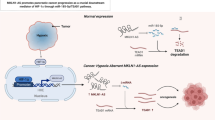
Abstract
Background
Hypoxia up-regulated 1 (HYOU1) was identified as a proto-oncogene and involved in tumorigenesis and progression in several cancer. Nonetheless, the biological function and mechanism of HYOU1 in bladder cancer (BCa) remian unclear.
Methods
The HYOU1 level in BCa tissues and cells was examined using RT-qPCR and western blot methods. The relationship between HYOU1 expression and clinicopathologic characteristics of BCa was analyzed. The biological role of HYOU1 on BCa cell proliferation, apoptosis, migration and invasion were analyzed via counting kit-8 (CCK-8), flow cytometry, wound healing and Transwell assays, respectively. The association between HYOU1 and the PI3K/AKT/Forkhead box O1 (FOXO1) signalling was assessed via western blot assay, meanwhile the the association of FOXO1 with HYOU1 was also investigated.
Results
HYOU1 was up-regulated in BCa tissues and cell lines, and the high level of HYOU1 was associated with bladder cancer histological grade and pathologic stage. Moreover, patients with high expression of HYOU1 showed poor overall survival from Kaplan-Meier Plotter. HYOU1 depletion impeded cell proliferation, migration and invasion, and induced cell apoptosis, while HYOU1 overexpression promoted cell proliferation, migration and invasion. Mechanically, our results showed that HYOU1 knockdown repressed PI3K/AKT/FOXO1 pathway and HYOU1 was negative regulated by FOXO1 in BCa. Significantly, we confirmed that the HYOU1/PI3K-AKT/FOXO1 negative feedback loop was involved in BCa cell proliferation, migration and invasion.
Conclusion
These findings revealed that HYOU1 acted as a pro-oncogene on BCa progression, and it will be a possible target for BCa treatment.






Similar content being viewed by others
Change history
18 April 2023
A Correction to this paper has been published: https://doi.org/10.1007/s11033-023-08325-4
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J Clin 68(6):394–424. https://doi.org/10.3322/caac.21492
Siegel RL, Miller KD, Jemal A (2018) Cancer statistics, 2018. CA: a cancer journal for clinicians 68. 7–30. https://doi.org/10.3322/caac.21442. 1
Rao S, Oyang L, Liang J, Yi P, Han Y, Luo X, Xia L, Lin J, Tan S, Hu J, Wang H, Tang L, Pan Q, Tang Y, Zhou Y, Liao Q (2021) Biological Function of HYOU1 in Tumors and Other Diseases. OncoTargets and therapy 14:1727–1735. https://doi.org/10.2147/OTT.S297332
Kretowski R, Stypulkowska A, Cechowska-Pasko M (2013) Low-glucose medium induces ORP150 expression and exerts inhibitory effect on apoptosis and senescence of human breast MCF7 cells. Acta Biochim Pol 60(2):167–173
Nimmo CM, Brooks PJ (1992) Process for implementing, structuring, and evaluating pharmacy practice residency experiences. Am J Hosp Pharm 49(9):2166–2174
Inoue T, Tsai B (2016) The Grp170 nucleotide exchange factor executes a key role during ERAD of cellular misfolded clients. Mol Biol Cell 27(10):1650–1662. https://doi.org/10.1091/mbc.E16-01-0033
Wang JM, Jiang JY, Zhang DL, Du X, Wu T, Du ZX (2021) HYOU1 facilitates proliferation, invasion and glycolysis of papillary thyroid cancer via stabilizing LDHB mRNA. J Cell Mol Med 25(10):4814–4825. https://doi.org/10.1111/jcmm.16453
Lee M, Song Y, Choi I, Lee SY, Kim S, Kim SH, Kim J, Seo HR (2021) Expression of HYOU1 via Reciprocal Crosstalk between NSCLC Cells and HUVECs Control Cancer Progression and Chemoresistance in Tumor Spheroids. Mol Cells 44(1):50–62. https://doi.org/10.14348/molcells.2020.0212
