Abstract
Background
Chronic kidney disease (CKD) is a serious health threat worldwide. Defective mitophagy has been reported to induce mitochondrial dysfunction, which is closely associated with CKD pathogenesis. Honokiol (HKL) is a bioactive component of Magnolia officinalis that has multiple efficacies. Our study aimed to investigate the effect of HKL on a CKD rat model and explore the possible mechanisms of mitophagy mediated by Bcl-2 interacting protein 3 and BNIP3-like (NIX) (also known as the BNIP3/NIX pathway) and FUN14 domain-containing 1 (the FUNDC1 pathway) and the role of the AMP-activated protein kinase (AMPK) pathway.
Methods
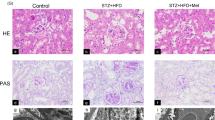
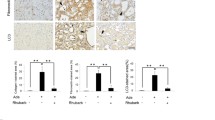
A CKD rat model was established by feeding the animals dietary adenine (0.75% w/w, 3 weeks). Simultaneously, the treatment group was given HKL (5 mg/kg/day, 4 weeks) by gavage. Renal function was assessed by measuring serum creatinine (Scr) and blood urea nitrogen (BUN) levels. Pathological changes were analyzed by periodic acid-Schiff (PAS) and Masson’s trichrome staining. Protein expression was evaluated by Western blotting and immunohistochemistry.
Results
HKL treatment ameliorated the decline in renal function and reduced tubular lesions and interstitial fibrosis in CKD rats. Accordingly, the renal fibrosis markers Col-IV and α-SMA were decreased by HKL. Moreover, HKL suppressed the upregulation of the proapoptotic proteins Bad and Bax and Cleaved caspase-3 expression in CKD rats. Furthermore, HKL suppressed BNIP3, NIX and FUNDC1 expression, leading to the reduction of excessive mitophagy in CKD rats. Additionally, AMPK was activated by adenine, and HKL reversed this change and significantly decreased the level of activated AMPK (phosphorylated AMPK, P-AMPK).
Conclusion
HKL exerted a renoprotective effect on CKD rats, which was possibly associated with BNIP3/NIX and FUNDC1-mediated mitophagy and the AMPK pathway.






Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AKI:
-
Acute kidney injury
- AMPK:
-
AMP-activated protein kinase
- BNIP3:
-
Bcl-2 interacting protein 3
- BUN:
-
Blood urea nitrogen
- CKD:
-
Chronic kidney disease
- Col-IV:
-
Type IV collagen
- DALYs:
-
Disability-adjusted life years
- ESRD:
-
End-stage renal disease
- FUNDC1:
-
FUN14 domain containing 1
- HKL:
-
Honokiol
- I/R:
-
Ischemia–reperfusion
- NIX:
-
BNIP3-like
- PAS:
-
Periodic acid-Schiff
- PINK1:
-
Phosphatase with tensin homolog (PTEN)-induced kinase 1
- RASIs:
-
Renin-angiotensin system inhibitors
- Scr:
-
Serum creatinine
- TCM:
-
Traditional Chinese medicine
References
Belousov DM, Mikhaylenko EV, Somasundaram SG et al (2020) Global, regional, and national burden of chronic kidney disease, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 395:709–733. https://doi.org/10.1016/S0140-6736(20)30045-3
Aashima M, Nanda RS, Jani C (2022) The burden of chronic kidney disease in Asia, 1990–2019: examination of estimates from global burden of disease 2019 study. Nephrology 27:610–620. https://doi.org/10.1111/nep.14051
Liyanage T, Ninomiya T, Jha V et al (2015) Worldwide access to treatment for end-stage kidney disease: a systematic review. Lancet 385:1975–1982. https://doi.org/10.1016/S0140-6736(14)61601-9
Xie Y, Bowe B, Mokdad AH et al (2018) Analysis of the Global Burden of Disease study highlights the global, regional, and national trends of chronic kidney disease epidemiology from 1990 to 2016. Kidney Int 94:567–581. https://doi.org/10.1016/j.kint.2018.04.011
Foreman KJ, Marquez N, Dolgert A et al (2018) Forecasting life expectancy, years of life lost, and all-cause and cause-specific mortality for 250 causes of death: reference and alternative scenarios for 2016–40 for 195 countries and territories. Lancet 392:2052–2090. https://doi.org/10.1016/S0140-6736(18)31694-5
