Abstract
Background
This study aimed to measure the expression levels of peripheral blood miRNAs in brucellosis and their involvement in the different phases of the brucellosis.
Methods
The expression levels of miRNAs including miR-210, miR-155, miR-150, miR-146a, miR-139-3p, miR-125a-5p, miR-29 and miR-21 were quantified in 57 brucellosis patients subgrouped into acute, under treatment & relapse phase and 30 healthy controls (HCs) using real-time polymerase chain reaction (RT-PCR). The receiver operating characteristic (ROC) analysis curve analysis was performed to find a biomarker for discrimination of different phases of brucellosis.
Results
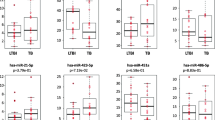
The expression of miR-155, miR-146a, miR-125a-5p, miR-29, and miR-21 was found to be elevated in the acute brucellosis patients compared to HCs. miR-29 changed in under-treatment patients, while miR-139-3p and miR-125a-5p showed alterations in relapse cases. The ROC curve analysis depicted the potential involvement of miR-21 in the pathogenesis of acute brucellosis.
Conclusion
The expression level of miR-21 is significantly augmented in acute brucellosis and has the potential to be a contributing diagnostic factor for acute infection.
Graphical Abstract




Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
de Figueiredo P, Ficht TA, Rice-Ficht A, Rossetti CA, Adams LG (2015) Pathogenesis and immunobiology of brucellosis: review of Brucella-host interactions. Am J Pathol 185:1505–1517
Pappas G, Papadimitriou P, Akritidis N, Christou L, Tsianos EV (2006) The new global map of human brucellosis. Lancet Infect Dis 6:91–99
Golshani M, Buozari S (2017) A review of brucellosis in Iran: epidemiology, risk factors, diagnosis, control, and prevention. Iran Biomed J 21:349
Abedi A-S, Hashempour-Baltork F, Alizadeh AM et al (2020) The prevalence of Brucella spp. in dairy products in the middle east region: a systematic review and meta-analysis. Acta Trop 202:105241
Hosseini SM, Abbasalipourkabir R, Jalilian FA et al (2019) Doxycycline-encapsulated solid lipid nanoparticles as promising tool against Brucella melitensis enclosed in macrophage: a pharmacodynamics study on J774A. 1 cell line. Antimicrob Resist Infect Control 8:1–12
Zowghi E, Ebadi AG, Yarahmadi M (2008) Isolation and identification of Brucella organisms in Iran. Arch Clin Infect Dis 3:185–188
Kazemi S, Mirzaei R, Sholeh M et al (2021) microRNAs in human brucellosis: a promising therapeutic approach and biomarker for diagnosis and treatment. Immun Inflamm Dis 9:1209–1218
Long H, Wang X, Chen Y, Wang L, Zhao M, Lu Q (2018) Dysregulation of microRNAs in autoimmune diseases: pathogenesis, biomarkers and potential therapeutic targets. Cancer Lett 428:90–103
Drury RE, O’Connor D, Pollard AJ (2017) The clinical application of microRNAs in infectious disease. Front Immunol 8:1182
Zhang D, Yi Z, Fu Y (2019) Downregulation of miR-20b-5p facilitates Mycobacterium tuberculosis survival in RAW 264.7 macrophages via attenuating the cell apoptosis by Mcl-1 upregulation. J Cell Biochem 120:5889–5896
Lee DY, Jeyapalan Z, Fang L et al (2010) Expression of versican 3′-untranslated region modulates endogenous microRNA functions. PLoS ONE 5:e13599
Zabaglia LM, Sallas ML, Santos MPD et al (2018) Expression of miRNA-146a, miRNA-155, IL-2, and TNF-α in inflammatory response to Helicobacter pylori infection associated with cancer progression. Ann Hum Genet 82:135–142
Castañeda-Ramírez A, González-Rodríguez D, Hernández-Pineda JA, Verdugo-Rodríguez A (2015) Blocking the expression of syntaxin 4 interferes with initial phagocytosis of Brucella melitensis in macrophages. Can J Vet Res 79:39–45
Xiao B, Liu Z, Li B-S et al (2009) Induction of microRNA-155 during Helicobacter pylori infection and its negative regulatory role in the inflammatory response. J Infect Dis 200:916–925
Budak F, Bal SH, Tezcan G, Akalın H, Goral G, Oral HB (2016) Altered expressions of miR-1238–3p, miR-494, miR-6069, and miR-139–3p in the formation of chronic brucellosis. J Immunol Res 2016:1–11
