Abstract
Background
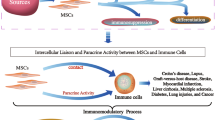
The immunomodulatory function of mesenchymal stem cells (MSCs) has been considered to be vital for MSC-based therapies. Many works have been devoted to excavate effective strategies for enhancing the immunomodulation effect of MSCs. Nonetheless, canine MSC-mediated immunomodulation is still poorly understood.
Methods and results
The inflammatory microenvironment was simulated through the employment of interferon-γ (IFN-γ) in a culture system. Compared with unstimulated cBMSCs, IFN-γ stimulation increased the mRNA levels of Toll-like receptor 3 (TLR3) and indoleamine 2, 3-dioxygenase 1 (IDO-1), and simultaneously enhanced the secretion of immunosuppressive molecules, including interleukin (IL)-10, hepatocyte growth factor (HGF), and kynurenine in cBMSCs. IFN-γ stimulation significantly enhanced the ability of cBMSCs and their supernatant to suppress the proliferation of murine spleen lymphocytes. Lymphocyte subtyping evaluation revealed that cBMSCs and their supernatant diminished the percentage of CD3+CD4+ and CD3+CD8+ lymphocytes compared with the control group, with a decreasing CD4+/CD8+ ratio. Notably, exposure to IFN-γ decreased the CD4+/CD8+ ratio more effectively than unstimulated cells or supernatant. Additionally, IFN-γ-stimulation increased the mRNA levels of the Th1 cytokines TNF-α, and remarkably decreased the mRNA level of the Th2 cytokine IL-4 and IL-10.
Conclusion
Our findings substantiate that IFN-γ stimulation can enhance the immunomodulatory properties of cBMSCs by promoting TLR3-dependent activation of the IDO/kynurenine pathway, increasing the secretion of immunoregulatory molecules and strengthening interactions with T lymphocytes, which may provide a meaningful strategy for the clinical application of cBMSCs in immune-related diseases.





Similar content being viewed by others
Data availability
Data are available from the corresponding author upon reasonable request.
References
Zhan XS, El-Ashram S, Luo DZ, Luo HN, Wang BY, Chen SF, Bai YS, Chen ZS, Liu CY, Ji HQ (2019) A Comparative study of biological characteristics and transcriptome profiles of mesenchymal stem cells from different canine tissues. Int J Mol Sci 20:1485. https://doi.org/10.3390/ijms20061485
Chen Y, Zeng Z, Sun J, Zeng H, Huang Y, Zhang Z (2015) Mesenchymal stem cell–conditioned medium prevents radiation-induced liver injury by inhibiting inflammation and protecting sinusoidal endothelial cells. J Radiat Res 56:700–708. https://doi.org/10.1093/jrr/rrv026
Samsonraj RM, Raghunath M, Nurcombe V, Hui JH, van Wijnen AJ, Cool SM (2017) Concise review: multifaceted characterization of human mesenchymal stem cells for use in regenerative medicine. Stem Cell Transl Med 6:2173–2185. https://doi.org/10.1002/sctm.17-0129
Ren G, Chen X, Dong F, Li W, Ren X, Zhang Y, Shi Y (2012) Concise review: mesenchymal stem cells and translational medicine: emerging issues. Stem Cells Transl Med 1:51–58. https://doi.org/10.5966/sctm.2011-0019
Glenn JD, Whartenby KA (2014) Mesenchymal stem cells: emerging mechanisms of immunomodulation and therapy. World J Stem Cells 6:526–539. https://doi.org/10.4252/wjsc.v6.i5.526
Kim HW, Song WJ, Li Q, Han SM, Jeon KO, Park SC, Ryu MO, Chae HK, Kyeong K, Youn HY (2016) Canine adipose tissue-derived mesenchymal stem cells ameliorate severe acute pancreatitis by regulating T cells in rats. J Vet Sci 17:539–548. https://doi.org/10.4142/jvs.2016.17.4.539
