Abstract
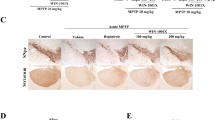
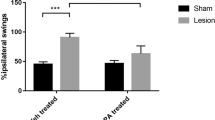
Parkinson’s disease (PD) is identified by the loss of dopaminergic neurons in the Substantia Nigra pars compacta (SNpc), and is correlated to aggregates of proteins such as α-synuclein, Lewy’s bodies. Although the PD etiology remains poorly understood, evidence suggests a main role of oxidative stress on this process. Lippia grata Schauer, known as “alecrim-do-mato”, “alecrim-de-vaqueiro”, “alecrim-da-chapada”, is a native bush from tropical areas mainly distributed throughout the Central and South America. This plant species is commonly used in traditional medicine for relief of pain and inflammation conditions, and that has proven antioxidant effects. We evaluated the effects of essential oil of the L. grata after its complexed with β-cyclodextrin (LIP) on PD animal model induced by reserpine (RES). Behavioral assessments were performed across the treatment. Upon completion the treatment, the animals were euthanized, afterwards their brains were isolated and processed for immunohistochemical and oxidative stress analysis. The LIP treatment delayed the onset of the behavior of catalepsy, decreased the number of oral movements and prevented the memory impairment on the novel object recognition task. In addition, the treatment with LIP protected against dopaminergic depletion in the SNpc and dorsal striatum (STRd), and decreased the α-syn immunoreactivity in the SNpc and hippocampus (HIP). Moreover, there was reduction of the oxidative stability index. These findings demonstrated that the LIP treatment has neuroprotective effect in a progressive parkinsonism model, suggesting that LIP could be an important source for novel treatment approaches in PD.








Similar content being viewed by others
Data availability
The datasets are available from the corresponding author on reasonable request.
References
Abilio VC, Araujo CC, Bergamo M, Calvente PR, D’Almeida V, Ribeiro Rde A, Frussa-Filho R (2003) Vitamin E attenuates reserpine-induced oral dyskinesia and striatal oxidized glutathione/reduced glutathione ratio (GSSG/GSH) enhancement in rats Progress in neuro-psychopharmacology &. Biol Psychiatry 27:109–114. doi:https://doi.org/10.1016/s0278-5846(02)00340-8
Abilio VC, Vera JAR, Ferreira LSM, Duarte CRM, Carvalho RC, Grassl C, Martins CR, Torres-Leite D, Bignotto M, Tufik S, de Ribeiro R, Frussa-Filho A (2002) R. Effects o fmelatonin on oro facial movements in rats Psychopharmacology 161:340–347
Alonso-Recio L, Martin-Plasencia P, Loeches-Alonso A, Serrano-Rodriguez JM (2014) Working memory and facial expression recognition in patients with Parkinson’s disease. J Int Neuropsychol Soc 20:496–505. doi:https://doi.org/10.1017/S1355617714000265
Alonso Canovas A et al (2014) Dopaminergic agonists in Parkinson’s disease. Neurologia 29:230–241. doi:https://doi.org/10.1016/j.nrl.2011.04.012
Alzoubi KH, Mokhemer E, Abuirmeileh AN (2018) Beneficial effect of etazolate on depression-like behavior and, learning, and memory impairment in a model of Parkinson’s disease. Behav Brain Res 350:109–115. doi:https://doi.org/10.1016/j.bbr.2018.05.004
Baig F et al (2017) Personality and addictive behaviours in early Parkinson’s disease and REM sleep behaviour disorder. Parkinsonism Relat Disord 37:72–78. doi:https://doi.org/10.1016/j.parkreldis.2017.01.017
