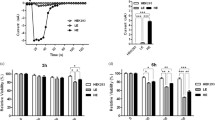
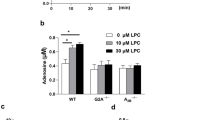
Abstract
P2X7 receptor (P2X7R) is an ATP-gated non-selective cation channel which mediates ATP-induced inflammation in macrophages. Transient receptor potential (TRP) receptors are nociceptors in cellular membrane which can perceive the stimuli of environmental irritant. The interaction between TRP channels and P2X7R has been found while the details about inflammation are still unclear. In this study, we suggested that transient receptor potential ankyrin 1 (TRPA1), a member of TRP superfamily, participates in ATP-induced oxidative stress and inflammation in human acute monocytic leukemia cell line (THP-1)-derived macrophage. The co-localization between TRPA1 and P2X7R was detected using immunofluorescence in THP-1-derived macrophage and transfected human embryonic kidney cell line (HEK293T). The mechanism by which ATP or 3′-O-(4-Benzoylbenzoyl)-ATP (BzATP) induces the activation of macrophages was verified by calcium imaging, mitochondrial reactive oxygen species (mtROS) detection, mitochondrial membrane potential (∆Ψm) measurement, flow cytometry, enzyme-linked immunosorbent assay (ELISA), western blotting, CCK-8 assay, and the lactate dehydrogenase (LDH) release cytotoxic assay. The BzATP and ATP induced calcium overload, mitochondria injury, interleukin-1β (IL-1β) secretion, and cytotoxicity can be inhibited by TRPA1 antagonists. These results indicated that TRPA1 can co-localize with P2X7R and mediate ATP-induced oxidative stress and inflammation. Therefore, the inhibition of TRPA1 may provide a potential therapy for ATP-elicited inflammatory diseases, including atherosclerosis.








Similar content being viewed by others
References
Idzko M, Ferrari D, Eltzschig HK (2014) Nucleotide signalling during inflammation. Nature 509:310–317. https://doi.org/10.1038/nature13085
Junger WG (2011) Immune cell regulation by autocrine purinergic signalling. Nat Rev Immunol 11:201–212. https://doi.org/10.1038/nri2938
Stachon P, Geis S, Peikert A, Heidenreich A, Michel NA, Unal F, Hoppe N, Dufner B, Schulte L, Marchini T, Cicko S, Ayata K, Zech A, Wolf D, Hilgendorf I, Willecke F, Reinohl J, von Zur MC, Bode C, Idzko M, Zirlik A (2016) Extracellular ATP induces vascular inflammation and atherosclerosis via purinergic receptor Y2 in mice. Arterioscler Thromb Vasc Biol 36:1577–1586. https://doi.org/10.1161/atvbaha.115.307397
Zhao TV, Li Y, Liu X, Xia S, Shi P, Li L, Chen Z, Yin C, Eriguchi M, Chen Y, Bernstein EA, Giani JF, Bernstein KE, Shen XZ (2019) ATP release drives heightened immune responses associated with hypertension. Sci Immunol. https://doi.org/10.1126/sciimmunol.aau6426
Peng K, Liu L, Wei D, Lv Y, Wang G, Xiong W, Wang X, Altaf A, Wang L, He D, Wang H, Qu P (2015) P2X7R is involved in the progression of atherosclerosis by promoting NLRP3 inflammasome activation. Int J Mol Med 35:1179–1188. https://doi.org/10.3892/ijmm.2015.2129
McCarthy AE, Yoshioka C, Mansoor SE (2019) Full-length P2X7 structures reveal how palmitoylation prevents channel desensitization. Cell 179:659–670.e13. https://doi.org/10.1016/j.cell.2019.09.017
Gicquel T, Robert S, Loyer P, Victoni T, Bodin A, Ribault C, Gleonnec F, Couillin I, Boichot E, Lagente V (2015) IL-1beta production is dependent on the activation of purinergic receptors and NLRP3 pathway in human macrophages. FASEB J 29:4162–4173. https://doi.org/10.1096/fj.14-267393