Li N, Hua J (2017) Interactions between mesenchymal stem cells and the immune system. Cell Mol Life Sci 74:2345–2360. https://doi.org/10.1007/s00018-017-2473-5
Zhou Y, Yamamoto Y, Xiao Z, Ochiya T (2019) The Immunomodulatory functions of mesenchymal stromal/stem cells mediated via paracrine activity. J Clin Med 8:1025. https://doi.org/10.3390/jcm8071025
Huaman O, Bahamonde J, Cahuascanco B, Jervis M, Palomino J, Torres CG, Peralta OA (2019) Immunomodulatory and immunogenic properties of mesenchymal stem cells derived from bovine fetal bone marrow and adipose tissue. Res Vet Sci 124:212–222. https://doi.org/10.1016/j.rvsc.2019.03.017
Castro-Manrreza ME, Montesinos JJ (2015) Immunoregulation by mesenchymal stem cells: biological aspects and clinical applications. J Immunol Res 2015:394917. https://doi.org/10.1155/2015/394917
Hawkins KE, Corcelli M, Dowding K, Ranzoni AM, Vlahova F, Hau KL, Hunjan A, Peebles D, Gressens P, Hagberg H, de Coppi P, Hristova M, Guillot PV (2018) Embryonic stem cell-derived Mesenchymal Stem Cells (MSCs) have a superior neuroprotective capacity over fetal MSCs in the hypoxic-ischemic mouse brain. Stem Cells Transl Med 7:439–449. https://doi.org/10.1002/sctm.17-0260
Chen X, Xu CX, Liang H, Xi Z, Pan J, Yang Y, Sun Q, Yang G, Sun Y, Bian L (2020) Bone marrow mesenchymal stem cells transplantation alleviates brain injury after intracerebral hemorrhage in mice through the Hippo signaling pathway. Aging 12:6306–6323. https://doi.org/10.18632/aging.103025
Song WJ, Li Q, Ryu MO, Ahn JO, Bhang DH, Jung YC, Youn HY (2018) TSG-6 released from intraperitoneally injected canine adipose tissue-derived mesenchymal stem cells ameliorate inflammatory bowel disease by inducing M2 macrophage switch in mice. Stem Cell Res Ther 9:91. https://doi.org/10.1186/s13287-018-0841-1
Cabezas J, Rojas D, Wong Y, Telleria F, Manriquez J, Mancanares ACF, Rodriguez-Alvarez LL, Castro FO (2020) In vitro preconditioning of equine adipose mesenchymal stem cells with prostaglandin E2, substance P and their combination changes the cellular protein secretomics and improves their immunomodulatory competence without compromising stemness. Vet Immunol Immunopathol 228:110100. https://doi.org/10.1016/j.vetimm.2020.110100
Song WJ, Li Q, Ryu MO, Nam A, An JH, Jung YC, Ahn JO, Youn HY (2019) Canine adipose tissue-derived mesenchymal stem cells pre-treated with TNF-alpha enhance immunomodulatory effects in inflammatory bowel disease in mice. Res Vet Sci 125:176–184. https://doi.org/10.1016/j.rvsc.2019.06.012
Krampera M, Cosmi L, Angeli R, Pasini A, Liotta F, Andreini A, Santarlasci V, Mazzinghi B, Pizzolo G, Vinante F, Romagnani P, Maggi E, Romagnani S, Annunziato F (2006) Role for interferon-gamma in the immunomodulatory activity of human bone marrow mesenchymal stem cells. Stem Cells 24:386–398. https://doi.org/10.1634/stemcells.2005-0008
Boland L, Burand AJ, Brown AJ, Boyt D, Lira VA, Ankrum JA (2018) IFN-gamma and TNF-alpha Pre-licensing protects mesenchymal stromal cells from the pro-inflammatory effects of palmitate. Mol Ther 26:860–873. https://doi.org/10.1016/j.ymthe.2017.12.013
Dang S, Xu H, Xu C, Cai W, Li Q, Cheng Y, Jin M, Wang RX, Peng Y, Zhang Y, Wu C, He X, Wan B, Zhang Y (2014) Autophagy regulates the therapeutic potential of mesenchymal stem cells in experimental autoimmune encephalomyelitis. Autophagy 10:1301–1315. https://doi.org/10.4161/auto.28771
Jie L, Lipeng B, Xi C, Fang Y, Liuhong S, Suizhong C, Zhicai Z, Zhihua R, Miaoping M, Shumin Y (2017) Isolation, cultivation and identification of canine bone marrow mesenchymal stem cells ( BMSCs) in vitro. Acta Agric Zhejiangensis 29:751–759. https://doi.org/10.3969/j.issn.1004-1524.2017.05.10