Bertrand E, Lechowicz W, Szpak GM, Dymecki J (1997) Qualitative and quantitative analysis of locus coeruleus neurons in Parkinson’s disease. Folia Neuropathol 35:80–86
Beserra-Filho JIA et al (2019) Eplingiella fruticosa leaf essential oil complexed with beta-cyclodextrin produces a superior neuroprotective and behavioral profile in a mice model of Parkinson’s disease. Food Chem Toxicol 124:17–29. doi:https://doi.org/10.1016/j.fct.2018.11.056
Blin P et al (2015) Parkinson’s disease incidence and prevalence assessment in France using the national healthcare insurance database European. J Neurol 22:464–471. doi:https://doi.org/10.1111/ene.12592
Braak H, Del Tredici K, Rub U, de Vos RA, Jansen Steur EN, Braak E (2003) Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging 24:197–211. doi:https://doi.org/10.1016/s0197-4580(02)00065-9
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. doi:https://doi.org/10.1006/abio.1976.9999
Brandao LEM et al (2017) Passiflora cincinnata Extract Delays the Development of Motor Signs and Prevents Dopaminergic Loss in a Mice Model of Parkinson’s Disease. Evid Based Complement Alternat Med 2017:8429290. doi:https://doi.org/10.1155/2017/8429290
Brito RG, Araujo AA, Quintans JS, Sluka KA, Quintans-Junior LJ (2015) Enhanced analgesic activity by cyclodextrins - a systematic review and meta-analysis. Expert Opin Drug Deliv 12:1677–1688. doi:https://doi.org/10.1517/17425247.2015.1046835
Burre J, Sharma M, Tsetsenis T, Buchman V, Etherton MR, Sudhof TC (2010) Alpha-synuclein promotes SNARE-complex assembly in vivo and in vitro. Science 329:1663–1667. doi:https://doi.org/10.1126/science.1195227
Butler B et al (2015) Dopamine Transporter Activity Is Modulated by alpha-Synuclein. J Biol Chem 290:29542–29554. doi:https://doi.org/10.1074/jbc.M115.691592
Campelo CLC et al (2017) Exposure to an enriched environment facilitates motor recovery and prevents short-term memory impairment and reduction of striatal BDNF in a progressive pharmacological model of parkinsonism in mice. Behav Brain Res 328:138–148. doi:https://doi.org/10.1016/j.bbr.2017.04.028
Carlsson A (1959) The occurrence, distribution and physiological role of catecholamines in the nervous system. Pharmacol Rev 11:490–493
Carlsson A, Lindqvist M, Magnusson T (1957) 3,4-Dihydroxyphenylalanine and 5-hydroxytryptophan as. reserpine antagonists Nature 180:1200. doi:https://doi.org/10.1038/1801200a0
Carneiro SB, Costa Duarte FI, Heimfarth L, Siqueira Quintans JS, Quintans-Junior LJ, Veiga Junior VFD, de Neves AA (2019) Cyclodextrin(-)Drug Inclusion Complexes: In Vivo and. Vitro Approaches International journal of molecular sciences 20. doi:https://doi.org/10.3390/ijms20030642
Cenini G, Lloret A, Cascella R (2019) Oxidative Stress in Neurodegenerative Diseases: From a Mitochondrial Point of View. Oxid Med Cell Longev 2019:2105607. doi:https://doi.org/10.1155/2019/2105607
Chaudhuri KR, Schapira AH (2009) Non-motor symptoms of Parkinson’s disease: dopaminergic pathophysiology and treatment. Lancet Neurol 8:464–474. doi:https://doi.org/10.1016/S1474-4422(09)70068-7
Chen MH et al (2016) Association Between Autonomic Impairment and Structural Deficit in Parkinson Disease. Med (Baltim) 95:e3086. doi:https://doi.org/10.1097/MD.0000000000003086
Chiang HL, Lin CH (2019) Altered Gut Microbiome and Intestinal Pathology in Parkinson’s Disease. J Mov Disord 12:67–83. doi:https://doi.org/10.14802/jmd.18067
