Abstract
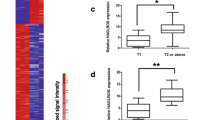
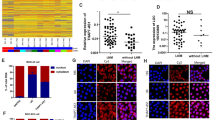
Long non-coding RNA (lncRNA) growth arrest-specific transcript 5 (GAS5) has been shown to be a regulator for many cancers, including non-small cell lung cancer (NSCLC). Therefore, its role and mechanism in the process of NSCLC deserve to be further revealed. The expression levels of GAS5, fat mass and obesity-associated protein (FTO) and bromodomain-containing protein 4 (BRD4) were detected by quantitative real-time PCR. Western blot analysis was used to examine the protein expression of FTO, BRD4, up-frameshift protein 1 (UPF1) and autophagy-related markers. Methylated RNA immunoprecipitation was used to assess the m6A level of GAS5 regulated by FTO. Cell proliferation and apoptosis were determined using MTT assay, EdU assay and flow cytometry. Autophagy ability was assessed by immunofluorescence staining and transmission electron microscope. Xenograft tumor model was constructed to explore the effects of FTO and GAS5 on NSCLC tumor growth in vivo. The interaction between UPF1 and GAS5 or BRD4 was confirmed by pull-down assay, RIP assay, dual-luciferase reporter assay, and chromatin immunoprecipitation. Fluorescent in situ hybridization was used to analyze the co-localization of GAS5 and UPF1. Actinomycin D treatment was employed to evaluate BRD4 mRNA stability. GAS5 was downregulated in NSCLC tissues and was associated with poor prognosis in NSCLC patients. FTO was highly expressed in NSCLC, and it inhibited GAS5 expression by reducing GAS5 m6A methylation level. GAS5 suppressed by FTO could promote the autophagic death of NSCLC cells in vitro and inhibit NSCLC tumor growth in vivo. In addition, GAS5 was able to interact with UPF1 to reduce the mRNA stability of BRD4. Knockdown of BRD4 reversed the inhibition of GAS5 or UPF1 silencing on the autophagic cell death of NSCLC. The findings of the study showed that lncRNA GAS5 mediated by FTO could contribute to the autophagic cell death of NSCLC by interacting with UPF1 to reduce BRD4 mRNA stability, suggesting that GAS5 might be a vital therapy target for NSCLC progression.









Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Herbst RS, Morgensztern D, Boshoff C (2018) The biology and management of non-small cell lung cancer. Nature 553:446–454. https://doi.org/10.1038/nature25183
Sezen CB, Celik A, Fazlioglu M, Memis L, Tastepe AI (2015) Lung cancer developing from tracheal bronchus. Asian Cardiovasc Thorac Ann 23:484–486. https://doi.org/10.1177/0218492314541450
Cheema PK, Rothenstein J, Melosky B, Brade A, Hirsh V (2019) Perspectives on treatment advances for stage III locally advanced unresectable non-small-cell lung cancer. Curr Oncol 26:37–42. https://doi.org/10.3747/co.25.4096
Soria JC, Massard C, Le Chevalier T (2010) Should progression-free survival be the primary measure of efficacy for advanced NSCLC therapy? Ann Oncol 21:2324–2332. https://doi.org/10.1093/annonc/mdq204
Jonna S, Subramaniam DS (2019) Molecular diagnostics and targeted therapies in non-small cell lung cancer (NSCLC): an update. Discov Med 27:167–170
Imyanitov EN, Iyevleva AG, Levchenko EV (2021) Molecular testing and targeted therapy for non-small cell lung cancer: current status and perspectives. Crit Rev Oncol Hematol 157:103194. https://doi.org/10.1016/j.critrevonc.2020.103194
Sanchez Calle A, Kawamura Y, Yamamoto Y, Takeshita F, Ochiya T (2018) Emerging roles of long non-coding RNA in cancer. Cancer Sci 109:2093–2100. https://doi.org/10.1111/cas.13642
