Abstract
Introduction
Right cerebral hemispheric glioblastomas (GBMs) often decrease the Karnofsky performance status (KPS) score postoperatively, despite the patient having sufficient patient function while performing daily living. This study aimed to evaluate the factors that could cause poor KPS scores during the postoperative chronic phase in patients with right cerebral hemispheric GBMs.
Methods
Data of 47 patients with newly diagnosed right cerebral hemispheric GBMs were analyzed. All patients were assessed preoperatively and 3 months postoperatively to determine KPS and brain function. To determine tumor location related to the postoperative KPS scores, we used voxel-based lesion symptom mapping (VLSM). The patients were divided into two groups (involvement and non-involvement groups) based on whether their lesion involved a significant region identified by VLSM. We then compared functional factors and prognosis between the groups using the chi-squared and log-rank tests, respectively.
Results
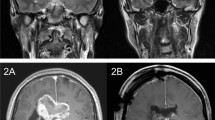
The KPS score significantly decreased after surgery compared to that preoperatively measured (p = 0.023). VLSM revealed that tumors in the white matter of temporo-parietal junction (WM-TPJ) caused a significant decline in the KPS score at three months postoperatively. The patients in the involvement group had a higher probability of impaired attention, visuospatial cognition, emotion recognition, and visual field than did those in the non-involvement group. In addition, tumor in the WM-TPJ were associated with shorter progression-free survival and overall survival (p = 0.039 and 0.023, respectively).
Conclusions
GBMs involving the right WM-TPJ are more likely to result in poor postoperative KPS scores and prognoses. Impairments of several kinds of brain functions caused by tumor invasion to the WM-TPJ may be associated with lower KPS scores.



Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Baba MA, Adali N (2021) Neurocognitive state and quality of life of patients with glioblastoma in Mediterranean countries: a systematic review. Ann Palliat Med 10:11980–11993. https://doi.org/10.21037/apm-21-1900
Rahman M, Abbatematteo J, De Leo EK, Kubilis PS, Vaziri S, Bova F, Sayour E, Mitchell D, Quinones-Hinojosa A (2017) The effects of new or worsened postoperative neurological deficits on survival of patients with glioblastoma. J Neurosurg 127:123–131. https://doi.org/10.3171/2016.7.JNS16396
Chambless LB, Kistka HM, Parker SL, Hassam-Malani L, McGirt MJ, Thompson RC (2015) The relative value of postoperative versus preoperative Karnofsky performance scale scores as a predictor of survival after surgical resection of Glioblastoma Multiforme. J Neuro-Oncol 121:359–364. https://doi.org/10.1007/s11060-014-1640-x
Osorio JA, Aghi MK (2014) Optimizing glioblastoma resection: intraoperative mapping and beyond. CNS Oncol 3:359–366. https://doi.org/10.2217/cns.14.36
Suarez-Meade P, Marenco-Hillembrand L, Sabsevitz D, Okromelidze L, Blake Perdikis BS, Sherman WJ, Quinones-Hinojosa A, Middlebrooks EH, Chaichana KL (2022) Surgical resection of gliomas in the dominant inferior frontal gyrus: consecutive case series and anatomy review of Broca’s area. Clin Neurol Neurosurg 223:107512. https://doi.org/10.1016/j.clineuro.2022.107512
Nakajima R, Kinoshita M, Okita H, Nakada M (2022) Quality of life following awake Surgery depends on ability of executive function, verbal fluency, and movement. J Neuro-Oncol 156:173–183. https://doi.org/10.1007/s11060-021-03904-6
Chaichana KL, Chaichana KK, Olivi A, Weingart JD, Bennett R, Brem H, Quinones-Hinojosa A (2011) Surgical outcomes for older patients with Glioblastoma Multiforme: preoperative factors associated with decreased survival. Clinical article. J Neurosurg 114:587–594. https://doi.org/10.3171/2010.8.JNS1081
Thomas R, O’Connor AM, Ashley S (1995) Speech and language disorders in patients with high grade glioma and its influence on prognosis. J Neuro-Oncol 23:265–270. https://doi.org/10.1007/BF01059960
Drewes C, Sagberg LM, Jakola AS, Solheim O (2016) Quality of life in patients with intracranial tumors: does Tumor laterality matter? J Neurosurg 125:1400–1407. https://doi.org/10.3171/2015.12.Jns152252
Nakada M, Nakajima R, Okita H, Nakade Y, Yuno T, Tanaka S, Kinoshita M (2021) Awake Surgery for right frontal lobe glioma can preserve visuospatial cognition and spatial working memory. J Neuro-Oncol 151:221–230. https://doi.org/10.1007/s11060-020-03656-9
