Abstract
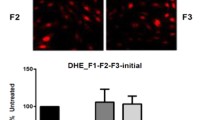
Bioactivities including cellular antioxidant activity in HepG2 and dipeptidylpeptidase-IV inhibitory effect of in vitro transepithelial transported peptides with ACE inhibitory activity from cooked chicken breast, namely, APP, KP, KPLL, LL, LVK and LVQ were investigated. HepG2 cells were the most susceptible to KP, however, none of them were toxic up to 100 µM. KP and KPLL showed cytoprotective effect and could reduce intracellular reactive oxygen species (ROS) level in a dose-dependent response (P < 0.05), particularly 100 µM KP showed comparable capacity with 100 µM ascorbic acid as demonstrated by using 2ʹ,7ʹ-dichlorofluorescein-diacetate (DCFH-DA) probe. KP induced expression of SOD1 and GPX1, while CAT appeared to be upregulated by KPLL (P < 0.05). Among the peptides studied, LL with an IC50 of 4.79 mM was the most active peptide against DPP-IV activity through a non-competitive action (Ki = 922.96 µM) as demonstrated by the Lineweaver–Burk plot, suggesting the ability of LL to bind with DPP-IV at various locations other than active site. Molecular docking revealed that LL interacted with the enzyme via van der Waals, salt bridge interactions, and hydrogen bonds.




Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Aguilar-Toalá JE, Santiago-López L, Peres CM et al (2017) Assessment of multifunctional activity of bioactive peptides derived from fermented milk by specific Lactobacillus plantarum strains. J Dairy Sci 100:65–75. https://doi.org/10.3168/jds.2016-11846
Casanova-Martí À, Bravo FI, Serrano J et al (2019) Antihyperglycemic effect of a chicken feet hydrolysate: Via the incretin system: DPP-IV-inhibitory activity and GLP-1 release stimulation. Food Funct 10:4062–4070. https://doi.org/10.1039/c9fo00695h
Chaicharoenaudomrung N, Jaroonwitchawan T, Noisa P (2018) Cordycepin induces apoptotic cell death of human brain cancer through the modulation of autophagy. Toxicol Vitr 46:113–121. https://doi.org/10.1016/j.tiv.2017.10.002
Chakrabarti AC (1994) Permeability of membranes to amino acids and modified amino acids: Mechanisms involved in translocation. Amino Acids 6:213–229. https://doi.org/10.1007/BF00813743
Chen HM, Muramoto K, Yamauchi F, Nokihara K (1996) Antioxidant activity of designed peptides based on the antioxidative peptide isolated from digests of a soybean protein. J Agric Food Chem 44:2619–2623. https://doi.org/10.1021/jf950833m
Chen T, Chen Z, Wang H et al (2018) Underlying action mechanism of a novel antioxidant peptide derived from Allium tuberosum Rottler protein hydrolysates and its protective effects on hydrogen peroxide induced cell injury. J Funct Foods 40:606–613. https://doi.org/10.1016/j.jff.2017.11.037
Chiu YW, Lo HJ, Huang HY et al (2013) The antioxidant and cytoprotective activity of Ocimum gratissimum extracts against hydrogen peroxide-induced toxicity in human HepG2 cells. J Food Drug Anal 21:253–260. https://doi.org/10.1016/j.jfda.2013.07.002
Connolly A, O’Keeffe MB, Nongonierma AB et al (2017) Isolation of peptides from a novel brewers spent grain protein isolate with potential to modulate glycaemic response. Int J Food Sci Technol 52:146–153. https://doi.org/10.1111/ijfs.13260
Fukada Y, Mizutani S, Nomura S et al (2016) Antioxidant activities of a peptide derived from chicken dark meat. J Food Sci Technol 53:2476–2481. https://doi.org/10.1007/s13197-016-2233-9
Giri A, Ohshima T (2012) Bioactive Marine peptides. Nutraceutical value and novel approaches, 1st edn. Elsevier Inc.
