Abstract
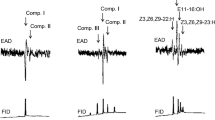
The common grass yellow Eurema mandarina has a characteristic patch (sex brand) composed of specialized scales (androconia) and wing intermembranous cells on the ventral surface of its male forewing. This structure is specific to males and is thought to release compounds that induce female mate acceptance. However, no study has demonstrated that these compounds function as sex pheromones in the genus Eurema. Here we report the identification of sex pheromones in males of E. mandarina. Chemical analyses revealed that 6,10,14-trimethylpentadecan-2-one (TMP) and (E/Z)-3,7,11,15-tetramethylhexadec-2-enal [(E/Z)-phytal] were male-specific and abundant in particular regions of the male forewings. TMP was highest in the sex brand, whereas (E/Z)-phytal was concentrated in the anal cell (cell 2 A), lacking androconia and intermembranous cells. The content of these compounds increases with age in males after emergence. In bioassays, virgin females displayed a posture of bending their abdomens as mating acceptance in response to stimulation by fresh male forewings. However, solvent-washed male wings did not induce such female responses, suggesting that some compounds from male wings serve as triggers. When we examined female responses to compounds applied to solvent-washed male wings, authentic TMP and (E/Z)-phytal alone showed little activity. However, the mixture elicited abdomen-bending responses in one-third of the females. Therefore, TMP and (E/Z)-phytal were found to act synergistically as aphrodisiac sex pheromones for E. mandarina females, although these activities were weak.







Similar content being viewed by others
Data Availability
All the data used in the present genetic analyses are included in this published article and its supplementary information.
References
Ahlquist L, Bergström G, Liljenberg C (1978) Acyclic diterpene alcohols: occurrence and synthesis of geranylcitronellol, phytol and geranylgeraniol. Prog Chem Fats other Lipids 16:231–255
Aoki S (2004) Steel-Dwass nonparametric multiple comparison test (in Japanese). https://aoki2.si.gunma-u.ac.jp/R/Steel-Dwass.html Accessed 5 Mar 2021
Bacquet PMB, Brattström O, Wang H-L, Allen CE, Löfstedt C, Brakefield PM, Nieberding CM (2014) Selection on male sex pheromone composition contributes to butterfly reproductive isolation. Proc R Soc B 282:20142734
Boppré M (1984) Chemically mediated interactions between butterflies. In: Vane-Wright RI, Ackery PR (eds) The biology of butterflies. Academic Press, London, pp 259–275
Constanzo K, Monteiro A (2007) The use of chemical and visual cues in female choice in the butterfly Bicyclus anynana. Proc R Soc B 274:845–851
Darragh K, Vanjari S, Mann F, Gonzalez-Rojas MF, Morrison CR, Salazar C, Pardo-Diaz C, Merrill RM, McMillan WO, Schulz S, Jiggins CD (2017) Male sex pheromone components in Heliconius butterflies released by the androconia affect female choice. PeerJ 5:e3953
Darragh K, Montejo-Kovacevich G, Kozak KM, Morrison CR, Figueiredo CME, Ready JS, Salazar C, Linares M, Byers KJRP, Merrill RM, McMillan WO, Schulz S, Jiggins CD (2020) Species specificity and intraspecific variation in the chemical profiles of Heliconius butterflies across a large geographic range. Ecol Evol 10:3895–3918
Eltz T, Hedenström E, Bång J, Wallin EA, Andersson J (2010) (6R,10R)-6,10,14-Trimethylpentadecan-2-one, a dominant and behaviorally active component in male orchid bee fragrances. J Chem Ecol 36:1322–1326
Fürstenau B, Muñoz L, Coca-Abia M, Rosell G, Guerrero A, Quero C (2013) Phytal: a candidate sex pheromone component of the Moroccan locust Dociostaurus maroccanus. ChemBioChem 14:450–1459