Nakajima R, Kinoshita M, Nakada M (2021) Simultaneous damage of the Cingulate Cortex Zone II and Fronto-Striatal Circuit causes prolonged selective attentional deficits. Front Hum Neurosci 15:762578. https://doi.org/10.3389/fnhum.2021.762578
Nakajima R, Kinoshita M, Nakada M, Herbet G (2021) Intraoperative mapping of cognitive networks. Springer, Switzerland
Nakajima R, Kinoshita M, Okita H, Liu ZW, Nakada M (2021) Preserving right pre-motor and Posterior Prefrontal Cortices Contribute to maintaining overall basic emotion. Front Hum Neurosci 15. ARTN 61289010.3389/fnhum.2021.612890
Nakajima R, Kinoshita M, Okita H, Yahata T, Matsui M, Nakada M (2018) Neural networks mediating high-level mentalizing in patients with Right Cerebral Hemispheric Gliomas. Front Behav Neurosci 12:33. https://doi.org/10.3389/fnbeh.2018.00033
Fyllingen EH, Bo LE, Reinertsen I, Jakola AS, Sagberg LM, Berntsen EM, Salvesen O, Solheim O (2021) Survival of glioblastoma in relation to Tumor location: a statistical Tumor atlas of a population-based cohort. Acta Neurochir (Wien) 163:1895–1905. https://doi.org/10.1007/s00701-021-04802-6
Roux A, Roca P, Edjlali M, Sato K, Zanello M, Dezamis E, Gori P, Lion S, Fleury A, Dhermain F, Meder JF, Chretien F, Lechapt E, Varlet P, Oppenheim C, Pallud J (2019) MRI Atlas of IDH Wild-Type Supratentorial Glioblastoma: probabilistic maps of phenotype, management, and outcomes. Radiology 293:633–643. https://doi.org/10.1148/radiol.2019190491
Mickevicius NJ, Carle AB, Bluemel T, Santarriaga S, Schloemer F, Shumate D, Connelly J, Schmainda KM, LaViolette PS (2015) Location of Brain Tumor intersecting white matter tracts predicts patient prognosis. J Neuro-Oncol 125:393–400. https://doi.org/10.1007/s11060-015-1928-5
Liu TT, Achrol AS, Mitchell LA, Du WA, Loya JJ, Rodriguez SA, Feroze A, Westbroek EM, Yeom KW, Stuart JM, Chang SD, Harsh GRt, Rubin DL (2016) Computational Identification of Tumor Anatomic Location Associated with Survival in 2 large cohorts of human primary glioblastomas. AJNR Am J Neuroradiol 37:621–628. https://doi.org/10.3174/ajnr.A4631
Ellingson BM, Lai A, Harris RJ, Selfridge JM, Yong WH, Das K, Pope WB, Nghiemphu PL, Vinters HV, Liau LM, Mischel PS, Cloughesy TF (2013) Probabilistic radiographic atlas of glioblastoma phenotypes. AJNR Am J Neuroradiol 34:533–540. https://doi.org/10.3174/ajnr.A3253
Schag CC, Heinrich RL, Ganz PA (1984) Karnofsky performance status revisited: reliability, validity, and guidelines. J Clin Oncol 2:187–193. https://doi.org/10.1200/JCO.1984.2.3.187
Sawner KA, LaVigne JM (1970) Brunnstrom’s movement therapy in hemiplegia: A neurophysiological approach. 2nd edition. J. B. Lippincitt Company, New York, pp 41–65
Bates E, Wilson SM, Saygin AP, Dick F, Sereno MI, Knight RT, Dronkers NF (2003) Voxel-based lesion-symptom mapping. Nat Neurosci 6:448–450. https://doi.org/10.1038/nn1050
Krason A, Vigliocco G, Mailend ML, Stoll H, Varley R, Buxbaum LJ (2023) Benefit of visual speech information for word comprehension in post-stroke aphasia. Cortex 165:86–100. https://doi.org/10.1016/j.cortex.2023.04.011
Middleton LE, Lam B, Fahmi H, Black SE, McIlroy WE, Stuss DT, Danells C, Ween J, Turner GR (2014) Frequency of domain-specific cognitive impairment in sub-acute and chronic Stroke. NeuroRehabilitation 34:305–312. https://doi.org/10.3233/NRE-131030
Di Carlo DT, Benedetto N, Duffau H, Cagnazzo F, Weiss A, Castagna M, Cosottini M, Perrini P (2019) Microsurgical anatomy of the sagittal stratum. Acta Neurochir (Wien) 161:2319–2327. https://doi.org/10.1007/s00701-019-04019-8
Di L, Shah AH, Mahavadi A, Eichberg DG, Reddy R, Sanjurjo AD, Morell AA, Lu VM, Ampie L, Luther EM, Komotar RJ, Ivan ME (2023) Radical supramaximal resection for newly diagnosed left-sided eloquent glioblastoma: safety and improved survival over gross-total resection. J Neurosurg 138:62–69. https://doi.org/10.3171/2022.3.JNS212399
Coluccia D, Roth T, Marbacher S, Fandino J (2018) Impact of Laterality on Surgical Outcome of Glioblastoma patients: a retrospective single-center study. World Neurosurg 114:e121–e128. https://doi.org/10.1016/j.wneu.2018.02.084
Roh TH, Kang SG, Moon JH, Sung KS, Park HH, Kim SH, Kim EH, Hong CK, Suh CO, Chang JH (2020) Survival benefit of lobectomy over gross-total resection without lobectomy in cases of glioblastoma in the noneloquent area: a retrospective study. J Neurosurg 132:895–901. https://doi.org/10.3171/2018.12.Jns182558