Fu Y, Liu L, Zhan J, Zhan H, Qiu C (2021) LncRNA GAS5 expression in non-small cell lung cancer tissues and its correlation with Ki67 and EGFR. Am J Transl Res 13:4900–4907
Yang X, Meng L, Zhong Y, Hu F, Wang L, Wang M (2021) The long intergenic noncoding RNA GAS5 reduces cisplatin-resistance in non-small cell lung cancer through the miR-217/LHPP axis. Aging 13:2864–2884. https://doi.org/10.18632/aging.202352
Ma J, Miao H, Zhang H, Ren J, Qu S, Da J, Xu F, Zhao H (2021) LncRNA GAS5 modulates the progression of non-small cell lung cancer through repressing mir-221-3p and up-regulating IRF2. Diagn Pathol 16:46. https://doi.org/10.1186/s13000-021-01108-0
Dong L, Li G, Li Y, Zhu Z (2019) Upregulation of long noncoding RNA GAS5 inhibits lung cancer cell proliferation and metastasis via miR-205/PTEN axis. Med Sci Monit 25:2311–2319. https://doi.org/10.12659/MSM.912581
Kamel LM, Atef DM, Mackawy AMH, Shalaby SM, Abdelraheim N (2019) Circulating long non-coding RNA GAS5 and SOX2OT as potential biomarkers for diagnosis and prognosis of non-small cell lung cancer. Biotechnol Appl Biochem 66:634–642. https://doi.org/10.1002/bab.1764
An Y, Duan H (2022) The role of m6A RNA methylation in cancer metabolism. Mol Cancer 21:14. https://doi.org/10.1186/s12943-022-01500-4
Chen J, Du B (2019) Novel positioning from obesity to cancer: FTO, an m(6)a RNA demethylase, regulates tumour progression. J Cancer Res Clin Oncol 145:19–29. https://doi.org/10.1007/s00432-018-2796-0
Wang Y, Li M, Zhang L, Chen Y, Zhang S (2021) m6A demethylase FTO induces NELL2 expression by inhibiting E2F1 m6A modification leading to metastasis of non-small cell lung cancer. Mol Ther Oncolytics 21:367–376. https://doi.org/10.1016/j.omto.2021.04.011
Li J, Han Y, Zhang H, Qian Z, Jia W, Gao Y, Zheng H, Li B (2019) The m6A demethylase FTO promotes the growth of lung cancer cells by regulating the m6A level of USP7 mRNA. Biochem Biophys Res Commun 512:479–485. https://doi.org/10.1016/j.bbrc.2019.03.093
Donati B, Lorenzini E, Ciarrocchi A (2018) BRD4 and cancer: going beyond transcriptional regulation. Mol Cancer 17:164. https://doi.org/10.1186/s12943-018-0915-9
Shi L, Xiong Y, Hu X, Wang Z, Xie C (2021) BRD4 inhibition promotes TRAIL-induced apoptosis by suppressing the transcriptional activity of NF-kappaB in NSCLC. Int J Med Sci 18:3090–3096. https://doi.org/10.7150/ijms.60776
Gao Z, Yuan T, Zhou X, Ni P, Sun G, Li P, Cheng Z, Wang X (2018) Targeting BRD4 proteins suppresses the growth of NSCLC through downregulation of eIF4E expression. Cancer Biol Ther 19:407–415. https://doi.org/10.1080/15384047.2018.1423923
Ouyang L, Zhang L, Liu J, Fu L, Yao D, Zhao Y, Zhang S, Wang G, He G, Liu B (2017) Discovery of a small-molecule bromodomain-containing protein 4 (BRD4) inhibitor that induces AMP-Activated protein kinase-modulated autophagy-associated cell death in breast cancer. J Med Chem 60:9990–10012. https://doi.org/10.1021/acs.jmedchem.7b00275
Lavysh D, Neu-Yilik G (2020) UPF1-mediated RNA decay—Danse Macabre in a cloud. Biomolecules 10. https://doi.org/10.3390/biom10070999
Mei Y, Si J, Wang Y, Huang Z, Zhu H, Feng S, Wu X, Wu L (2017) Long noncoding RNA GAS5 suppresses tumorigenesis by inhibiting miR-23a expression in non-small cell lung cancer. Oncol Res 25:1027–1037. https://doi.org/10.3727/096504016X14822800040451
Xue Y, Ni T, Jiang Y, Li Y (2017) Long noncoding RNA GAS5 inhibits tumorigenesis and enhances radiosensitivity by suppressing miR-135b expression in non-small cell lung cancer. Oncol Res 25:1305–1316. https://doi.org/10.3727/096504017X14850182723737