Chu Y, Kordower JH (2007) Age-associated increases of alpha-synuclein in monkeys and humans are associated with nigrostriatal dopamine depletion: Is this the target for Parkinson’s disease? Neurobiol Dis 25:134–149. doi:https://doi.org/10.1016/j.nbd.2006.08.021
Cohen J (1988a) Statistical Power analysis for the behavioral science vol, vol 2nd. Erlbaum, Hillsdale, NJ
Cohen J (1988b) Statistical Power analysis for the behavioral sciences. 2nd ed
edn., Hillsdale, NJ:Erlbaum
Connolly B, Fox SH (2014) Treatment of cognitive, psychiatric, and affective disorders associated with Parkinson’s disease. Neurotherapeutics 11:78–91. doi:https://doi.org/10.1007/s13311-013-0238-x
da Silva RER et al (2018) Vasorelaxant effect of the Lippia alba essential oil and its major constituent, citral, on the contractility of isolated rat aorta. Biomed Pharmacother 108:792–798. doi:https://doi.org/10.1016/j.biopha.2018.09.073
de Farias CC et al (2016) Highly specific changes in antioxidant levels and lipid peroxidation in Parkinson’s disease and its progression: Disease and staging biomarkers and new drug targets. Neurosci Lett 617:66–71. doi:https://doi.org/10.1016/j.neulet.2016.02.011
De Gennaro L et al (2016) Dopaminergic system and dream recall: An MRI study in Parkinson’s disease patients. Hum Brain Mapp 37:1136–1147. doi:https://doi.org/10.1002/hbm.23095
Dissanayaka NNW et al (2017) Anxiety is associated with cognitive impairment in newly-diagnosed Parkinson’s disease. Parkinsonism Relat Disord 36:63–68. doi:https://doi.org/10.1016/j.parkreldis.2017.01.001
Doukkali Z et al (2016) Anti-anxiety effects of mercurialis annua aqueous extract in the elevated plus maze test. Pharm Bioprocess 4:52–57
Duncan GW et al (2014) Health-related quality of life in early Parkinson’s disease: the impact of nonmotor symptoms. Mov Disord 29:195–202. doi:https://doi.org/10.1002/mds.25664
Egan SJ, Laidlaw K, Starkstein S (2015) Cognitive Behaviour Therapy for Depression and Anxiety in Parkinson’s Disease. J Parkinsons Dis 5:443–451. doi:https://doi.org/10.3233/JPD-150542
Erel O (2004a) A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin Biochem 37:277–285. doi:https://doi.org/10.1016/j.clinbiochem.2003.11.015
Erel O (2004b) A novel automated method to measure total antioxidant response against potent free radical reactions. Clin Biochem 37:112–119. doi:https://doi.org/10.1016/j.clinbiochem.2003.10.014
Fernandes VS et al (2012) Repeated treatment with a low dose of reserpine as a progressive model of Parkinson’s disease. Behav Brain Res 231:154–163. doi:https://doi.org/10.1016/j.bbr.2012.03.008
Freis ED (1954) Mental depression in hypertensive patients treated for long periods with large doses of reserpine. N Engl J Med 251:1006–1008. doi:https://doi.org/10.1056/NEJM195412162512504
Gandhi SR, Quintans JSS, Gandhi GR, Araujo AAS, Quintans Junior LJ (2020) The use of cyclodextrin inclusion complexes to improve anticancer drug profiles: a systematic review. Expert Opin Drug Deliv 1–11. doi:https://doi.org/10.1080/17425247.2020.1776261
Grover S, Somaiya M, Kumar S, Avasthi A (2015) Psychiatric aspects of Parkinson’s disease. J Neurosci Rural Pract 6:65–76
Guo JT, Chen AQ, Kong Q, Zhu H, Ma CM, Qin C (2008) Inhibition of vesicular monoamine transporter-2 activity in alpha-synuclein stably transfected SH-SY5Y cells. Cell Mol Neurobiol 28:35–47. doi:https://doi.org/10.1007/s10571-007-9227-0