Hao A, Wang Y, Zhang X, Li J, Li Y, Li D, Kulik G, Sui G (2021) Long non-coding antisense RNA HYOU1-AS is essential to human breast cancer development through competitive binding hnRNPA1 to promote HYOU1 expression. Biochim et Biophys acta Mol cell Res 1868(4):118951. https://doi.org/10.1016/j.bbamcr.2021.118951
Li X, Zhang NX, Ye HY, Song PP, Chang W, Chen L, Wang Z, Zhang L, Wang NN (2019) HYOU1 promotes cell growth and metastasis via activating PI3K/AKT signaling in epithelial ovarian cancer and predicts poor prognosis. Eur Rev Med Pharmacol Sci 23(10):4126–4135. https://doi.org/10.26355/eurrev_201901_17914
Li F, Xie W, Fang Y, Xie K, Liu W, Hou L, Tan W (2021) HnRNP-F promotes the proliferation of bladder cancer cells mediated by PI3K/AKT/FOXO1. J Cancer 12(1):281–291. https://doi.org/10.7150/jca.50490
Ling J, Wang F, Liu C, Dong X, Xue Y, Jia X, Song W, Li Q (2019) FOXO1-regulated lncRNA LINC01197 inhibits pancreatic adenocarcinoma cell proliferation by restraining Wnt/beta-catenin signaling. J experimental Clin cancer research: CR 38(1):179. https://doi.org/10.1186/s13046-019-1174-3
Yang JB, Zhao ZB, Liu QZ, Hu TD, Long J, Yan K, Lian ZX (2018) FoxO1 is a regulator of MHC-II expression and anti-tumor effect of tumor-associated macrophages. Oncogene 37(9):1192–1204. https://doi.org/10.1038/s41388-017-0048-4
Zhang H, Pan Y, Zheng L, Choe C, Lindgren B, Jensen ED, Westendorf JJ, Cheng L, Huang H (2011) FOXO1 inhibits Runx2 transcriptional activity and prostate cancer cell migration and invasion. Cancer Res 71(9):3257–3267. https://doi.org/10.1158/0008-5472.CAN-10-2603
Liu JQ, Zhang L, Yao J, Yao S, Yuan T (2018) AMPK alleviates endoplasmic reticulum stress by inducing the ER-chaperone ORP150 via FOXO1 to protect human bronchial cells from apoptosis. Biochem Biophys Res Commun 497(2):564–570. https://doi.org/10.1016/j.bbrc.2018.02.095
Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J (2016) Cancer statistics in China, 2015. Cancer J Clin 66(2):115–132. https://doi.org/10.3322/caac.21338
Fresno Vara JA, Casado E, de Castro J, Cejas P, Belda-Iniesta C, Gonzalez-Baron M (2004) PI3K/Akt signalling pathway and cancer. Cancer Treat Rev 30(2):193–204. https://doi.org/10.1016/j.ctrv.2003.07.007
Calderaro J, Rebouissou S, de Koning L, Masmoudi A, Herault A, Dubois T, Maille P, Soyeux P, Sibony M, de la Taille A, Vordos D, Lebret T, Radvanyi F, Allory Y (2014) PI3K/AKT pathway activation in bladder carcinogenesis. Int J Cancer 134(8):1776–1784. https://doi.org/10.1002/ijc.28518
Sun CH, Chang YH, Pan CC (2011) Activation of the PI3K/Akt/mTOR pathway correlates with tumour progression and reduced survival in patients with urothelial carcinoma of the urinary bladder. Histopathology 58(7):1054–1063. https://doi.org/10.1111/j.1365-2559.2011.03856.x
Mitsudome A, Takeshita S, Ohfu M, Uchida T, Ogata H, Irie K (1988) [Analysis of normal F-wave of the tibial nerve in childhood]. No to hattatsu = Brain and development 20(1):76–78
Park C, Cha HJ, Lee H, Hwang-Bo H, Ji SY, Kim MY, Hong SH, Jeong JW, Han MH, Choi SH, Jin CY, Kim GY, Choi YH (2019) Induction of G2/M Cell Cycle Arrest and Apoptosis by Genistein in Human Bladder Cancer T24 Cells through Inhibition of the ROS-Dependent PI3k/Akt Signal Transduction Pathway. Antioxidants 8(9). https://doi.org/10.3390/antiox8090327
Yao J, Qian K, Chen C, Liu X, Yu D, Yan X, Liu T, Li S (2020) ZNF139/circZNF139 promotes cell proliferation, migration and invasion via activation of PI3K/AKT pathway in bladder cancer. Aging 12(10):9915–9934. https://doi.org/10.18632/aging.103256