Singh J, Dhanoa JK, Choudhary RK et al (2020) MicroRNA expression profiling in PBMCs of Indian water Buffalo (Bubalus bubalis) infected with Brucella and Johne’s disease. ExRNA 2:1–13
Zhang C, Fu Q, Ding M et al (2019) Comprehensive analysis of differentially expressed serum microRNAs in humans responding to Brucella infection. Ann Transl Med 7:301
Condrat CE, Thompson DC, Barbu MG et al (2020) miRNAs as biomarkers in disease: latest findings regarding their role in diagnosis and prognosis. Cells 9:276
Rahmanpour M, Keramat F, Jourghasemi S et al (2019) Direct correlation between Th1 and Th17 responses in immunity to Brucella infection. Microbes Infect 21:441–448
Eini P, Keramat F, Hasanzadehhoseinabadi M (2012) Epidemiologic, clinical and laboratory findings of patients with brucellosis in Hamadan, west of Iran. J Res Health Sci 12:105–108
Zhou Y, Zhou B, Pache L et al (2019) Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat Commun 10:1523
Virga F, Cappellesso F, Stijlemans B et al (2021) Macrophage miR-210 induction and metabolic reprogramming in response to pathogen interaction boost life-threatening inflammation. Sci Adv 7:eabf0466
Bandyopadhyay S, Long ME, Allen L-AH (2014) Differential expression of microRNAs in Francisella tularensis-infected human macrophages: miR-155-dependent downregulation of MyD88 inhibits the inflammatory response. PLoS ONE 9:e109525
Rezaeepoor M, Pourjafar M, Tahamoli-Roudsari A, Basiri Z, Hajilooi M, Solgi G (2020) Altered expression of microRNAs may predict therapeutic response in rheumatoid arthritis patients. Int Immunopharmacol 83:106404
Cui B, Liu W, Wang X et al (2017) Brucella Omp25 upregulates miR-155, miR-21-5p, and miR-23b to inhibit interleukin-12 production via modulation of programmed death-1 signaling in human monocyte/macrophages. Front Immunol 8:708
Cremer T, Ravneberg D, Clay C, Piper-Hunter M, Marsh C (2009) MiR-155 induction by F. novicida but not the virulent F. tularensis results in SHIP down-regulation and enhanced pro-inflammatory cytokine response. PLoS ONE 4:e8508
Taganov KD, Boldin MP, Chang K-J, Baltimore D (2006) NF-κB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc Natl Acad Sci 103:12481–12486
Zheng K, Chen D-S, Wu Y-Q et al (2012) MicroRNA expression profile in RAW264. 7 cells in response to Brucella melitensis infection. Int J Biol Sci 8:1013
Monk CE, Hutvagner G, Arthur JSC (2010) Regulation of miRNA transcription in macrophages in response to Candida albicans. PLoS ONE 5:e13669
Cameron JE, Yin Q, Fewell C et al (2008) Epstein-barr virus latent membrane protein 1 induces cellular MicroRNA miR-146a, a modulator of lymphocyte signaling pathways. J Virol 82:1946–1958
Li D, Kong C, Tsun A et al (2015) MiR-125a-5p decreases the sensitivity of treg cells toward IL-6-mediated conversion by inhibiting IL-6R and STAT3 expression. Sci Rep 5:14615
Wang Y, Chen C, Xu X-d et al (2020) Levels of miR-125a-5p are altered in mycobacterium avium-infected macrophages and associate with the triggering of an autophagic response. Microbes Infect Microbes Infect 22:31–39
Potenza N, Papa U, Mosca N, Zerbini F, Nobile V, Russo A (2011) Human microRNA hsa-miR-125a-5p interferes with expression of hepatitis B virus surface antigen. Nucleic Acids Res 39:5157–5163
Banerjee S, Cui H, Xie N et al (2013) miR-125a-5p regulates differential activation of macrophages and inflammation*. J Biol Chem 288:35428–35436
Liu N, Wang L, Sun C, Yang L, Sun W, Peng Q (2016) MicroRNA-125b-5p suppresses brucella abortus intracellular survival via control of A20 expression. BMC Microbiol 16:171
Steiner David F, Thomas Molly F, Hu Joyce K et al (2011) MicroRNA-29 regulates T-box transcription factors and Interferon-γ production in helper T cells. Immunity 35:169–181
Akbulut H, Celik I, Akbulut A (2007) Cytokine levels in patients with brucellosis and their relations with the treatment. Indian J Med Microbiol 25:387
Gross A, Terraza A, Ouahrani-Bettache S, Liautard J-P, Dornand J (2000) In vitro Brucella suis infection prevents the programmed cell death of human monocytic cells. Infect Immun 68:342–351
