Abstract
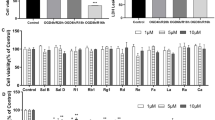
Recently, necroptosis has emerged as one of the important mechanisms of ischemia stroke. Necroptosis can be rapidly activated in endothelial cells to cause vascular damage and neuroinflammation. Panax notoginseng saponins (PNS), an ingredient extracted from the root of Panax notoginseng (Burk.) F.H. Chen, was commonly used for ischemic stroke, while its molecular mechanism and targets have not been fully clarified. Our study aimed to clarify the anti-necroptosis effect of PNS by regulating RIP1-RIP3-MLKL signaling pathway in brain microvascular endothelial cells (BMECs) subjected to transient oxygen-glucose deprivation (OGD/resupply [R]). In vitro, the necroptosis model of rat BMECs was established by testing the effect of OGD/R in the presence of the pan-caspase inhibitor z-VAD-FMK. After administration of PNS and Nec-1, cell viability, cell death modality, the expression of RIP1-RIP3-MLKL pathway and mitochondrial membrane potential (Δψm) level were investigated in BMECs upon OGD/R injury. The results showed that PNS significantly enhanced cell viability of BMECs determined by CCK-8 analysis, and protected BMECs from necroptosis by Flow cytometry and TEM. In addition, PNS inhibited the phosphorylation of RIP1, RIP3, MLKL and the downstream expression of PGAM5 and Drp1, while similar results were observed in Nec-1 intervention. We further investigated whether PNS prevented the Δψm depolarization. Our current findings showed that PNS effectively reduced the occurrence of necroptosis in BMECs exposed to OGD/R by inhibition of the RIP1-RIP3-MLK signaling pathway and mitigation of mitochondrial damage. This study provided a novel insight of PNS application in clinics.




Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Benjamin EJ, Muntner P, Alonso A et al (2019) Heart disease and stroke statistics—2019 update: a report from the American Heart Association. Circulation 139:e56–e528
Chen AQ, Fang Z, Chen XL et al (2019) Microglia-derived TNF-α mediates endothelial necroptosis aggravating blood brain–barrier disruption after ischemic stroke. Cell Death Dis 10:487
Degterev A, Huang Z, Boyce M et al (2005) Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat Chem Biol 1:112–119
Naito MG, Xu D, Amin P et al (2020) Sequential activation of necroptosis and apoptosis cooperates to mediate vascular and neural pathology in stroke. Proceedings of the National Academy of Sciences 117: 4959–4970
Wehn AC, Khalin I, Duering M et al (2021) RIPK1 or RIPK3 deletion prevents progressive neuronal cell death and improves memory function after traumatic brain injury. Acta Neuropathol Commun 9:1–18
Zhang Y, Li K, Wang X et al (2021) CSE-Derived H2S Inhibits Reactive Astrocytes Proliferation and Promotes Neural Functional Recovery after Cerebral Ischemia/Reperfusion Injury in Mice Via Inhibition of RhoA/ROCK2 Pathway. ACS Chem Neurosci 12:2580–2590
Sun T, Wang P, Deng T et al (2021) Effect of Panax notoginseng Saponins on Focal Cerebral Ischemia-Reperfusion in Rat Models: A Meta-Analysis. Front Pharmacol 11:2491
Zhang J, Guo F, Zhou R et al (2021) Proteomics and transcriptome reveal the key transcription factors mediating the protection of Panax notoginseng saponins (PNS) against cerebral ischemia/reperfusion injury. Phytomedicine 92:153613
Geng H, Zhang L, Xin C et al (2021) Xuesaitong oral preparation as adjuvant therapy for treating acute cerebral infarction: A systematic review and meta-analysis of randomized controlled trials. J Ethnopharmacol 285:114849
Wang L, Xu Z, Liang X et al (2021) Systematic Review and Meta-Analysis on Randomized Controlled Trials on Efficacy and Safety of Panax Notoginseng Saponins in Treatment of Acute Ischemic Stroke. Evidence-Based Complementary and Alternative Medicine:4694076
Li W, Li P, Liu Z et al (2014) A Chinese medicine preparation induces neuroprotection by regulating paracrine signaling of brain microvascular endothelial cells. J Ethnopharmacol 151:686–693
Zhang C, Zhang S, Wang L et al (2021) The RIG-I Signal Pathway Mediated Panax notoginseng Saponin Anti-Inflammatory Effect in Ischemia Stroke. Evidence-Based Complementary and Alternative Medicine:8878428
Li W, Li P, Hua Q et al (2009) The impact of paracrine signaling in brain microvascular endothelial cells on the survival of neurons. Brain Res 1287:28–38
Hu Y, Lei H, Wang S et al (2017) The Establishment of Necroptosis Model on Mimic Ischemic Reperfusion Injury in Brain Microvascular Endothelial Cells. World Chin Med 12:879–883
Uzunparmak B, Gao M, Lindemann A et al (2020) Caspase-8 loss radiosensitizes head and neck squamous cell carcinoma to SMAC mimetic–induced necroptosis. JCI insight 5:e139837
Yuan Z, Yi YS, Hai YY (2020) Triad3A displays a critical role in suppression of cerebral ischemic/reperfusion (I/R) injury by regulating necroptosis. Biomed Pharmacother 128:110045