Graziani D, Ribeiro JV V, Cruz VS, et al (2021) Oxidonitrergic and antioxidant effects of a low molecular weight peptide fraction from hardened bean (Phaseolus vulgaris ) on endothelium. 54:1–11. doi: https://doi.org/10.1590/1414-431X202010423
Gülçin İ (2012) Antioxidant activity of food constituents: an overview. Arch Toxicol 86(3):345–391. https://doi.org/10.1007/s00204-011-0774-2
Gülçin İ (2020) Antioxidants and antioxidant methods-An updated overview. Arch Toxicol 94(3):651–715. https://doi.org/10.1007/s00204-020-02689-3
Gülçin İ, Gören AC, Taslimi P, Alwasel SH, Kılıc O, Bursal E (2020) Anticholinergic, antidiabetic and antioxidant activities of Anatolian pennyroyal (Mentha pulegium)-Analysis of its polyphenol contents by LC-MS/MS. Biocatal Agric Biotechnol 23:101441. https://doi.org/10.1016/j.bcab.2019.101441
Hamzeh A, Noisa P, Yongsawatdigul J (2020) Characterization of the antioxidant and ACE-inhibitory activities of Thai fish sauce at different stages of fermentation. J Funct Foods 64:103699. https://doi.org/10.1016/j.jff.2019.103699
Hatanaka T, Inoue Y, Arima J et al (2012) Production of dipeptidyl peptidase IV inhibitory peptides from defatted rice bran. Food Chem 134:797–802. https://doi.org/10.1016/j.foodchem.2012.02.183
Hatanaka T, Kawakami K, Uraji M (2014) Inhibitory effect of collagen-derived tripeptides on dipeptidylpeptidase-IV activity. J Enzyme Inhib Med Chem 29:823–828. https://doi.org/10.3109/14756366.2013.858143
Homayouni-Tabrizi M, Asoodeh A, Soltani M (2017) Cytotoxic and antioxidant capacity of camel milk peptides: Effects of isolated peptide on superoxide dismutase and catalase gene expression. J Food Drug Anal 25:567–575. https://doi.org/10.1016/j.jfda.2016.10.014
Ighodaro OM, Akinloye OA (2018) First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alexandria J Med 54:287–293. https://doi.org/10.1016/j.ajme.2017.09.001
Ji W, Zhang C, Ji H (2017) Two novel bioactive peptides from Antarctic krill with dual angiotensin converting enzyme and dipeptidyl peptidase IV inhibitory activities. J Food Sci 82:1742–1749. https://doi.org/10.1111/1750-3841.13735
Jones G, Willett P, Glen RC et al (1997) Development and validation of a genetic algorithm for flexible docking. J Mol Biol 267:727–748. https://doi.org/10.1006/jmbi.1996.0897
Kazir M, Abuhassira Y, Robin A et al (2019) Extraction of proteins from two marine macroalgae, Ulva sp. and Gracilaria sp., for food application, and evaluating digestibility, amino acid composition and antioxidant properties of the protein concentrates. Food Hydrocoll 87:194–203. https://doi.org/10.1016/j.foodhyd.2018.07.047
Lacroix IME, Li-Chan ECY (2012) Evaluation of the potential of dietary proteins as precursors of dipeptidyl peptidase (DPP)-IV inhibitors by an in silico approach. J Funct Foods 4:403–422. https://doi.org/10.1016/j.jff.2012.01.008
Lan VTT, Ito K, Ohno M et al (2015) Analyzing a dipeptide library to identify human dipeptidyl peptidase IV inhibitor. Food Chem 175:66–73. https://doi.org/10.1016/j.foodchem.2014.11.131
Li YW, Li B (2013) Characterization of structure-antioxidant activity relationship of peptides in free radical systems using QSAR models: Key sequence positions and their amino acid properties. J Theor Biol 318:29–43. https://doi.org/10.1016/j.jtbi.2012.10.029
Liu H, Ye H, Sun C et al (2018a) Antioxidant activity in HepG2 cells, immunomodulatory effects in RAW 264.7 cells and absorption characteristics in Caco-2 cells of the peptide fraction isolated from Dendrobium aphyllum. Int J Food Sci Technol 53:2027–2036. https://doi.org/10.1111/ijfs.13783
Liu Z, Ren Z, Zhang J et al (2018b) Role of ROS and nutritional antioxidants in human diseases. Front Physiol 9:1–14. https://doi.org/10.3389/fphys.2018.00477
Lu Y, Lu P, Wang Y et al (2019) A novel dipeptidyl peptidase IV inhibitory tea peptide improves pancreatic β-cell function and reduces α-cell proliferation in streptozotocin-induced diabetic mice. Int J Mol Sci 20:1–15. https://doi.org/10.3390/ijms20020322