Yaron JR, Gangaraju S, Rao MY, Kong X, Zhang L, Su F, Tian Y, Glenn HL, Meldrum DR (2015) K(+) regulates Ca(2+) to drive inflammasome signaling: dynamic visualization of ion flux in live cells. Cell Death Dis 6:e1954. https://doi.org/10.1038/cddis.2015.277
Gong T, Liu L, Jiang W, Zhou R (2019) DAMP-sensing receptors in sterile inflammation and inflammatory diseases. Nat Rev Immunol. https://doi.org/10.1038/s41577-019-0215-7
Janks L, Sprague RS, Egan TM (2019) ATP-gated P2X7 receptors require chloride channels to promote inflammation in human macrophages. J Immunol 202:883–898. https://doi.org/10.4049/jimmunol.1801101
Bautista DM, Pellegrino M, Tsunozaki M (2013) TRPA1: A gatekeeper for inflammation. Annu Rev Physiol 75:181–200. https://doi.org/10.1146/annurev-physiol-030212-183811
Hajna Z, Saghy E, Payrits M, Aubdool AA, Szoke E, Pozsgai G, Batai IZ, Nagy L, Filotas D, Helyes Z, Brain SD, Pinter E (2016) Capsaicin-sensitive sensory nerves mediate the cellular and microvascular effects of H2S via TRPA1 receptor activation and neuropeptide release. J Mol Neurosci 60:157–170. https://doi.org/10.1007/s12031-016-0802-z
Guntur AR, Gu P, Takle K, Chen J, Xiang Y, Yang CH (2015) Drosophila TRPA1 isoforms detect UV light via photochemical production of H2O2. Proc Natl Acad Sci U S A 112:E5753–E5761. https://doi.org/10.1073/pnas.1514862112
Liu B, Tai Y, Caceres AI, Achanta S, Balakrishna S, Shao X, Fang J, Jordt SE (2016) Oxidized phospholipid OxPAPC activates TRPA1 and contributes to chronic inflammatory pain in mice. PLoS ONE 11:e0165200. https://doi.org/10.1371/journal.pone.0165200
Tian C, Huang R, Tang F, Lin Z, Cheng N, Han X, Li S, Zhou P, Deng S, Huang H, Zhao H, Xu J, Li Z (2020) Transient receptor potential ankyrin 1 contributes to lysophosphatidylcholine-induced intracellular calcium regulation and THP-1-derived macrophage activation. J Membr Biol 253:43–55. https://doi.org/10.1007/s00232-019-00104-2
Faria RX, Reis RA, Ferreira LG, Cezar-de-Mello PF, Moraes MO (2016) P2X7R large pore is partially blocked by pore forming proteins antagonists in astrocytes. J Bioenerg Biomembr 48:309–324. https://doi.org/10.1007/s10863-016-9649-9
Faria RX, Freitas HR, Reis RAM (2017) P2X7 receptor large pore signaling in avian Muller glial cells. J Bioenerg Biomembr 49:215–229. https://doi.org/10.1007/s10863-017-9717-9
Stanchev D, Blosa M, Milius D, Gerevich Z, Rubini P, Schmalzing G, Eschrich K, Schaefer M, Wirkner K, Illes P (2009) Cross-inhibition between native and recombinant TRPV1 and P2X(3) receptors. Pain 143:26–36. https://doi.org/10.1016/j.pain.2009.01.006
Heid ME, Keyel PA, Kamga C, Shiva S, Watkins SC, Salter RD (2013) Mitochondrial reactive oxygen species induces NLRP3-dependent lysosomal damage and inflammasome activation. J Immunol 191:5230–5238. https://doi.org/10.4049/jimmunol.1301490
Schilling T, Eder C (2009) Non-selective cation channel activity is required for lysophosphatidylcholine-induced monocyte migration. J Cell Physiol 221:325–334. https://doi.org/10.1002/jcp.21857
