Abstract
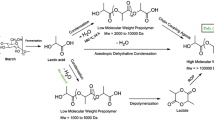
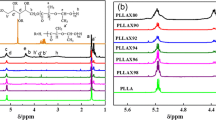
Biodegradable four-arm star-shaped poly(lactic acid) (4sPLA) was synthesized from l-Lactic acid (l-LA) and pentaerythritol (PENTA), and the polymerization kinetics was studied. The effects of reaction time, reaction temperature and molar ratio on the polymerization of 4sPLA were discussed. The molecular structure of 4sPLA was characterized by Fourier transform infrared spectroscopy (FTIR) and 1H nuclear magnetic resonance spectra (1H-NMR). The results showed that the optimum reaction conditions were as follows: the molar ratio of l-LA to PENTA was 12:1, and the polymerization reaction occurred at 160 ℃ for 5 h. Gel permeation chromatography method was used to determine the polymerization kinetics of 4sPLA consistent with the first-order reaction kinetics.













Similar content being viewed by others
References
Norazlina H, Kamal Y (2015) Polym Bull 72:931–961
Garlotta D (2001) J Polym Environ 9:63–84
Zaaba NF, Jaafar M (2020) Polym Eng Sci 60:2061–2075
Braun B, Dorgan JR, Knauss DM (2006) J Polym Environ 14:49–58
Abu Ghalia M, Dahman Y (2018) J Polym Environ 26:1903–1919
Liao QH, Peng XF, Fang H, Turng LS, Huang A, Chang CC (2020) J Polym Environ 28:295–303
Jiang N, Yu T, Li Y, Pirzada TJ, Marrow TJ (2019) Compos Sci Technol 173:15–23
Burgos N, Tolaguera D, Fiori S, Jimenez A (2014) J Polym Environ 22:227–235
Zheng L, Geng ZX, Zhen WJ (2019) J Polym Res 26:78
Fang F, Huo SQ, Shen HF, Ran SY, Wang H, Song PA, Fang ZP (2020) Compos Commun 17:104–108
Fang F, Ran SY, Fang ZP, Song PA, Wang H (2019) Compos Part B-Eng 165:406–416
Sai T, Ran SY, Guo ZH, Yan HQ, Zhang Y, Wang H, Song PG, Fang ZP (2021) Chem Eng J 409:128223
Chomachayi MD, Jalali-arani A, Urreaga JM (2021) J Polym Environ 29:2585–2597
Chen P, Yu KS, Wang YQ, Wang WB, Zhou HF, Li HQ, Mi JG, Wang XD (2018) J Polym Environ 26:3718–3730
Shin BY, Han DH, Narayan R (2010) J Polym Environ 18:558–566
Michalski A, Brzezinski M, Lapienis G, Biela T (2019) Prog Polym Sci 89:159–212
Jahandideh A, Muthukumarappan K (2017) Eur Polym J 87:360–379
Hachana N, Wongwanchai T, Chaochanchaikul K, Harnnarongchai W (2017) J Polym Environ 25:323–333
Yan XQ, Li JB, Ren TB (2018) E-Polymers 18:559–568
Jing ZX, Shi XT, Zhang GC (2016) Polym Composite 37:2744–2755
Lee SH, Kim SH, Han YK, Kim YH (2001) J Polym Sci Pol Chem 39:973–985
Esmaeili N, Jahandideh A, Muthukumarappan K, Akesson D, Skrifvars M (2017) J Appl Polym Sci 134:45341
Choi EJ, Son B, Hwang TS, Hwang EH (2011) J Ind Eng Chem 17:691–695
Salaam LE, Dean D, Bray TL (2006) Polymer 47:310–318
Zhang CX, Wang B, Chen Y, Cheng F, Jiang SC (2012) Polymer 53:3900–3909
Cameron DJA, Shaver MP (2011) Chem Soc Rev 40:1761–1776
Yuan MW, He ZG, Li HL, Jiang L, Yuan ML (2014) Polym Bull 71:1331–1347
Gorczynski JL, Chen JB, Fraser CL (2005) J Am Chem Soc 127:14956–14957
Tsuji H, Miyase T, Tezuka Y, Saha SK (2005) Biomacromol 6:244–254
Costa LI, Tancini F, Hofmann S, Codari F, Trommsdorff U (2016) Macromol Symp 360:40–48
Chu SL, Li X, Robertson AW, Sun ZY (2021) Acta Phys-Chim Sin 37:2009023
Medina DA, Contreras JM, Lopez-Carrasquero FJ, Cardozo EJ, Contreras RR (2018) Polym Bull 75:1253–1263
Moravek SJ, Messman JM, Storey RF (2009) J Polym Sci Pol Chem 47:797–803
George KA, Schue F, Chirila TV, Wentrup-Byrne E (2009) J Polym Sci Pol Chem 47:4736–4748
Burdick JA, Lovestead TM, Anseth KS (2003) Biomacromol 4:149–156
Sekkar V, Devi KA, Ninan KN (2001) J Appl Polym Sci 79:1869–1876
Fukuda T (2004) J Polym Sci Pol Chem 42:4743–4755
Akesson D, Skrifvars M, Seppala J, Turunen M, Martinelli A, Matic A (2010) J Appl Polym Sci 115:480–486
Jahandideh A, Muthukumarappan K (2016) Eur Polym J 83:344–358
Bakare FO, Ramamoorthy SK, Akesson D, Skrifvars M (2016) Compos Part a-Appl S 83:176–184
Zhao YL, Qing C, Jing J, Shuai XT, Bei JZ, Chen CF, Fu X (2002) Polymer 43:5819–5825
Ehsani M, Khodabakhshi K, Asgari M (2014) E-Polymers 14:353–361
Eldessouki M, Buschle-Diller G, Gowayed Y (2016) Des Monomers Polym 19:180–192
Kundys A, Plichta A, Florjanczyk Z, Zychewicz A, Lisowska P, Parzuchowski P, Wawrzynska E (2016) Polym Int 65:927–937
Nagahama K, Ohya Y, Ouchi T (2006) Polym J 38:852–860
Acknowledgements
This work is supported by National Natural Science Foundation of China (No. 11872279, No. 12172258 and No. 11625210) and Natural Science Foundation of Shanghai (No. 18ZR1440700).
Funding
Funding was provided by National Natural Science Foundation of China (Grant Nos.: 11872279, 12172258, 11625210) and by Natural Science Foundation of Shanghai (Grant No.: 18ZR1440700)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interests
There are no competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
He, J., Yu, T., Yang, W. et al. Biodegradable Star-Shaped Poly(lactic acid): Synthesis, Characterization and Its Reaction Kinetics. J Polym Environ 30, 3121–3128 (2022). https://doi.org/10.1007/s10924-022-02416-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-022-02416-2