Malaypally SP, Liceaga AM, Kim KH et al (2015) Influence of molecular weight on intracellular antioxidant activity of invasive silver carp (Hypophthalmichthys molitrix) protein hydrolysates. J Funct Foods 18:1158–1166. https://doi.org/10.1016/j.jff.2014.06.011
Marseglia A, Dellafiora L, Prandi B et al (2019) Simulated gastrointestinal digestion of cocoa: detection of resistant peptides and in silico/in vitro prediction of their ace inhibitory activity. Nutrients. https://doi.org/10.3390/nu11050985
Mattson MP (2008) Hormesis defined. Ageing Res Rev 7:1–7. https://doi.org/10.1016/j.arr.2007.08.007
Miquel E, Alegría A, Barberá R, Farré R (2006) Casein phosphopeptides released by simulated gastrointestinal digestion of infant formulas and their potential role in mineral binding. Int Dairy J 16:992–1000. https://doi.org/10.1016/j.idairyj.2005.10.010
Mirzaei M, Mirdamadi S, Ehsani MR, Aminlari M (2018) Production of antioxidant and ACE-inhibitory peptides from Kluyveromyces marxianus protein hydrolysates: purification and molecular docking. J Food Drug Anal 26:696–705. https://doi.org/10.1016/j.jfda.2017.07.008
Mora L, Bolumar T, Heres A, Toldrá F (2017) Effect of cooking and simulated gastrointestinal digestion on the activity of generated bioactive peptides in aged beef meat. Food Funct 8:4347–4355. https://doi.org/10.1039/c7fo01148b
Nishitani S, Takehana K, Fujitani S, Sonaka I (2005) Branched-chain amino acids improve glucose metabolism in rats with liver cirrhosis. Am J Physiol Gastrointest Liver Physiol 288:G1292–G1300. https://doi.org/10.1152/ajpgi.00510.2003
Nongonierma AB, Fitzgerald RJ (2013) Inhibition of dipeptidyl peptidase IV (DPP-IV) by tryptophan containing dipeptides. Food Funct 4:1843–1849. https://doi.org/10.1039/c3fo60262a
Nongonierma AB, Hennemann M, Paolella S, Fitzgerald RJ (2017) Generation of wheat gluten hydrolysates with dipeptidyl peptidase IV (DPP-IV) inhibitory properties. Food Funct 8:2249–2257. https://doi.org/10.1039/c7fo00165g
Nongonierma AB, Mooney C, Shields DC, FitzGerald RJ (2013) Inhibition of dipeptidyl peptidase IV and xanthine oxidase by amino acids and dipeptides. Food Chem 141:644–653. https://doi.org/10.1016/j.foodchem.2013.02.115
Pantaleão SQ, Philot EA, de Resende-Lara PT et al (2018) Structural dynamics of DPP-4 and its influence on the projection of bioactive ligands. Molecules 23:1–10. https://doi.org/10.3390/molecules23020490
Picot L, Bordenave S, Didelot S, et al (2006) Antiproliferative activity of fish protein hydrolysates on human breast cancer cell lines
Saiga A, Okumura T, Makihara T et al (2003) Angiotensin I-converting enzyme inhibitory peptides in a hydrolyzed chicken breast muscle extract. J Agric Food Chem 51:1741–1745. https://doi.org/10.1021/jf020604h
Samaranayaka AGP, Kitts DD, Li-Chan ECY (2010) Antioxidative and angiotensin-I-converting enzyme inhibitory potential of a pacific hake (merlucclus productos) fish protein hydrolysate subjected to simulated gastrointestinal digestion and caco-2 cell permeation. J Agric Food Chem 58:1535–1542. https://doi.org/10.1021/jf9033199
Sangsawad P, Choowongkomon K, Kitts DD et al (2018a) Transepithelial transport and structural changes of chicken angiotensin I-converting enzyme (ACE) inhibitory peptides through Caco-2 cell monolayers. J Funct Foods 45:401–408. https://doi.org/10.1016/j.jff.2018.04.020
Sangsawad P, Roytrakul S, Choowongkomon K et al (2018b) Transepithelial transport across Caco-2 cell monolayers of angiotensin converting enzyme (ACE) inhibitory peptides derived from simulated in vitro gastrointestinal digestion of cooked chicken muscles. Food Chem 251:77–85. https://doi.org/10.1016/j.foodchem.2018.01.047
Sangsawad P, Roytrakul S, Yongsawatdigul J (2017) Angiotensin converting enzyme (ACE) inhibitory peptides derived from the simulated in vitro gastrointestinal digestion. J Funct Foods 29:77–83. https://doi.org/10.1016/j.jff.2016.12.005
Segura-Campos M, Chel-Guerrero L, Betancur-Ancona D, Hernandez-Escalante VM (2011) Bioavailability of bioactive peptides. Food Rev Int 27:213–226. https://doi.org/10.1080/87559129.2011.563395
Shen W, Matsui T (2019) Intestinal absorption of small peptides: a review. Int J Food Sci Technol 54:1942–1948. https://doi.org/10.1111/ijfs.14048