Hu CD, Kosaka Y, Marcus P, Rashedi I, Keating A (2019) Differential immunomodulatory effects of human bone marrow-derived mesenchymal stromal cells on natural killer cells. Stem Cells Dev 28:933–943. https://doi.org/10.1089/scd.2019.0059
Xin Y, Gao J, Hu R, Li H, Li Q, Han F, He Z, Lai L, Su M (2020) Changes of immune parameters of T lymphocytes and macrophages in EAE mice after BM-MSCs transplantation. Immunol Lett 225:66–73. https://doi.org/10.1016/j.imlet.2020.05.005
Deng J, Ouyang P, Li W, Zhong L, Gu C, Shen L, Cao S, Yin L, Ren Z, Zuo Z, Deng J, Yan Q, Yu S (2021) Curcumin alleviates the senescence of canine bone marrow mesenchymal stem cells during in vitro expansion by activating the autophagy pathway. Int J Mol Sci 22:11356. https://doi.org/10.3390/ijms222111356
Haque N, Kasim NH, Rahman MT (2015) Optimization of pre-transplantation conditions to enhance the efficacy of mesenchymal stem cells. Int J Biol Sci 11:324–334. https://doi.org/10.7150/ijbs.10567
Lian Q, Zhang Y, Liang X, Gao F, Tse H-F (2016) Directed Differentiation of Human-Induced Pluripotent Stem Cells to Mesenchymal Stem Cells. In: Gnecchi M (ed) Mesenchymal Stem Cells: Methods and Protocols. Springer, New York, pp 289–298. https://doi.org/10.1007/978-1-4939-3584-0_17
Sun YQ, Zhang Y, Li X, Deng MX, Gao WX, Yao Y, Chiu SM, Liang X, Gao F, Chan CW, Tse HF, Shi J, Fu QL, Lian Q (2015) Insensitivity of human iPS cells-derived mesenchymal stem cells to interferon-gamma-induced HLA expression potentiates repair efficiency of hind limb ischemia in immune humanized NOD scid gamma mice. Stem Cells 33:3452–3467. https://doi.org/10.1002/stem.2094
Delarosa O, Dalemans W, Lombardo E (2012) Toll-like receptors as modulators of mesenchymal stem cells. Front Immunol 3:182. https://doi.org/10.3389/fimmu.2012.00182
Cassano JM, Schnabel LV, Goodale MB, Fortier LA (2018) The immunomodulatory function of equine MSCs is enhanced by priming through an inflammatory microenvironment or TLR3 ligand. Vet Immunol Immunopathol 195:33–39. https://doi.org/10.1016/j.vetimm.2017.10.003
Jafari M, Asghari A, Delbandi AA, Jalessi M, Jazayeri MH, Samarei R, Tajik N (2020) Priming TLR3 and TLR4 in human adipose- and olfactory mucosa-derived mesenchymal stromal cells and comparison of their cytokine secretions. Cytotechnology 72:57–68. https://doi.org/10.1007/s10616-019-00357-8
Li Y, Huang L, Cai Z, Deng W, Wang P, Su H, Wu Y, Shen H (2019) A study of the immunoregulatory function of TLR3 and TLR4 on mesenchymal stem cells in ankylosing spondylitis. Stem Cells Dev 28:1398–1412. https://doi.org/10.1089/scd.2019.0039
Opitz CA, Litzenburger UM, Lutz C, Lanz TV, Tritschler I, Koppel A, Tolosa E, Hoberg M, Anderl J, Aicher WK, Weller M, Wick W, Platten M (2009) Toll-like receptor engagement enhances the immunosuppressive properties of human bone marrow-derived mesenchymal stem cells by inducing indoleamine-2,3-dioxygenase-1 via interferon-beta and protein kinase R. Stem Cells 27:909–919. https://doi.org/10.1002/stem.7
Liang C, Jiang E, Yao J, Wang M, Chen S, Zhou Z, Zhai W, Ma Q, Feng S, Han M (2018) Interferon-gamma mediates the immunosuppression of bone marrow mesenchymal stem cells on T-lymphocytes in vitro. Hematology 23:44–49. https://doi.org/10.1080/10245332.2017.1333245
Kim DS, Jang IK, Lee MW, Ko YJ, Lee DH, Lee JW, Sung KW, Koo HH, Yoo KH (2018) Enhanced immunosuppressive properties of human mesenchymal stem cells primed by interferon-gamma. EBioMedicine 28:261–273. https://doi.org/10.1016/j.ebiom.2018.01.002