Li X, Fang P, Li Y, Kuo YM, Andrews AJ, Nanayakkara G, Johnson C, Fu H, Shan H, Du F, Hoffman NE, Yu D, Eguchi S, Madesh M, Koch WJ, Sun J, Jiang X, Wang H, Yang X (2016) Mitochondrial reactive oxygen species mediate lysophosphatidylcholine-induced endothelial cell activation. Arterioscler Thromb Vasc Biol 36:1090–1100. https://doi.org/10.1161/atvbaha.115.306964
Deveci HA, Akyuva Y, Nur G, Naziroglu M (2019) Alpha lipoic acid attenuates hypoxia-induced apoptosis, inflammation and mitochondrial oxidative stress via inhibition of TRPA1 channel in human glioblastoma cell line. Biomed Pharmacother 111:292–304. https://doi.org/10.1016/j.biopha.2018.12.077
Zhou R, Yazdi AS, Menu P, Tschopp J (2011) A role for mitochondria in NLRP3 inflammasome activation. Nature 469:221–225. https://doi.org/10.1038/nature09663
Hamilton NB, Kolodziejczyk K, Kougioumtzidou E, Attwell D (2016) Proton-gated Ca(2+)-permeable TRP channels damage myelin in conditions mimicking ischaemia. Nature 529:523–527. https://doi.org/10.1038/nature16519
Mackenzie AB, Young MT, Adinolfi E, Surprenant A (2005) Pseudoapoptosis induced by brief activation of ATP-gated P2X7 receptors. J Biol Chem 280:33968–33976. https://doi.org/10.1074/jbc.M502705200
Adinolfi E, Callegari MG, Ferrari D, Bolognesi C, Minelli M, Wieckowski MR, Pinton P, Rizzuto R, Di Virgilio F (2005) Basal activation of the P2X7 ATP receptor elevates mitochondrial calcium and potential, increases cellular ATP levels, and promotes serum-independent growth. Mol Biol Cell 16:3260–3272. https://doi.org/10.1091/mbc.e04-11-1025
Zhao J, Xu S, Song F, Nian L, Zhou X, Wang S (2014) 2,3,5,4'-tetrahydroxystilbene-2-O-beta-d-glucoside protects human umbilical vein endothelial cells against lysophosphatidylcholine-induced apoptosis by upregulating superoxide dismutase and glutathione peroxidase. IUBMB Life 66:711–722. https://doi.org/10.1002/iub.1321
Grebe A, Hoss F, Latz E (2018) NLRP3 inflammasome and the IL-1 pathway in atherosclerosis. Circ Res 122:1722–1740. https://doi.org/10.1161/circresaha.118.311362
Wytrykowska A, Prosba-Mackiewicz M, Nyka WM (2016) IL-1beta, TNF-alpha, and IL-6 levels in gingival fluid and serum of patients with ischemic stroke. J Oral Sci 58:509–513. https://doi.org/10.2334/josnusd.16-0278
Dinarello CA (2019) The IL-1 family of cytokines and receptors in rheumatic diseases. Nat Rev Rheumatol 15:612–632. https://doi.org/10.1038/s41584-019-0277-8
Acknowledgements
This study was funded by The National Natural Science Foundation of China (31671211), Science and Technology Planning Project of Guangdong Province, China (2017B030314056), frontier Research Program of Guangzhou Regenerative Medicine and Health Guangdong Laboratory (2018GZR110105020) and Guangdong Provincial Natural Science Foundation (2017A030313757 and 2016A030313170). Kind gift of THP-1 cell line from Dr. Peng Li, Guangzhou Institute of Biomedicine and Health, Chinese Academy of Sciences, China and kind gift of rat TRPA1 plasmid from Dr. Zhaobing Gao, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, China, are deeply appreciated.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tian, C., Han, X., He, L. et al. Transient receptor potential ankyrin 1 contributes to the ATP-elicited oxidative stress and inflammation in THP-1-derived macrophage. Mol Cell Biochem 473, 179–192 (2020). https://doi.org/10.1007/s11010-020-03818-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-020-03818-3