Mao XP, Zhang LS, Huang B, Zhou SY, Liao J, Chen LW, Qiu SP, Chen JX (2015) Mir-135a enhances cellular proliferation through post-transcriptionally regulating PHLPP2 and FOXO1 in human bladder cancer. J translational Med 13:86. https://doi.org/10.1186/s12967-015-0438-8
Wang Y, Zhou Y, Graves DT (2014) FOXO transcription factors: their clinical significance and regulation. Biomed Res Int 2014:925350. https://doi.org/10.1155/2014/925350
Zhu J, Li Y, Tian Z, Hua X, Gu J, Li J, Liu C, Jin H, Wang Y, Jiang G, Huang H, Huang C (2017) ATG7 Overexpression Is Crucial for Tumorigenic Growth of Bladder Cancer In Vitro and In Vivo by Targeting the ETS2/miRNA196b/FOXO1/p27 Axis. Mol therapy Nucleic acids 7:299–313. https://doi.org/10.1016/j.omtn.2017.04.012
Lloreta J, Font-Tello A, Juanpere N, Frances A, Lorenzo M, Nonell L, de Muga S, Vazquez I, Cecchini L, Hernandez-Llodra S (2017) FOXO1 down-regulation is associated with worse outcome in bladder cancer and adds significant prognostic information to p53 overexpression. Hum Pathol 62:222–231. https://doi.org/10.1016/j.humpath.2016.12.022
Ide H, Mizushima T, Jiang G, Goto T, Nagata Y, Teramoto Y, Inoue S, Li Y, Kashiwagi E, Baras AS, Netto GJ, Kawahara T, Miyamoto H (2020) FOXO1 as a tumor suppressor inactivated via AR/ERbeta signals in urothelial cells. Endocrine-related Cancer 27(4):231–244. https://doi.org/10.1530/ERC-20-0004
Ide H, Goto T, Teramoto Y, Mizushima T, Jiang G, Nagata Y, Inoue S, Baras AS, Kashiwagi E, Miyamoto H (2020) FOXO1 inactivation induces cisplatin resistance in bladder cancer. Cancer Sci 111(9):3397–3400. https://doi.org/10.1111/cas.14557
Wang L, Yang C, Chu M, Wang ZW, Xue B (2021) Cdc20 induces the radioresistance of bladder cancer cells by targeting FoxO1 degradation. Cancer Lett 500:172–181. https://doi.org/10.1016/j.canlet.2020.11.052
Song G, Ouyang G, Bao S (2005) The activation of Akt/PKB signaling pathway and cell survival. J Cell Mol Med 9(1):59–71. https://doi.org/10.1111/j.1582-4934.2005.tb00337.x
Wang Y, Wu Z, Li D, Wang D, Wang X, Feng X, Xia M (2011) Involvement of oxygen-regulated protein 150 in AMP-activated protein kinase-mediated alleviation of lipid-induced endoplasmic reticulum stress. J Biol Chem 286(13):11119–11131. https://doi.org/10.1074/jbc.M110.203323
Funding
None.
Author information
Authors and Affiliations
Contributions
CC supervised the whole project, designed the experiments, and analyzed the data. WW performed the experiments, analyzed the data, wrote the manuscript. JX and XF contributed to the experiments and analyzed the data. CX prepared the figures. LG, LL, XQ, and ZM contributed to the experiments and provided technical support.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest in this work.
Ethics approval
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the institutional ethics committee of Xiangya Changde Hospital (approval No. 201812628).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent to publish
The authors affirm that human research participants provided informed consent for publication of the images in Fig. 1 A and 1B.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The figures 1-6 are incorrectly sequenced. This error has been rectified.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, W., Jiang, X., Xia, F. et al. HYOU1 promotes cell proliferation, migration, and invasion via the PI3K/AKT/FOXO1 feedback loop in bladder cancer. Mol Biol Rep 50, 453–464 (2023). https://doi.org/10.1007/s11033-022-07978-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07978-x