Habr S, Bernardi M, Conceição I, Freitas T, Felicio L (2011) Open field behavior and intra-nucleus accumbens dopamine release in vivo in virgin and lactating rats. Psychol Neurosci 4(1):115–121
Haliwell BG, J. M. C (1999) Free radicals in biology and medicine. Oxford University Press
Halliwell B (1996) Antioxidants in human health and disease. Annu Rev Nutr 16:33–50. doi:https://doi.org/10.1146/annurev.nu.16.070196.000341
Hemmati-Dinarvand M et al (2019) Oxidative stress and Parkinson’s disease: conflict of oxidant-antioxidant systems. Neurosci Lett 709:134296. doi:https://doi.org/10.1016/j.neulet.2019.134296
Herrera-Rodríguez SE L-RR, García-Márquez E, Estarrón-Espinosa M, Espinosa-Andrews H (2018) Mexican oregano (Lippia graveolens) essential oil-in-water emulsions: impact of emulsifier type on the antifungal activity of Candida albicans. Food Sci Biotechnol 28:441–448
Holdorff B, Rodrigues e Silva AM, Dodel R (2013) Centenary of Lewy bodies (1912–2012. J Neural Transm (Vienna) 120:509–516. doi:https://doi.org/10.1007/s00702-013-0984-2
Hornykiewicz O (1993) Parkinson’s disease and the adaptive capacity of the nigrostriatal dopamine system: possible neurochemical mechanisms. Adv Neurol 60:140–147
Huh YE, Hwang S, Kim K, Chung WH, Youn J, Cho JW (2016) Postural sensory correlates of freezing of gait in Parkinson’s disease. Parkinsonism Relat Disord 25:72–77. doi:https://doi.org/10.1016/j.parkreldis.2016.02.004
Jellinger KA (2012) Neuropathology of sporadic Parkinson’s disease: evaluation and changes of concepts. Mov Disord 27:8–30. doi:https://doi.org/10.1002/mds.23795
Jost WH (2017) Autonomic Dysfunction in Parkinson’s Disease: Cardiovascular Symptoms, Thermoregulation, and Urogenital Symptoms. Int Rev Neurobiol 134:771–785. doi:https://doi.org/10.1016/bs.irn.2017.04.003
Jung UJ, Kim SR (2018) Beneficial Effects of Flavonoids Against Parkinson’s Disease. J Med Food 21:421–432. doi:https://doi.org/10.1089/jmf.2017.4078
Kannappan R, Gupta SC, Kim JH, Reuter S, Aggarwal BB (2011) Neuroprotection by spice-derived nutraceuticals: you are what you eat! Mol Neurobiol 44:142–159. doi:https://doi.org/10.1007/s12035-011-8168-2
Kohlert C, van Rensen I, Marz R, Schindler G, Graefe EU, Veit M (2000) Bioavailability and pharmacokinetics of natural volatile terpenes in animals and humans. Planta Med 66:495–505. doi:https://doi.org/10.1055/s-2000-8616
Kraeuter A, Guest PC, Sarnyai Z (2019) The Open Field Test for Measuring Locomotor Activity and Anxiety-Like Behavior 99–103. https://doi.org/10.1007/978-1-4939-8994-2_9
Kumar V, Khan AA, Tripathi A (2015) Role of oxidative stress in various diseases: relevance of dietary antioxidants. J Pharm Exp Ther 4:126–132
Lam HA et al (2011) Elevated tonic extracellular dopamine concentration and altered dopamine modulation of synaptic activity precede dopamine loss in the striatum of mice overexpressing human alpha-synuclein. J Neurosci Res 89:1091–1102. doi:https://doi.org/10.1002/jnr.22611
Langston JW, Forno LS (1978) The hypothalamus in Parkinson disease. Ann Neurol 3:129–133. doi:https://doi.org/10.1002/ana.410030207
Leao AH et al (2017) Spontaneously Hypertensive Rats (SHR) Are Resistant to a Reserpine-Induced Progressive Model of Parkinson’s Disease: Differences in Motor Behavior, Tyrosine Hydroxylase and alpha-Synuclein Expression. Front Aging Neurosci 9:78. doi:https://doi.org/10.3389/fnagi.2017.00078