Rauf A, Olatunde A, Imran M et al (2021) Honokiol: a review of its pharmacological potential and therapeutic insights. Phytomedicine 92:153769. https://doi.org/10.1016/j.phymed.2021.153647
Fried LE, Arbiser JL (2009) Honokiol, a multifunctional antiangiogenic and antitumor agent. Antioxid Redox Signal 11:1139–1148. https://doi.org/10.1089/ars.2009.2440
Debsharma S, Pramanik S, Bindu S, Bandyopadhyay U et al (2023) Honokiol, an inducer of Sirtuin 3, protects against NSAID-induced gastric mucosal mitochondrial pathology, apoptosis and inflammatory tissue injury. Br J Pharmacol 2023:14765381. https://doi.org/10.1111/bph.1607010
Huang K-J, Kuo C-H, Chen S-H et al (2018) Honokiol inhibits in vitro and in vivo growth of oral squamous cell carcinoma through induction of apoptosis, cell cycle arrest and autophagy. J Cell Mol Med 22:1894–1908. https://doi.org/10.1111/jcmm.13474
Pillai VB, Zhou Y, Tang J et al (2023) Honokiol alleviated neurodegeneration by reducing oxidative stress and improving mitochondrial function in mutant SOD1 cellular and mouse models of amyotrophic lateral sclerosis. Acta Pharm Sin B 13:577–597. https://doi.org/10.1016/j.apsb.2022.07.019
Chen C, Zhang Q-W, Ye Y, Lin L-G (2021) Honokiol: a naturally occurring lignan with pleiotropic bioactivities. Chin J Nat Med 19:481–490. https://doi.org/10.1016/S1875-5364(21)60047-X
Yi X, Guo W, Shi Q et al (2019) SIRT3-dependent mitochondrial dynamics remodeling contributes to oxidative stress-induced melanocyte degeneration in vitiligo. Theranostics 9:1614–1633. https://doi.org/10.7150/thno.30398
Ye J-S, Chen L, Lu Y-Y et al (2019) Honokiol-mediated mitophagy ameliorates postoperative cognitive impairment induced by surgery/sevoflurane via inhibiting the activation of NLRP3 inflammasome in the hippocampus. Oxid Med Cell Longev 2019:8639618. https://doi.org/10.1155/2019/8639618
Mao R, He S, Lan J, Zhu W (2022) Honokiol ameliorates cisplatin-induced acute kidney injury via inhibition of mitochondrial fission. Br J Pharmacol 179:3886–3904. https://doi.org/10.1111/bph.15837
Park EJ, Dusabimana T, Je J et al (2020) Honokiol protects the kidney from renal ischemia and reperfusion injury by upregulating the glutathione biosynthetic enzymes. Biomedicines 8:352. https://doi.org/10.3390/biomedicines8090352
Tang C, He L, Liu J, Dong Z (2015) Mitophagy: basic mechanism and potential role in kidney diseases. Kidney Dis 1:71–79. https://doi.org/10.1159/000381510
Belousov DM, Mikhaylenko EV, Somasundaram SG et al (2020) The dawn of mitophagy: what do we know by now?”. CN 19:170–192. https://doi.org/10.2174/1570159X18666200522202319
Narendra DP (2021) Managing risky assets—mitophagy in vivo. J Cell Sci 134:jcs240465. https://doi.org/10.1242/jcs.240465
Tang C, Livingston MJ, Liu Z, Dong Z (2020) Autophagy in kidney homeostasis and disease. Nat Rev Nephrol 16:489–508. https://doi.org/10.1038/s41581-020-0309-2
Kubli DA, Ycaza JE, Gustafsson AB (2007) Bnip3 mediates mitochondrial dysfunction and cell death through Bax and Bak. Biochem J 405:407–415. https://doi.org/10.1042/BJ20070319
Wang D-X, Yang Y, Huang X-S et al (2021) Pramipexole attenuates neuronal injury in Parkinson’s disease by targeting miR-96 to activate BNIP3-mediated mitophagy. Neurochem Int 146:104972. https://doi.org/10.1016/j.neuint.2021.104972
Lampert MA, Orogo AM, Najor RH et al (2019) BNIP3L/NIX and FUNDC1-mediated mitophagy is required for mitochondrial network remodeling during cardiac progenitor cell differentiation. Autophagy 15:1182–1198. https://doi.org/10.1080/15548627.2019.1580095