Li Y, Sun G, Wang L (2022) MiR-21 participates in LPS-induced myocardial injury by targeting Bcl-2 and CDK6. Inflamm Res 71:205–214
He Y, Reichow S, Ramamoorthy S et al (2006) Brucella melitensis triggers time-dependent modulation of apoptosis and down-regulation of mitochondrion-associated gene expression in mouse macrophages. Infect Immun 74:5035–5046
Zhao Z, Hao J, Li X, Chen Y, Qi X (2019) MiR-21-5p regulates mycobacterial survival and inflammatory responses by targeting Bcl-2 and TLR4 in mycobacterium tuberculosis-infected macrophages. FEBS Lett 593:1326–1335
Sathiyaseelan J, Goenka R, Parent M et al (2006) Treatment of Brucella-susceptible mice with IL-12 increases primary and secondary immunity. Cell Immunol 243:1–9
Li S, Fan Q, He S, Tang T, Liao Y, Xie J (2015) MicroRNA-21 negatively regulates treg cells through a TGF-β1/Smad-independent pathway in patients with coronary heart disease. Cell Physiol Biochem 37:866–878
Rafiei A, Ardestani SK, Kariminia A, Keyhani A, Mohraz M, Amirkhani A (2006) Dominant Th1 cytokine production in early onset of human brucellosis followed by switching towards Th2 along prolongation of disease. J Infect 53:315–324
Elfaki MG, Al-Hokail AA (2009) Transforming growth factor β production correlates with depressed lymphocytes function in humans with chronic brucellosis. Microbes Infect 11:1089–1096
Maheshwari A, Kelly DR, Nicola T et al (2011) TGF-β2 suppresses macrophage cytokine production and mucosal inflammatory responses in the developing intestine. Gastroenterology 140:242–253
Wang H, Xu W, Shao Q, Ding Q (2017) miR-21 silencing ameliorates experimental autoimmune encephalomyelitis by promoting the differentiation of IL-10-producing B cells. Oncotarget 8:94069
Xavier MN, Winter MG, Spees AM et al (2013) CD4+ T cell-derived IL-10 promotes Brucella abortus persistence via modulation of macrophage function. PLoS Pathog 9:e1003454
Wang Z, Brandt S, Medeiros A et al (2015) MicroRNA 21 is a homeostatic regulator of macrophage polarization and prevents prostaglandin E2-mediated M2 generation. PLoS ONE 10:e0115855
Yao T, Lin Z (2012) MiR-21 is involved in cervical squamous cell tumorigenesis and regulates CCL20. Biochim Biophys Acta Mol Basis Dis 1822:248–260
Acknowledgements
We acknowledge the support from Hamadan University of Medical Science.
Funding
This study was funded by Hamadan University of Medical Sciences (Grant Number: # 9607044220), Hamadan, Iran.
Author information
Authors and Affiliations
Contributions
MR: Investigation, conducting of the experiments, analysis, writing. FK: Brucellosis diagnosis and introducing patients. SJ: Investigation, conducting of the experiments. MR: Investigation, conducting of the experiments. AL: Editing and analysis. MH: Project administration, funding acquisition GS: Supervision, Review, Editing.
Corresponding authors
Ethics declarations
Conflict of interests
The authors declare no competing interests.
Ethical approval
This study was approved by the Ethics Committee of Hamadan University of Medical Sciences (No: IR.UMSHA.REC.1396.441).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11033_2023_9193_MOESM1_ESM.tif
Supplementary file1 (TIF 97 KB)—The network was designed using the STRING11.0 database. Red nodes represent proteins with a negative regulatory role in cytokine production by macrophage. Blue nodes represent related molecules that have roles in the negative regulation of cytokines. Green, yellow, and purple nodes were used for indicating molecules that participate in negative regulation of immune response, immune effector process, and inflammatory responses, respectively.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rezaeepoor, M., Keramat, F., Jourghasemi, S. et al. MicroRNA -21 expression as an auxiliary diagnostic biomarker of acute brucellosis. Mol Biol Rep 51, 264 (2024). https://doi.org/10.1007/s11033-023-09193-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11033-023-09193-8