Grula JW, McChesney JD, Taylor OR Jr (1980) Aphrodisiac pheromones of the sulfur butterflies Colias eurytheme and C. philodice (Lepidoptera, Pieridae). J Chem Ecol 6:241–256
Guerrero A, Ramos VE, López S, Alvarez JM, Domínguez A, Coca-Abia M, Bosch MP, Quero C (2019) Enantioselective synthesis and activity of all diastereoisomers of (E)-phytal, a pheromone component of the Moroccan locust, Dociostaurus maroccanus. J Agric Food Chem 67:72–80
Hedenström E, Wallin EA, Andersson J, Bång J, Wang H-L, Löfstedt C, Brattström O, Baquet P (2015) Stereoisomeric analysis of 6,10,14-trimethylpentadecan-2-ol and the corresponding ketone in wing extracts from African Bicyclus butterfly species. J Chem Ecol 51:44–51
Hervé M (2019) RVAideMemoire: Testing and Plotting Procedures for Biostatistics. R package version 0:9–73
Honda (2008) Addiction to pyrrolizidine alkaloids in male danaine butterflies: a quest for the evolutionary origin of pharmacophagy. In: Maes RP (ed) Insect Physiology: New Research. Nova Science Publishers, New York, pp 73–118
Jeratthitikul E, Lewvanich A, Butcher BA, Lekprayoon C (2009) A taxonomic study of the genus Eurema Hübner, [1819] (Lepidoptera: Pieridae) in Thailand. Nat Hist J Chulalongkorn Univ 9:1–20
Kato Y (1989) Differences in reproductive behavior among seasonal wing morphs of the butterfly Eurema hecabe. J Insect Behav 2:419–429
Kato Y, Nakane T (1989) Male approach to pupae in the yellow butterfly, Eurema hecabe. J Ethol 7:59–61
Kemp DJ (2006) Ultraviolet ornamentation and male mating success in a high-density assemblage of the butterfly Colias eurytheme. J Insect Behav 19:669–684
Kemp DJ (2007) Female mating biases for bright ultraviolet iridescence in the butterfly Eurema hecabe (Pieridae). Behav Ecol 19:1–8
Kemp DJ, Rutowski RL (2011) The role of coloration in mate choice and sexual interactions in butterflies. Adv Study Behav 43:55–92
Khan D, Sahito ZA (2012) Eurema hecabe (Linnaeus, 1758) [Lepidoptera: Pieridae], a serious seedling pest of Acacia stenophylla A. Cunn. Ex. Benth., in Karachi. Int J Biol Biotech 9:307–312
Kristensen NP, Simonsen TJ (2003) ‘Hairs’ and scales. In: Kristensen NP (ed) Lepidoptera, moths and butterflies. Volume 2: Morphology, physiology, and development. Walter de Gruyter, Berlin, pp 9–22
Mérot C, Frérot B, Leppik E, Joron M (2015) Beyond magic traits: Multimodal mating cues in Heliconius butterflies. Evolution 69:2891–2904
Mohamed MA, Quisenberry SS, Moellenbeck DJ (1992) 6,10,14-Trimethylpentadecan-2-one: a Bermuda grass phagostimulant to fall armyworm (lepidoptera: Noctuidae). J Chem Ecol 18:673–682
Nieberding CM, de Vos H, Schneider MV, Lassance J-M, Estramil N, Andersson J, Bång J, Hedenström E, Löfstedt C, Brakefield PM (2008) The male sex pheromone of the butterfly Bicyclus anynana: towards an evolutionary analysis. PLoS ONE 3:e2751
Nieberding CM, Fischer K, Saastamoinen M, Allen CE, Wallin EA, Hedenström E, Brakefield PM (2012) Cracking the olfactory code of a butterfly: the scent of ageing. Ecol Lett 15:415–424
Nishida R, Schulz S, Kim CS, Fukami H, Kuwahara Y, Honda K, Hayashi N (1996) Male sex pheromone of a giant danaine butterfly, Idea leuconoe. J Chem Ecol 22:949–972
Nobre CEB, Lucas LAS, Padilha RJR, Navarro DMAF, Alves LC, Maia ACD (2021) Specialized androconial scales conceal species-specific semiochemicals of sympatric sulphur butterflies (Lepidoptera: Pieridae: Coliadinae). Org Divers Evol 21:93–105
Okumura Y, Ozeki Y, Itoh T, Ohta S, Ômura H (2016) Volatile terpenoids from male wings lacking scent scales in Anthocharis scolymus (Lepidoptera: Pieridae). Appl Entomol Zool 51:385–392
Ômura H, Yotsuzuka S (2015) Male-specific epicuticular compounds of the sulfur butterfly Colias erate poliographus (Lepidoptera: Pieridae). Appl Entomol Zool 50:191–199