Huang C, Yi H, Shi Y et al (2021) KCa3.1 mediates dysregulation of mitochondrial quality control in diabetic kidney disease. Front. Cell Dev. Biol 9:573814. https://doi.org/10.3389/fcell.2021.573814
Tang C, Han H, Liu Z et al (2019) Activation of BNIP3-mediated mitophagy protects against renal ischemia–reperfusion injury. Cell Death Dis 10:677. https://doi.org/10.1038/s41419-019-1899-0
Lu Y, Zhang C, Chen J et al (2022) Hypothermia preconditioning improves cardiac contractility after cardiopulmonary resuscitation through AMPK-activated mitophagy. Exp Biol Med (Maywood) 2022:153537022210815. https://doi.org/10.1177/15353702221081546
Iorio R, Celenza G, Petricca S (2021) Mitophagy: molecular mechanisms, new concepts on parkin activation and the emerging role of AMPK/ULK1 axis. Cells 11:30. https://doi.org/10.3390/cells11010030
Wei X, Wang Y, Weng J et al (2022) Combination of perindopril erbumine and Huangqi-Danshen decoction protects against chronic kidney disease via Sirtuin3/mitochondrial dynamics pathway. Evid Based Complement Altern Med 2022:5812105. https://doi.org/10.1155/2022/5812105
Elfeky MG, Mantawy EM, Gad AM et al (2020) Mechanistic aspects of antifibrotic effects of honokiol in Con A-induced liver fibrosis in rats: emphasis on TGF-β/SMAD/MAPK signaling pathways. Life Sci 240:117096. https://doi.org/10.1016/j.lfs.2019.117096
Liu X, Gao L, Huang X, Deng R et al (2022) Lipidomics reveals the potential mechanism of honokiol against adenine-induced chronic kidney disease. Front Pharmacol 13:1019629. https://doi.org/10.3389/fphar.2022.1019629
Cortes AL, Gonsalez SR, Rioja LS et al (2018) Protective outcomes of low-dose doxycycline on renal function of Wistar rats subjected to acute ischemia/reperfusion injury. Biochim Biophys Acta (BBA) Mol Basis Dis 1864:102–114. https://doi.org/10.1016/j.bbadis.2017.10.005
Zhao W, Zhao T, Chen Y et al (2018) Differential expression of hypertensive phenotypes in BXD mouse strains in response to angiotensin II. Am J Hypertens 31:108–114. https://doi.org/10.1093/ajh/hpx144
Schindelin J, Arganda-Carreras I, Frise E et al (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682. https://doi.org/10.1038/nmeth.2019
Dumont L, Levacher N, Schapman D et al (2021) IHC_Tool: an open-source Fiji procedure for quantitative evaluation of cross sections of testicular explants. Reproduct Biol 21:100507. https://doi.org/10.1016/j.repbio.2021.100507
Su L, Zhang J, Gomez H et al (2022) Mitochondria ROS and mitophagy in acute kidney injury. Autophagy 2022:1–14. https://doi.org/10.1080/15548627.2022.2084862
Liang Q, Wan J, Liu H et al (2022) A plant nonenveloped double-stranded RNA virus activates and co-opts BNIP3-mediated mitophagy to promote persistent infection in its insect vector. Autophagy 2022:1–16. https://doi.org/10.1080/15548627.2022.2091904
Yu Y, Li M, Su N et al (2016) Honokiol protects against renal ischemia/reperfusion injury via the suppression of oxidative stress, iNOS, inflammation and STAT3 in rats. Mol Med Rep 13:1353–1360. https://doi.org/10.3892/mmr.2015.4660
Li N, Xie H, Li L et al (2014) Effects of honokiol on sepsis-induced acute kidney injury in an experimental model of sepsis in rats. Inflammation 37:1191–1199. https://doi.org/10.1007/s10753-014-9845-x
Sowter HM, Ratcliffe PJ, Watson P et al (2001) HIF-1-dependent regulation of hypoxic induction of the cell death factors BNIP3 and NIX in human tumors. Cancer Res 61:6669–6673 (PMID: 11559532)
Okami J, Simeone DM, Logsdon CD (2004) Silencing of the hypoxia-inducible cell death protein BNIP3 in pancreatic cancer. Cancer Res 64:5338–5346. https://doi.org/10.1158/0008-5472.CAN-04-0089
Fei P, Wang W, Kim S et al (2004) Bnip3L is induced by p53 under hypoxia, and its knockdown promotes tumor growth. Cancer Cell 6:597–609. https://doi.org/10.1016/j.ccr.2004.10.012