Waterman RS, Tomchuck SL, Henkle SL, Betancourt AM (2010) A New Mesenchymal Stem Cell (MSC) Paradigm: Polarization into a Pro-Inflammatory MSC1 or an Immunosuppressive MSC2 Phenotype. PLoS One 5:e10088. https://doi.org/10.1371/journal.pone.0010088
Kim KD, Lim HY, Lee HG, Yoon DY, Choe YK, Choi I, Paik SG, Kim YS, Yang Y, Lim JS (2005) Apolipoprotein A-I induces IL-10 and PGE2 production in human monocytes and inhibits dendritic cell differentiation and maturation. Biochem Biophys Res Commun 338:1126–1136. https://doi.org/10.1016/j.bbrc.2005.10.065
Ryan JM, Barry F, Murphy JM, Mahon BP (2007) Interferon-gamma does not break, but promotes the immunosuppressive capacity of adult human mesenchymal stem cells. Clin Exp Immunol 149:353–363. https://doi.org/10.1111/j.1365-2249.2007.03422.x
Ramaswamy K, Kumar P, He YX (2000) A role for parasite-induced PGE2 in IL-10-mediated host immunoregulation by skin stage schistosomula of Schistosoma mansoni. J Immunol 165:4567–4574. https://doi.org/10.4049/jimmunol.165.8.4567
Connard SS, Linardi RL, Even KM, Berglund AK, Schnabel LV, Ortved KF (2021) Effects of continuous passage on the immunomodulatory properties of equine bone marrow-derived mesenchymal stem cells in vitro. Vet Immunol Immunopathol 234:110203. https://doi.org/10.1016/j.vetimm.2021.110203
Lu Z, Chang W, Meng S, Xu X, Xie J, Guo F, Yang Y, Qiu H, Liu L (2019) Mesenchymal stem cells induce dendritic cell immune tolerance via paracrine hepatocyte growth factor to alleviate acute lung injury. Stem Cell Res Ther 10:372. https://doi.org/10.1186/s13287-019-1488-2
Shi Y, Wang Y, Li Q, Liu K, Hou J, Shao C, Wang Y (2018) Immunoregulatory mechanisms of mesenchymal stem and stromal cells in inflammatory diseases. Nat Rev Nephrol 14:493–507. https://doi.org/10.1038/s41581-018-0023-5
Fan LX, Hu CX, Chen JJ, Cen PP, Wang J, Li LJ (2016) Interaction between mesenchymal stem cells and B-cells. Int J Mol Sci 17:650. https://doi.org/10.3390/ijms17050650
Ma D, Xu K, Zhang G, Liu Y, Gao J, Tian M, Wei C, Li J, Zhang L (2019) Immunomodulatory effect of human umbilical cord mesenchymal stem cells on T lymphocytes in rheumatoid arthritis. Int Immunopharmacol 74:105687. https://doi.org/10.1016/j.intimp.2019.105687
Meisel R, Zibert A, Laryea M, Gobel U, Daubener W, Dilloo D (2004) Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase-mediated tryptophan degradation. Blood 103:4619–4621. https://doi.org/10.1182/blood-2003-11-3909
Chen J, Zhang X, Xie J, Xue M, Liu L, Yang Y, Qiu H (2020) Overexpression of TGFbeta1 in murine mesenchymal stem cells improves lung inflammation by impacting the Th17/Treg balance in LPS-induced ARDS mice. Stem Cell Res Ther 11:311. https://doi.org/10.1186/s13287-020-01826-0
Luz-Crawford P, Kurte M, Bravo-Alegria J, Contreras R, Nova-Lamperti E, Tejedor G, Noel D, Jorgensen C, Figueroa F, Djouad F, Carrion F (2013) Mesenchymal stem cells generate a CD4+CD25+Foxp3+ regulatory T cell population during the differentiation process of Th1 and Th17 cells. Stem Cell Res Ther 4:65. https://doi.org/10.1186/scrt216
Krampera M, Glennie S, Dyson J, Scott D, Laylor R, Simpson E, Dazzi F (2003) Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood 101:3722–3729. https://doi.org/10.1182/blood-2002-07-2104
Bruno G, Saracino A, Monno L, Angarano G (2017) The revival of an “Old” marker: CD4/CD8 ratio. AIDS Rev 19:81–88