Leao AH, Sarmento-Silva AJ, Santos JR, Ribeiro AM, Silva RH (2015) Molecular, Neurochemical, and Behavioral Hallmarks of Reserpine as a Model for Parkinson’s Disease: New Perspectives to a Long-Standing. Model Brain Pathol 25:377–390. doi:https://doi.org/10.1111/bpa.12253
Leentjens AF (2011) The role of dopamine agonists in the treatment of depression in patients with Parkinson’s disease: a. Syst Rev Drugs 71:273–286. doi:https://doi.org/10.2165/11585380-000000000-00000
Lees AJ, Hardy J, Revesz T (2009) Parkinson’s disease Lancet 373:2055–2066. doi:https://doi.org/10.1016/S0140-6736(09)60492-X
Li J et al (2019) Multiple pathways for natural product treatment of Parkinson’s disease: A. mini Rev Phytomedicine 60:152954. doi:https://doi.org/10.1016/j.phymed.2019.152954
Lima BDS et al (2019) Development of morin/hydroxypropyl-beta-cyclodextrin inclusion complex: Enhancement of bioavailability, antihyperalgesic and anti-inflammatory effects. Food Chem Toxicol 126:15–24. doi:https://doi.org/10.1016/j.fct.2019.01.038
Lima PSS, Lucchese AM, Araujo-Filho HG, Menezes PP, Araujo AAS, Quintans-Junior LJ, Quintans JSS (2016) Inclusion of terpenes in cyclodextrins: Preparation, characterization and pharmacological approaches. Carbohydr Polym 151:965–987. doi:https://doi.org/10.1016/j.carbpol.2016.06.040
Lobay D (2015) Rauwolfia in the Treatment of Hypertension Integr Med. (Encinitas) 14:40–46
Loeffler DA, Klaver AC, Coffey MP, Aasly JO, LeWitt PA (2017) Increased Oxidative Stress Markers in Cerebrospinal Fluid from Healthy Subjects with Parkinson’s Disease-Associated LRRK2 Gene Mutations. Front Aging Neurosci 9:89. doi:https://doi.org/10.3389/fnagi.2017.00089
Lohr JB, Kuczenski R, Bracha HS, Moir M, Jeste DV (1990) Increased indices of free radical activity in the cerebrospinal fluid of patients with tardive dyskinesia. Biol Psychiatry 28:535–539. doi:https://doi.org/10.1016/0006-3223(90)90490-s
Marsh L (2013) Depression and Parkinson’s disease: current knowledge Curr. Neurol Neurosci Rep 13:409. doi:https://doi.org/10.1007/s11910-013-0409-5
Martens KAE, Hall JM, Gilat M, Georgiades MJ, Walton CC, Lewis SJG (2016) Anxiety is associated with freezing of gait and attentional set-shifting in Parkinson’s disease: A new perspective for early intervention. Gait Posture 49:431–436. doi:https://doi.org/10.1016/j.gaitpost.2016.07.182
Mayeux R (2003) Epidemiology of neurodegeneration Annual. Rev Neurosci 26:81–104. doi:https://doi.org/10.1146/annurev.neuro.26.043002.094919
Mc QE, Doyle AE, Smirk FH (1954) Mechanism of hypotensive action of reserpine, an alkaloid of Rauwolfia. serpentina Nat 174:1015. doi:https://doi.org/10.1038/1741015b0
Mhyre TR, Boyd JT, Hamill RW, Maguire-Zeiss KA (2012) Parkinson’s disease Subcell Biochem 65:389–455. doi:https://doi.org/10.1007/978-94-007-5416-4_16
Milanese C et al (2018) Activation of the DNA damage response in vivo in synucleinopathy models of Parkinson’s disease. Cell Death Dis 9:818. doi:https://doi.org/10.1038/s41419-018-0848-7
Nisihara CMH, Tumas V, Loureiro SR, Lemos Correa AC, Kawasaki Nakabayashi TI, Crippa JA (2009) Does the association between anxiety and Parkinson’s disease really exist? A literature review. Curr Psychiatry Rev 5:29–36
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. doi:https://doi.org/10.1016/0003-2697(79)90738-3
Peng X, Tehranian R, Dietrich P, Stefanis L, Perez RG (2005) Alpha-synuclein activation of protein phosphatase 2A reduces tyrosine hydroxylase phosphorylation in dopaminergic cells. J Cell Sci 118:3523–3530. doi:https://doi.org/10.1242/jcs.02481