Koncsos G, Varga ZV, Baranyai T et al (2016) Diastolic dysfunction in prediabetic male rats: role of mitochondrial oxidative stress. Am J Physiol Heart Circ Physiol 311:H927–H943. https://doi.org/10.1152/ajpheart.00049.2016
Nair S, Leverin AL, Rocha-Ferreira E et al (2022) Induction of mitochondrial fragmentation and mitophagy after neonatal hypoxia-ischemia. Cells 11:1193. https://doi.org/10.3390/cells11071193
Wen W, Yu G, Liu W et al (2019) Silencing FUNDC1 alleviates chronic obstructive pulmonary disease by inhibiting mitochondrial autophagy and bronchial epithelium cell apoptosis under hypoxic environment. J Cell Biochem 120:17602–17615. https://doi.org/10.1002/jcb.29028
Su SH, Wu YF, Wang DP, Hai J (2018) Inhibition of excessive autophagy and mitophagy mediates neuroprotective effects of URB597 against chronic cerebral hypoperfusion. Cell Death Dis 9:733. https://doi.org/10.1038/s41419-018-0755-y
Sassone F, Margulets V, Maraschi A et al (2015) Bcl-2/adenovirus E1B 19-kDa interacting protein (BNip3) has a key role in the mitochondrial dysfunction induced by mutant huntingtin. Hum Mol Genet 24:6530–6539. https://doi.org/10.1093/hmg/ddv362
Wei X, Wei X, Lu Z et al (2020) Activation of TRPV1 channel antagonizes diabetic nephropathy through inhibiting endoplasmic reticulum-mitochondria contact in podocytes. Metabolism 105:154182. https://doi.org/10.1016/j.metabol.2020.154182
Zhou L, Zhang L, Zhang Y et al (2019) PINK1 deficiency ameliorates cisplatin-induced acute kidney injury in rats. Front Physiol 10:1225. https://doi.org/10.3389/fphys.2019.01225
Zhang H, Liu B, Li T et al (2017) AMPK activation serves a critical role in mitochondria quality control via modulating mitophagy in the heart under chronic hypoxia. Int J Mol Med 41:69–76. https://doi.org/10.3892/ijmm.2017.3213
Juszczak F, Caron N, Mathew AV, Declèves A-E (2020) Critical role for AMPK in metabolic disease-induced chronic kidney disease. IJMS 21:7994. https://doi.org/10.3390/ijms21217994
Li C, Tan Y, Wu J et al (2020) Resveratrol improves Bnip3-related mitophagy and attenuates high-fat-induced endothelial dysfunction. Front Cell Dev Biol 8:796. https://doi.org/10.3389/fcell.2020.00796
Mao S, Tian S, Luo X et al (2021) Overexpression of PLK1 relieved the myocardial ischemia–reperfusion injury of rats through inducing the mitophagy and regulating the p-AMPK/FUNDC1 axis. Bioengineered 12:2676–2687. https://doi.org/10.1080/21655979.2021.1938500
Funding
This study was supported by the Shenzhen Science and Technology Plan Project (Grant number JCYJ20190812161001790) and Shenzhen Fund for Guangdong Provincial Highlevel Clinical Key Specialties.
Author information
Authors and Affiliations
Contributions
XW: conceptualization, investigation, data curation, writing—original draft. YW: investigation. YL: investigation. JW: investigation. RD: investigation. SL: funding acquisition. JL: resources. SY: supervision, project administration. XL: conceptualization, funding acquisition, supervision, writing—review and editing.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
All animal experiments were carried out in accordance with the National Institutes of Health guide for the care and use of Laboratory animals (NIH Publications No. 8023, revised 1978) and approved by the Ethics Committee of the Guangzhou University of Chinese Medicine.
Consent for publication
Not applicable.
Consent for participate
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wei, X., Wang, Y., Lao, Y. et al. Effects of honokiol protects against chronic kidney disease via BNIP3/NIX and FUNDC1-mediated mitophagy and AMPK pathways. Mol Biol Rep 50, 6557–6568 (2023). https://doi.org/10.1007/s11033-023-08592-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08592-1