Abstract
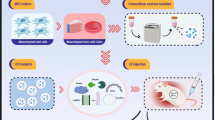
Human amniotic membrane mesenchymal stem cells-derived conditioned medium (hAM-MSCs-CM) has positive effects against myocardial ischemia/reperfusion (MI/R) injury. However, it needs further investigations how hAM-MSCs-CM leads to the cell survival under MI/R via modulation of autophagy. The purpose of this study is investigating the effects of hAM-MSCs-CM in a rat model of MI/R injury by focusing on the role of autophagy as one of its possible mechanisms. Male Wistar rats (44 rats, 175–200 g) were randomly divided into four groups: Sham, MI/R, culture media-receiving and conditioned medium-receiving. MI/R was induced by 30 min of left anterior descending coronary artery ligation. After 15 min reperfusion, culture media or hAM-MSCs-CM (150 μl) were injected intramyocardially. At the end of the experiment, CK-MB, autophagy markers, phosphorylated and total forms of mTOR and ULK1, cardiac function and fibrosis were measured. hAM-MSCs-CM significantly decreased CK-MB levels (P < 0.0001), and also the mRNA levels of Beclin1 (P < 0.0001), LC3 (P = 0.012) and p62 (P = 0.003). In addition, hAM-MSCs-CM significantly reduced Beclin1, LC3II/LC3I and p62 protein levels (P < 0.0001), and increased p-mTOR/mTOR (P = 0.022) and p-ULK1/ULK1 (P < 0.0001) expressions. Moreover, hAM-MSCs-CM improved cardiac function and decreased fibrosis (P < 0.0001). This study showed cardioprotective effects of hAM-MSCs-CM against MI/R injury through modulation of autophagy via mTOR/ULK1 pathway. Based on these findings, it can be concluded that hAM-MSCs-CM can be offered as an attractive candidate for attenuation of MI/R injury in future, but needs further investigations.





Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article [and/or] its supplementary materials.
References
Bayrami G, Karimi P, Agha-Hosseini F, Feyzizadeh S, Badalzadeh R (2018) Effect of ischemic postconditioning on myocardial function and infarct size following reperfusion injury in diabetic rats pretreated with vildagliptin. J Cardiovasc Pharmacol Ther 23:174–183
Najafi M, Noroozi E, Javadi A, Badalzadeh R (2018) Anti-arrhythmogenic and anti-inflammatory effects of troxerutin in ischemia/reperfusion injury of diabetic myocardium. Biomed Pharmacother 102:385–391
Badalzadeh R, Azimi A, Alihemmati A, Yousefi B (2017) Chronic type-I diabetes could not impede the anti-inflammatory and anti-apoptotic effects of combined postconditioning with ischemia and cyclosporine A in myocardial reperfusion injury. J Physiol Biochem 73:111–120
Bélanger M, Rodrigues PH, Dunn WA Jr, Progulske-Fox A (2006) Autophagy: a highway for Porphyromonas gingivalis in endothelial cells. Autophagy 2:165–170
Ma S, Wang Y, Chen Y, Cao F (2015) The role of the autophagy in myocardial ischemia/reperfusion injury. Biochim Biophys Acta Mol Basis Dis 1852:271–276
Timmers L et al (2008) Reduction of myocardial infarct size by human mesenchymal stem cell conditioned medium. Stem Cell Res 1:129–137
Spees JL, Lee RH, Gregory CA (2016) Mechanisms of mesenchymal stem/stromal cell function. Stem Cell Res Ther. https://doi.org/10.1186/s13287-016-0363-7
Timmers L et al (2011) Human mesenchymal stem cell-conditioned medium improves cardiac function following myocardial infarction. Stem Cell Res 6:206–214
Francisco JC et al (2013) Amniotic membrane as a potent source of stem cells and a matrix for engineering heart tissue. J Biomed Sci Eng. https://doi.org/10.4236/jbise.2013.612147
Danieli P et al (2015) Conditioned medium from human amniotic mesenchymal stromal cells limits infarct size and enhances angiogenesis. Stem Cells Transl Med 4:448–458
Yamaguchi S et al (2015) Dental pulp-derived stem cell conditioned medium reduces cardiac injury following ischemia-reperfusion. Sci Rep. https://doi.org/10.1038/srep16295
Mokhtari B et al (2020) Human amniotic membrane mesenchymal stem cells-conditioned medium attenuates myocardial ischemia-reperfusion injury in rats by targeting oxidative stress. Iran J Basic Med Sci 23:1453–1461
Razavi Tousi SMT et al (2017) A rapid and cost-effective protocol for isolating mesenchymal stem cells from the human amniotic membrane. Galen Med J 6:217–225
Pfaffl MW, Horgan GW, Dempfle L (2002) Relative expression software tool (REST©) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res. https://doi.org/10.1093/nar/30.9.e36
Obara K, Sekito T, Niimi K, Ohsumi Y (2008) The Atg18-Atg2 complex is recruited to autophagic membranes via phosphatidylinositol 3-phosphate and exerts an essential function. J Biol Chem 283:23972–23980
Xiao-Fang T, Shi-Wei Y, Yu-Jie Z (2017) Autophagy, dysglycemia and myocardial infarction. IJC Metab Endocr 14:40–44
Ma X et al (2012) Impaired autophagosome clearance contributes to cardiomyocyte death in ischemia/reperfusion injury. Circulation 125:3170–3181
Fan G et al (2016) Danshensu alleviates cardiac ischaemia/reperfusion injury by inhibiting autophagy and apoptosis via activation of mTOR signalling. J Cell Mol Med 20:1908–1919
Wang J-L et al (2020) Postconditioning with calreticulin attenuates myocardial ischemia/reperfusion injury and improves autophagic flux. Shock 53:363–372
Aoyagi T et al (2012) Cardiac mTOR protects the heart against ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol 303:H75–H85
Ouyang C, You J, Xie Z (2014) The interplay between autophagy and apoptosis in the diabetic heart. J Mol Cell Cardiol 71:71–80
Acknowledgements
This study was part of a Ph.D. thesis of Behnaz Mokhtari and financially supported by Physiology Research Center in Iran University of Medical Sciences, Tehran, Iran.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by a grant (Grant Number: 97-2-3-33054) from Iran University of Medical Sciences, Tehran-Iran.
Author information
Authors and Affiliations
Contributions
BM and NA did the study design. BM performed experimental tests, gathered and analyzed the data, and drafted the manuscript. RB contributed in interpretation of the results and finalized the manuscript editing and critically revised the manuscript. NA supervised the whole project and revised the manuscript. All gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Animal care procedures, as well as all experimental protocols were approved by the Institutional Animal Ethical Committee of Iran University of Medical Sciences (Ethical code: IR.IUMS.FMD.REC.1397.234).
Informed consent
Amniotic membranes were supplied from Shahid Akbar Abadi Hospital under informed agreement from each participants in accordance with ethical standards of the institutional and/or national research committee and with the 1975 Declaration of Helsinki as reflected in a prior approval by the institution's human research committee.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mokhtari, B., Badalzadeh, R. & Aboutaleb, N. Modulation of autophagy as the target of mesenchymal stem cells-derived conditioned medium in rat model of myocardial ischemia/reperfusion injury. Mol Biol Rep 48, 3337–3348 (2021). https://doi.org/10.1007/s11033-021-06359-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06359-0