Sila A, Alvarez OM, Haddar A et al (2016) Purification, identification and structural modelling of DPP-IV inhibiting peptides from barbel protein hydrolysate. J Chromatogr B Anal Technol Biomed Life Sci 1008:260–269. https://doi.org/10.1016/j.jchromb.2015.11.054
Slim MK (2001) Cardiovascular actions of chicken-meat extract in normo- and hypertensive rats. Br J Nutr 86:97–103. https://doi.org/10.1079/bjn2001367
Taslimi P, Gulçin İ (2018) Antioxidant and anticholinergic properties of olivetol. J Food Biochem 42:e12516. https://doi.org/10.1111/jfbc.12516
Uchida M, Ohshiba Y, Mogami O (2011) Novel dipeptidyl peptidase-4-inhibiting peptide derived from β-lactoglobulin. J Pharmacol Sci 117:63–66. https://doi.org/10.1254/jphs.11089SC
Van Amerongen A, Beelen-Thomissen MJC, Van Zeeland-Wolbers LAM, et al (2009) Egg protein hydrolysates
Van Dijk A, Van Eldik M, Veldhuizen EJA et al (2016) Immunomodulatory and anti-inflammatory activities of chicken cathelicidin-2 derived peptides. PLoS ONE 11:1–16. https://doi.org/10.1371/journal.pone.0147919
Vilcacundo R, Martínez-Villaluenga C, Miralles B, Hernández-Ledesma B (2019) Release of multifunctional peptides from kiwicha (Amaranthus caudatus) protein under in vitro gastrointestinal digestion. J Sci Food Agric 99:1225–1232. https://doi.org/10.1002/jsfa.9294
Wang B, Xie N, Li B (2019) Influence of peptide characteristics on their stability, intestinal transport, and in vitro bioavailability: A review. J Food Biochem 43:1–6. https://doi.org/10.1111/jfbc.12571
Wolfe KL, Rui HL (2007) Cellular antioxidant activity (CAA) assay for assessing antioxidants, foods, and dietary supplements. J Agric Food Chem 55:8896–8907. https://doi.org/10.1021/jf0715166
Yoshii K, Ogasawara M, Wada J et al (2020) Exploration of dipeptidyl-peptidase IV (DPP IV) inhibitors in a low-molecular mass extract of the earthworm Eisenia fetida and identification of the inhibitors as amino acids like methionine, leucine, histidine, and isoleucine. Enzyme Microb Technol 137:109534. https://doi.org/10.1016/j.enzmictec.2020.109534
Young D, Nau F, Pasco M, Mine Y (2011) Identification of hen egg yolk-derived phosvitin phosphopeptides and their effects on gene expression profiling against oxidative stress-induced caco-2 cells. J Agric Food Chem 59:9207–9218. https://doi.org/10.1021/jf202092d
Zhang Q, Tong X, Qi B et al (2018) Changes in antioxidant activity of Alcalase-hydrolyzed soybean hydrolysate under simulated gastrointestinal digestion and transepithelial transport. J Funct Foods 42:298–305. https://doi.org/10.1016/j.jff.2018.01.017
Zhang Y, Guo K, LeBlanc RE et al (2007) Increasing dietary leucine intake reduces diet-induced obesity and improves glucose and cholesterol metabolism in mice via multimechanisms. Diabetes 56:1647–1654. https://doi.org/10.2337/db07-0123
Zou T, He T, Li H, et al (2016) The structure-activity relationship of the antioxidant peptides from natural proteins. Molecules 21:72. https://doi.org/10.3390/molecules21010072
Funding
This work was financially supported by the National Research Council of Thailand under the project Food Innovation for Safety and Value Creation of Nakhonchaiburin (SUT3-305-61-12-06). Postdoctoral fellowship of AH was supported by Suranaree University of Technology (SUT), by Thailand Science Research and Innovation (TSRI), and by National Science, Research, and Innovation Fund (NSRF, Project no. 90464).
Author information
Authors and Affiliations
Contributions
JY, the PI, conceived overall experiment and write/correct the manuscript. AH conducted the experiments, data analysis and the manuscript preparation. PS conducted DPP-IV related experiments. PN gave advice on cellular studies. KC performed molecular docking.
Corresponding author
Ethics declarations
Conflict of Interest
All authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hamzeh, A., Sangsawad, P., Noisa, P. et al. Bioactivities of In Vitro Transepithelial Transported Peptides from Cooked Chicken Breast. Int J Pept Res Ther 28, 31 (2022). https://doi.org/10.1007/s10989-021-10326-5
Accepted:
Published:
DOI: https://doi.org/10.1007/s10989-021-10326-5