Maldonado IL, Destrieux C, Ribas EC, Siqueira de Abreu Brito Guimaraes B, Cruz PP, Duffau H (2021) Composition and organization of the sagittal stratum in the human brain: a fiber dissection study. J Neurosurg 135:1214–1222. https://doi.org/10.3171/2020.7.JNS192846
Ryan NP, Genc S, Beauchamp MH, Yeates KO, Hearps S, Catroppa C, Anderson VA, Silk TJ (2018) White matter microstructure predicts longitudinal social cognitive outcomes after paediatric traumatic brain injury: a diffusion tensor imaging study. Psychol Med 48:679–691. https://doi.org/10.1017/S0033291717002057
Chen LZ, Hu XY, Ouyang L, He N, Liao Y, Liu Q, Zhou M, Wu M, Huang XQ, Gong QY (2016) A systematic review and meta-analysis of tract-based spatial statistics studies regarding attention-deficit/hyperactivity disorder. Neurosci Biobehav R 68:838–847. https://doi.org/10.1016/j.neubiorev.2016.07.022
Berro DH, Herbet G, Duffau H (2021) New insights into the anatomo-functional architecture of the right sagittal stratum and its surrounding pathways: an axonal electrostimulation mapping study. Brain Struct Funct 226:425–441. https://doi.org/10.1007/s00429-020-02186-4
Wangaryattawanich P, Hatami M, Wang JX, Thomas G, Flanders A, Kirby J, Wintermark M, Huang ES, Bakhtiari AS, Luedi MM, Hashmi SS, Rubin DL, Chen JY, Hwang SN, Freymann J, Holder CA, Zinn PO, Colen RR (2015) Multicenter imaging outcomes study of the Cancer Genome Atlas glioblastoma patient cohort: imaging predictors of overall and progression-free survival. Neurooncology 17:1525–1537. https://doi.org/10.1093/neuonc/nov117
Henker C, Kriesen T, Glass A, Schneider B, Piek J (2017) Volumetric quantification of glioblastoma: experiences with different measurement techniques and impact on survival. J Neuro-Oncol 135:391–402. https://doi.org/10.1007/s11060-017-2587-5
Starnoni D, Berthiller J, Idriceanu TM, Meyronet D, d’Hombres A, Ducray F, Guyotat J (2018) Returning to work after multimodal treatment in glioblastoma patients. Neurosurg Focus 44:E17. https://doi.org/10.3171/2018.3.FOCUS1819
Karsy M, Yoon N, Boettcher L, Jensen R, Shah L, MacDonald J, Menacho ST (2018) Surgical treatment of glioblastoma in the elderly: the impact of Complications. J Neuro-Oncol 138:123–132. https://doi.org/10.1007/s11060-018-2777-9
Huq S, Khalafallah AM, Ruiz-Cardozo MA, Botros D, Oliveira LAP, Dux H, White T, Jimenez AE, Gujar SK, Sair HI, Pillai JJ, Mukherjee D (2021) A novel radiographic marker of Sarcopenia with prognostic value in glioblastoma. Clin Neurol Neurosurg 207:106782. https://doi.org/10.1016/j.clineuro.2021.106782
Gresham G, Hendifar AE, Spiegel B, Neeman E, Tuli R, Rimel BJ, Figlin RA, Meinert CL, Piantadosi S, Shinde AM (2018) Wearable activity monitors to assess performance status and predict clinical outcomes in advanced cancer patients. NPJ Digit Med 1:27. https://doi.org/10.1038/s41746-018-0032-6
Keith RA, Granger CV, Hamilton BB, Sherwin FS (1987) The functional independence measure: a new tool for rehabilitation. Adv Clin Rehabil 1:6–18
Acknowledgements
None.
Funding
This work was supported by JSPS KAKENHI Grant Number 21H03301 and 21K19705.
Author information
Authors and Affiliations
Contributions
Conception and design: MN and RN Acquisition of data: RN. Analysis and interpretation of data: RN. Drafting article: MN, RN. Critically revising the article: All authors Reviewed the final version of the manuscript and approved it for submission. Study supervision: MN.
Corresponding author
Ethics declarations
Conflicts of interest
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Ethics approval
This study was conducted in accordance with the guidelines of the Internal Review Board of Kanazawa University and was approved by the Medical Ethics Committee of Kanazawa University (approval numbers 1797 and 3284).
Consent to participate
Written informed consent to participate in this study was obtained from all patients.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nakajima, R., Kinoshita, M., Okita, H. et al. Glioblastomas at the white matter of temporo-parietal junction cause a poor postoperative independence level. J Neurooncol 165, 191–199 (2023). https://doi.org/10.1007/s11060-023-04479-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-023-04479-0