Pirogovsky-Turk E, Moore RC, Filoteo JV, Litvan I, Song DD, Lessig SL, Schiehser DM (2017) Neuropsychiatric Predictors of Cognitive Decline in Parkinson Disease: A Longitudinal Study. Am J Geriatr Psychiatry 25:279–289. doi:https://doi.org/10.1016/j.jagp.2016.10.004
Politi C, Ciccacci C, Novelli G, Borgiani P (2018) Genetics and Treatment Response in Parkinson’s Disease: An Update on Pharmacogenetic Studies. Neuromolecular Med 20:1–17. doi:https://doi.org/10.1007/s12017-017-8473-7
Pringsheim T, Jette N, Frolkis A, Steeves TD (2014) The prevalence of Parkinson’s disease: a systematic review and meta-analysis. Mov Disord 29:1583–1590. doi:https://doi.org/10.1002/mds.25945
Quintans-Júnior LJ, Barreto RSS, Menezes PP, Almeida JRGS, Viana AFSC, Oliveira RCM et al (2013) β-Cyclodextrin-complexed (–)-linalool produces antinociceptive effect superior to that of (–)-linalool in experimental pain protocols Basic. Clin Pharmacol Toxicol 113:167–172
Reynolds GO, Hanna KK, Neargarder S, Cronin-Golomb A (2017) The relation of anxiety and cognition in Parkinson’. s disease Neuropsychology 31:596–604. doi:https://doi.org/10.1037/neu0000353
Richard IH et al (2012) A randomized, double-blind, placebo-controlled trial of antidepressants in Parkinson. disease Neurol 78:1229–1236. doi:https://doi.org/10.1212/WNL.0b013e3182516244
Riederer P, Muller T (2018) Monoamine oxidase-B inhibitors in the treatment of Parkinson’s disease: clinical-pharmacological aspects. J Neural Transm (Vienna) 125:1751–1757. doi:https://doi.org/10.1007/s00702-018-1876-2
Rodriguez Diaz M, Abdala P, Barroso-Chinea P, Obeso J, Gonzalez-Hernandez T (2001) Motor behavioural changes after intracerebroventricular injection of 6-hydroxydopamine in the rat: an animal model of Parkinson’s disease. Behav Brain Res 122:79–92. doi:https://doi.org/10.1016/s0166-4328(01)00168-1
Sagna A, Gallo JJ, Pontone GM (2014) Systematic review of factors associated with depression and anxiety disorders among older adults with Parkinson’s disease. Parkinsonism Relat Disord 20:708–715. doi:https://doi.org/10.1016/j.parkreldis.2014.03.020
Santos JR et al (2013) Cognitive, motor and tyrosine hydroxylase temporal impairment in a model of parkinsonism induced by reserpine. Behav Brain Res 253:68–77. doi:https://doi.org/10.1016/j.bbr.2013.06.031
Santos PL, Brito RG, Quintans JSS, Araujo AAS, Menezes IRA, Brogden NK, Quintans-Junior LJ (2017) Cyclodextrins as Complexation Agents to Improve the Anti-inflammatory Drugs Profile: a Systematic Review and Meta-Analysis. Curr Pharm Des 23:2096–2107. doi:https://doi.org/10.2174/1381612823666170126121926
Sarmento AJ, Lima RH, Cabral A, Meurer Y, Ribeiro AM, Silva RH (2015) Alpha-tocopherol counteracts cognitive and motor deficits induced by repeated treatment with reserpine. Biochem Pharmacol 4:1–6
Sauer H, Oertel WH (1994) Progressive degeneration of nigrostriatal dopamine neurons following intrastriatal terminal lesions with 6-hydroxydopamine: A combined retrograde tracing and immunocytochemical study in the rat Neuroscience. 59:401–4152
Schapira AHV, Chaudhuri KR, Jenner P (2017) Non-motor features of Parkinson disease. Nat Rev Neurosci 18:509. doi:https://doi.org/10.1038/nrn.2017.91
Singulani JL, Silva PS, Raposo NR, de Siqueira EP, Zani CL, Alves TM, Viccini LF (2012) Chemical composition and antioxidant activity of Lippia species. J Med Plants Res 6:4416–4422
