhury, Acta Pol. Pharm. Drug Res., 67(3), 217 – 223 (2010).
R. Manadas, M. E. Pina, and F. Veiga, Rev. Bras. Ciências Farm., 38(4) (2002).
Conflict of Interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bertol, C.D., Schonhorst, É.M., Tristacci, N.D.B. et al. Extended-Release Tablets of Nitrofurantoin: An Old Antibiotic Carried in a New Formulation. Pharm Chem J 55, 808–813 (2021). https://doi.org/10.1007/s11094-021-02499-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-021-02499-6