Zhu YM, Lin L, Wei C et al (2021) The Key Regulator of Necroptosis, RIP1 Kinase, Contributes to the Formation of Astrogliosis and Glial Scar in Ischemic Stroke. Translational Stroke Research 12:991–1017
Zhou T, DeRoo E, Yang H et al (2021) MLKL and CaMKII Are Involved in RIPK3-Mediated Smooth Muscle Cell Necroptosis. Cells 10:2397
de Almagro MC, Vucic D (2015) Necroptosis: Pathway diversity and characteristics[C]//Seminars in cell & developmental biology. Acad Press 39:56–62
Verduijn J, Van der Meeren L, Krysko DV et al (2021) Deep learning with digital holographic microscopy discriminates apoptosis and necroptosis. Cell death discovery 7:1–10
Vandenabeele P, Galluzzi L, Vanden Berghe T et al (2010) Molecular mechanisms of necroptosis: an ordered cellular explosion. Nat Rev Mol Cell Biol 11:700–714
Linkermann A, Green DR (2014) Necroptosis. N Engl J Med 370:455–465
Zhu H, Tan Y, Du W et al (2021) Phosphoglycerate mutase 5 exacerbates cardiac ischemia-reperfusion injury through disrupting mitochondrial quality control. Redox Biol 38:101777
Xu Q, Guo J, Li X et al (2021) Necroptosis Underlies Hepatic Damage in a Piglet Model of Lipopolysaccharide-Induced Sepsis. Front Immunol 12:745
Cheng M, Lin N, Dong D et al (2021) PGAM5: a crucial role in mitochondrial dynamics and programmed cell death. Eur J Cell Biol 100:151144
Xue C, Gu X, Li G et al (2021) Mitochondrial mechanisms of necroptosis in liver diseases. Int J Mol Sci 22:66
Moujalled D, Strasser A, Liddell JR (2021) Molecular mechanisms of cell death in neurological diseases. Cell Death & Differentiation 28:2029–2044
Adameova A, Horvath C, Abdul-Ghani S et al (2022) Interplay of Oxidative Stress and Necrosis-like Cell Death in Cardiac Ischemia/Reperfusion Injury: A Focus on Necroptosis. Biomedicines 10:127
Hua X, Li B, Yu F et al (2022) Protective Effect of MFG-E8 on Necroptosis-Induced Intestinal Inflammation and Enteroendocrine Cell Function in Diabetes. Nutrients 14:604
Dong K, Zhu Y, Deng Q et al (2022) Salmonella pSLT-encoded effector SpvB promotes RIPK3-dependent necroptosis in intestinal epithelial cells. Cell Death Discovery 8:1–11
Kamiya M, Mizoguchi F, Kawahata K et al (2022) Targeting necroptosis in muscle fibers ameliorates inflammatory myopathies. Nat Commun 13:1–12
Das N, Samantaray S, Ghosh C et al (2022) Chimaphila umbellata extract exerts anti-proliferative effect on human breast cancer cells via RIP1K/RIP3K-mediated necroptosis. Phytomedicine Plus 2:100159
Tuo Q, Zhang S, Lei P (2022) Mechanisms of neuronal cell death in ischemic stroke and their therapeutic implications. Med Res Rev 42:259–305
Khuanjing T, Ongnok B, Maneechote C et al (2021) Acetylcholinesterase inhibitor ameliorates doxorubicin-induced cardiotoxicity through reducing RIP1-mediated necroptosis. Pharmacol Res 173:105882
Wu L, Chung JY, Cao T et al (2021) Genetic inhibition of RIPK3 ameliorates functional outcome in controlled cortical impact independent of necroptosis. Cell Death Dis 12:1–16
Seo J, Nam YW, Kim S et al (2021) Necroptosis molecular mechanisms: Recent findings regarding novel necroptosis regulators. Exp Mol Med 53:1007–1017
Huang HR, Cho SJ, Harris RM et al (2021) RIPK3 activates MLKL-mediated necroptosis and inflammasome signaling during Streptococcus infection. Am J Respir Cell Mol Biol 64:579–591
Liu Y, Xu Q, Wang Y et al (2021) Necroptosis is active and contributes to intestinal injury in a piglet model with lipopolysaccharide challenge. Cell Death Dis 12:1–14
Hu Y, Feng X, Chen J et al (2022) Hydrogenrich saline alleviates early brain injury through inhibition of necroptosis and neuroinflammation via the ROS/HO1 signaling pathway after traumatic brain injury. Experimental and therapeutic medicine 23:1–10
Lee AJ, Liao HJ, Hong JR (2022) Overexpression of Bcl2 and Bcl2L1 Can Suppress Betanodavirus-Induced Type III Cell Death and Autophagy Induction in GF-1 Cells. Symmetry 14:360
Acknowledgements
The authors would like to acknowledge financial support from the National Natural Science Foundation of China (Grant No.82104673,81273885) and The Fundamental Research Funds for the Central Public Welfare Research Institutes (Grant No. ZZ15-YQ-056).
Author information
Authors and Affiliations
Contributions
YH performed all experiments and wrote the manuscript; SZ, JM, SK, LW, FL, FZ, TS and CZ helped with the biological study and data statistics; WL conducted and supervised the whole experiments. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hu, Y., Lei, H., Zhang, S. et al. Panax notoginseng Saponins Protect Brain Microvascular Endothelial Cells against Oxygen-Glucose Deprivation/Resupply-Induced Necroptosis via Suppression of RIP1-RIP3-MLKL Signaling Pathway. Neurochem Res 47, 3261–3271 (2022). https://doi.org/10.1007/s11064-022-03675-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-022-03675-0