Siqueira-Lima PS et al (2014) beta-cyclodextrin complex containing Lippia grata leaf essential oil reduces orofacial nociception in mice - evidence of possible involvement of descending inhibitory pain modulation pathway Basic. Clin Pharmacol Toxicol 114:188–196. doi:https://doi.org/10.1111/bcpt.12145
Siqueira-Lima PS et al (2017) Anti-hyperalgesic effect of Lippia grata leaf essential oil complexed with beta-cyclodextrin in a chronic musculoskeletal pain animal model: Complemented with a molecular docking and antioxidant screening. Biomed Pharmacother 91:739–747. doi:https://doi.org/10.1016/j.biopha.2017.05.009
Siqueira-Lima PS et al (2019) Involvement of the PKA pathway and inhibition of voltage gated Ca2 + channels in antihyperalgesic activity of Lippia grata/beta-cyclodextrin Life sciences 239. 116961. https://doi.org/10.1016/j.lfs.2019.116961
Skorvanek M, Gdovinova Z, Rosenberger J, Saeedian RG, Nagyova I, Groothoff JW, van Dijk JP (2015) The associations between fatigue, apathy, and depression in Parkinson’s disease. Acta Neurol Scand 131:80–87. doi:https://doi.org/10.1111/ane.12282
Spillantini MG, Schmidt ML, Lee VM, Trojanowski JQ, Jakes R, Goedert M (1997) Alpha-synuclein in Lewy bodies. Nature 388:839–840. doi:https://doi.org/10.1038/42166
Stayte S, Vissel B (2014) Advances in non-dopaminergic treatments for Parkinson’s disease. Front Neurosci 8:113. doi:https://doi.org/10.3389/fnins.2014.00113
Su CF, Jiang L, Zhang XW, Iyaswamy A, Li M (2021) Resveratrol in Rodent Models of Parkinson’s Disease: A Systematic Review of Experimental Studies Frontiers in Pharmacology 12,1–10. https://doi.org/10.3389/fphar.2021.644219
Suppa A, Bologna M, Conte A, Berardelli A, Fabbrini G (2017) The effect of L-dopa in Parkinson’s disease as revealed by neurophysiological studies of motor and sensory functions. Expert Rev Neurother 17:181–192. doi:https://doi.org/10.1080/14737175.2016.1219251
Taimur M, Shah MAA, Ali M, Barry HD, Hussain SZM, Shahzad H, Rizwan A (2019) Frequency of Cognitive Impairment in Patients with. Parkinson’s Disease Cureus 11:e4733. doi:https://doi.org/10.7759/cureus.4733
Teerapattarakan N, Benya-Aphikul H, Tansawat R, Wanakhachornkrai O, Tantisira MH, Rodsiri R (2018) Neuroprotective effect of a standardized extract of Centella asiatica ECa233 in rotenone-induced parkinsonism. rats Phytomedicine 44:65–73. doi:https://doi.org/10.1016/j.phymed.2018.04.028
Terblanché FCK, G (1996) Essential oil constituents of the genus Lippia (Verbenaceae) - A literature review. J Essencial Oil Res 8:471–485
Todorova A, Jenner P, Ray Chaudhuri K (2014) Non-motor Parkinson’s: integral to motor Parkinson’s. yet often neglected Pract Neurol 14:310–322. doi:https://doi.org/10.1136/practneurol-2013-000741
Tong J et al (2010) Brain alpha-synuclein accumulation in multiple system atrophy, Parkinson’s disease and progressive supranuclear palsy: a. Comp Invest Brain 133:172–188. doi:https://doi.org/10.1093/brain/awp282
Uhl GR, Hedreen JC, Price DL (1985) Parkinson’s disease: loss of neurons from the ventral tegmental area contralateral to therapeutic. Surg lesions Neurol 35:1215–1218. doi:https://doi.org/10.1212/wnl.35.8.1215
Ullah R, Khan M, Shah SA, Saeed K, Kim MO (2019) Natural Antioxidant Anthocyanins-A Hidden Therapeutic Candidate in Metabolic Disorders with Major Focus in Neurodegeneration Nutrients. 11. https://doi.org/10.3390/nu11061195