Crespo AC, van der Zwan A, Ramalho-Santos J, Strominger JL, Tilburgs T (2017) Cytotoxic potential of decidual NK cells and CD8+ T cells awakened by infections. J Reprod Immunol 119:85–90. https://doi.org/10.1016/j.jri.2016.08.001
Basu R, Whitlock BM, Husson J, Le Floc’h A, Jin WY, Oyler-Yaniv A, Dotiwala F, Giannone G, Hivroz C, Biais N, Lieberman J, Kam LC, Huse M (2016) Cytotoxic T cells use mechanical force to potentiate target cell killing. Cell 165:100–110. https://doi.org/10.1016/j.cell.2016.01.021
Duggleby R, Danby RD, Madrigal JA, Saudemont A (2018) Clinical grade regulatory CD4(+) T cells (Tregs): moving toward cellular-based immunomodulatory therapies. Front Immunol 9:252. https://doi.org/10.3389/fimmu.2018.00252
Le Blanc K, Rasmusson I, Gotherstrom C, Seidel C, Sundberg B, Sundin M, Rosendahl K, Tammik C, Ringden O (2004) Mesenchymal stem cells inhibit the expression of CD25 (interleukin-2 receptor) and CD38 on phytohaemagglutinin-activated lymphocytes. Scand J Immunol 60:307–315. https://doi.org/10.1111/j.0300-9475.2004.01483.x
Groh ME, Maitra B, Szekely E, Koc ON (2005) Human mesenchymal stem cells require monocyte-mediated activation to suppress alloreactive T cells. Exp Hematol 33:928–934. https://doi.org/10.1016/j.exphem.2005.05.002
Luckheeram RV, Zhou R, Verma AD, Xia B (2012) CD4(+)T cells: differentiation and functions. Clin Dev Immunol 2012:925135. https://doi.org/10.1155/2012/925135
Ezquer F, Ezquer M, Contador D, Ricca M, Simon V, Conget P (2012) The antidiabetic effect of mesenchymal stem cells is unrelated to their transdifferentiation potential but to their capability to restore Th1/Th2 balance and to modify the pancreatic microenvironment. Stem Cells 30:1664–1674. https://doi.org/10.1002/stem.1132
Tian J, Zhu Q, Zhang Y, Bian Q, Hong Y, Shen Z, Xu H, Rui K, Yin K, Wang S (2020) Olfactory ecto-mesenchymal stem cell-derived exosomes ameliorate experimental colitis via modulating Th1/Th17 and treg cell responses. Front Immunol 11:598322. https://doi.org/10.3389/fimmu.2020.598322
Tay SS, Plain KM, Bishop GA (2009) Role of IL-4 and Th2 responses in allograft rejection and tolerance. Curr Opin Organ Transplant 14:16–22. https://doi.org/10.1097/MOT.0b013e32831ebdf5
Acknowledgements
The authors appreciate the experimental equipment support provided by Sichuan Agricultural University. Thanks are also due to Liping Yang for taking care of the animals used in this work.
Funding
This work was funded by the dual support plan for discipline construction of Sichuan. Agricultural University (035/2121993256).
Author information
Authors and Affiliations
Contributions
JQD, DTL, and XYH: made substantial contributions to the conception and design of the study, acquisition of data, analysis and interpretation of data. JQD, DTL, XYH, WYL, FFZ, and CWG: contributed to manuscript preparation. LHS, SZC, ZHR, ZCZ, JLD, and SMY: gave critical revision for important intellectual content and final approval for the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare no conflict of interest.
Ethics approval and consent to participate
This study was approved by the Faculty Animal Care and Use Committee of Sichuan Agricultural University (Ya’an, China; Approval No.2013–028) for the use of animals in this research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Deng, J., Li, D., Huang, X. et al. Interferon-γ enhances the immunosuppressive ability of canine bone marrow-derived mesenchymal stem cells by activating the TLR3-dependent IDO/kynurenine pathway. Mol Biol Rep 49, 8337–8347 (2022). https://doi.org/10.1007/s11033-022-07648-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07648-y