Ni W, Yao S, Zhou Y, Liu Y, Huang P, Zhou A, Liu J, Che L, Li J (2019) Long noncoding RNA GAS5 inhibits progression of colorectal cancer by interacting with and triggering YAP phosphorylation and degradation and is negatively regulated by the m(6)a reader YTHDF3. Mol Cancer 18:143. https://doi.org/10.1186/s12943-019-1079-y
Dong Q, Long X, Cheng J, Wang W, Tian Q, Di W (2021) LncRNA GAS5 suppresses ovarian cancer progression by targeting the miR-96-5p/PTEN axis. Ann Transl Med 9:1770. https://doi.org/10.21037/atm-21-6134
Yang W, Hong L, Xu X, Wang Q, Huang J, Jiang L (2017) LncRNA GAS5 suppresses the tumorigenesis of cervical cancer by downregulating miR-196a and miR-205. Tumour Biol 39:1010428317711315. https://doi.org/10.1177/1010428317711315
Xu T, Xu X, Chu Y, Jiang D, Xu G (2021) Longchain noncoding RNA GAS5 promotes cell autophagy by modulating the miR181c5p/ATG5 and miR1192/ATG12 axes. Int J Mol Med. https://doi.org/10.3892/ijmm.2021.5042
Zhang N, Yang GQ, Shao XM, Wei L (2016) GAS5 modulated autophagy is a mechanism modulating cisplatin sensitivity in NSCLC cells. Eur Rev Med Pharmacol Sci 20:2271–2277
Jiang X, Liu B, Nie Z, Duan L, Xiong Q, Jin Z, Yang C, Chen Y (2021) The role of m6A modification in the biological functions and diseases. Signal Transduct Target Ther 6:74. https://doi.org/10.1038/s41392-020-00450-x
Tao L, Mu X, Chen H, Jin D, Zhang R, Zhao Y, Fan J, Cao M, Zhou Z (2021) FTO modifies the m6A level of MALAT and promotes bladder cancer progression. Clin Transl Med 11:e310. https://doi.org/10.1002/ctm2.310
Zhang L, Wan Y, Zhang Z, Jiang Y, Lang J, Cheng W, Zhu L (2021) FTO demethylates m6A modifications in HOXB13 mRNA and promotes endometrial cancer metastasis by activating the WNT signalling pathway. RNA Biol 18:1265–1278. https://doi.org/10.1080/15476286.2020.1841458
Sommer G, Heise T (2021) Role of the RNA-binding protein La in cancer pathobiology. RNA Biol 18:218–236. https://doi.org/10.1080/15476286.2020.1792677
Han S, Cao D, Sha J, Zhu X, Chen D (2020) LncRNA ZFPM2-AS1 promotes lung adenocarcinoma progression by interacting with UPF1 to destabilize ZFPM2. Mol Oncol 14:1074–1088. https://doi.org/10.1002/1878-0261.12631
Xie X, Lin J, Liu J, Huang M, Zhong Y, Liang B, Song X, Gu S, Chang X, Huang D, Tang A (2019) A novel lncRNA NR4A1AS up-regulates orphan nuclear receptor NR4A1 expression by blocking UPF1-mediated mRNA destabilization in colorectal cancer. Clin Sci (Lond) 133:1457–1473. https://doi.org/10.1042/CS20181061
Pellicori P, Zhang J, Cuthbert J, Urbinati A, Shah P, Kazmi S, Clark AL, Cleland JGF (2020) High-sensitivity C-reactive protein in chronic heart failure: patient characteristics, phenotypes, and mode of death. Cardiovasc Res 116:91–100. https://doi.org/10.1093/cvr/cvz198
Funding
This study was supported by Hainan Province Science and Technology Special Fund (Grant No. ZDYF2023SHFZ133) and Hainan Provincial Natural Science Foundation of China (Grant No. 823MS142).
Author information
Authors and Affiliations
Contributions
YHF and LRL conceived and designed research. YHF and HHW conducted experiments. HHW, YMZ and HJZ analyzed data. LL wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
Each patient signed written informed consent, and the research was approved by the Ethics Committee of the Hainan Affiliated Hospital of Hainan Medical University.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fu, Y., Liu, L., Wu, H. et al. LncRNA GAS5 regulated by FTO-mediated m6A demethylation promotes autophagic cell death in NSCLC by targeting UPF1/BRD4 axis. Mol Cell Biochem 479, 553–566 (2024). https://doi.org/10.1007/s11010-023-04748-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-023-04748-6