Ulusoy A, Bjorklund T, Buck K, Kirik D (2012) Dysregulated dopamine storage increases the vulnerability to alpha-synuclein in nigral neurons. Neurobiol Dis 47:367–377. doi:https://doi.org/10.1016/j.nbd.2012.05.012
Veras HNAM, Costa JG, Coutinho HD, Kerntopf MR, Botelho MA, Menezes IR (2013) Topical antiinflammatory activity of essential oil of Lippia sidoides cham
possible mechanism of action Phytother Res 27:179–185
Viana GSBM, Araujo FF;WL, Matos FJA, Craveiro AA (1981) Essential Oil of Lippia grata: Pharmacological Effects and Main Constituents Quart. J Crude Drug Res 19:1–10
Vida C, Kobayashi H, Garrido A, Martinez de Toda I, Carro E, Molina JA, De la Fuente M (2019) Lymphoproliferation Impairment and Oxidative Stress in Blood Cells from Early Parkinson’s. Disease Patients International journal of molecular sciences 20. doi:https://doi.org/10.3390/ijms20030771
Weintraub D, Moberg PJ, Duda JE, Katz IR, Stern MB (2004) Effect of psychiatric and other nonmotor symptoms on disability in Parkinson’s disease. J Am Geriatr Soc 52:784–788. doi:https://doi.org/10.1111/j.1532-5415.2004.52219.x
Acknowledgements
In this study grants and fellowships were supported by Fundação de Amparo à Pesquisa no Estado de São Paulo (FAPESP - grant #2015/20785-8, #2015/03354-3, #2018/26609-5 and #2020/09015-4), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq - grant #425694/2016-0, #310403/2021-9 and #408377/2021-6) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) – Finance Code 001; Pró-reitoria de Pós-graduação Pesquisa da Universidade Federal de São Paulo (PROPESQ/UNIFESP).
Funding
In this study grants and fellowships were supported by Fundação de Amparo à Pesquisa no Estado de São Paulo (FAPESP - grant #2015/20785-8, #2015/03354-3 and #2018/26609-5), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq - grant #425694/2016-0) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) – Finance Code 001; Pró-reitoria de Pós-graduação Pesquisa da Universidade Federal de São Paulo (PROPESQ/UNIFESP).
Author information
Authors and Affiliations
Contributions
JIABF and AMR designed the study. JIABF, AMM, SSM, ACC, BS, RHL, JRS performed the experiments and collected the data. JIABF, AMM, LJQJ and AMR analyzed data and wrote the manuscript. JRS, RHL, LJQJ, RHS and AMR contributed with theoretical discussions and technical insights. JIABF, RHL, JRS, RHS, LJQJ and AMR contributed in analysis and writing, and revised the final manuscript. All the authors have read the final manuscript and approved the submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no financial interests.
Informed consent
None.
Ethical approval
All procedures involving animals were in compliance with the Brazilian law for the use of animals in scientific research (Law number 11.794) of October 8, 2008, Decree 6899 of July 15, 2009, as well as with the rules issued by the National Council for Control of Animal Experimentation (CONCEA), and was approved by the Ethic Committee on Animal Use of the Federal University of Sao Paulo (CEUA/UNIFESP Protocol No. 9407030517/2017) in the meeting of May 22, 2017.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Beserra-Filho, J.I.A., Maria-Macêdo, A., Silva-Martins, S. et al. Lippia grata essential oil complexed with β-cyclodextrin ameliorates biochemical and behavioral deficits in an animal model of progressive parkinsonism. Metab Brain Dis 37, 2331–2347 (2022). https://doi.org/10.1007/s11011-022-01032-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-01032-2