Ômura H, Noguchi T, Ohta S (2020) The male swallowtail butterfly, Papilio polytes, uses cuticular hydrocarbons for mate discrimination. Anim Behav 170:133–145
Ômura H, Morozumi Y, Noguchi T, Ohta S (2021) Variation in cuticular lipid profiles of black butterflies of the genus Papilio in Japan. Biochem Syst Ecol 96:104265
Ômura H, Noguchi T, Ohta S (2022) Chemical identity of cuticula lipid components in the mimetic swallowtail butterfly Papilio polytes. Chem Biodivers 19:e202100879
Papke RS, Kemp DJ, Rutowski RL (2007) Multimodal signalling: structural ultraviolet reflectance predicts male mating success better than pheromones in the butterfly Colias eurytheme L. (Pieridae). Anim Behav 73:47–54
Pliske TE (1975) Courtship behavior and use of chemical communication by males of certain species of ithomiine butterflies (Nymphalidae: Lepidoptera). Ann Entomol Soc Am 68:935–942
R Development Core Team (2019) R: A language and environment for statistical computing. Vienaa, Austria: R Foundation for Statistical Computing. https://www.R-project.org
Rutowski RL (1977a) The use of visual cues in sexual and species discrimination by males of the small sulphur butterfly Eurema lisa (Lepidoptera, Pieridae). J Comp Physiol A 115:61–74
Rutowski RL (1977b) Chemical communication in the courtship of the small sulphur butterfly Eurema lisa (Lepidoptera, Pieridae). J Comp Physiol A 115:75–85
Rutowski RL (1978) The courtship behaviour of the small sulphur butterfly, Eurema lisa (Lepidoptera: Pieridae). Anim Behav 26:892–903
Rutowski RL (1980) Male scent-producing structures in Colias butterflies–function, localization, and adaptive features. J Chem Ecol 6:13–26
Sappington TW, Taylor OR (1990a) Developmental and environmental sources of pheromone variation in Colias eurytheme butterflies. J Chem Ecol 16:2771–2786
Sappington TW, Taylor OR (1990b) Disruptive sexual selection in Colias eurytheme butterflies. Proc Natl Acad USA 87:6132–6135
Sasaerila Y, Gries R, Gries G, Khaskin G, King S, Takács S, Hardi(2003) Sex pheromone components of male Tirathaba mundella (Lepidoptera: Pyralidae). Chemoecology 13:89–93
Schiestl FP, Wallin EA, Beck JJ, Friberg M, Thompson JN (2021) Generalized olfactory detection of floral volatiles in the highly specialized Greya-Lithophragma nursery pollination system. Arthrop Plant Interact 15:209–221
Schulz S, Nishida R (1996) The pheromone system of the male danaine butterfly, Idea leuconoe. Bioorg Medic Chem 4:341–349
Schulz S, Boppré M, Vane-Wright RJ (1993) Specific mixtures of secretions from male scent organs of African milkweed butterflies (Danainae). Philos Trans R Soc Lond B 342:161–181
Schulz S, Yildizhan S, van Loon JJA (2011) The biosynthesis of hexahydrofarnesylacetone in the butterfly Pieris brassicae. J Chem Ecol 37:360–363
Shimomura K, Matsui S, Ohsawa K, Yajima S (2016) Saltational evolution of contact sex pheromone compounds of Callosobruchus rhodesianus (Pic). Chemoecology 26:15–23
Silberglied RE (1984) Visual communication and sexual selection among butterflies. In: Vane-Wright RI, Ackery PR (eds) The biology of butterflies. Academic Press, London, pp 207–223
Silberglied RE, Taylor OR(1973) Ultraviolet differences between sulfur butterflies, Colias eurytheme and C. philodice, and a possible isolating mechanism. Nature 241:406–408
Silberglied RE, Taylor OR(1978) Ultraviolet reflection and its behavioral role in the courtship of the sulphur butterflies Colias eurytheme and C. philodice (Lepidoptera, Pieridae). Behav Ecol Sociobiol 3:203–243
Takanashi T, Hiroki M, Obara Y (2001) Evidence for male and female sex pheromones in the sulfur butterfly, Eurema hecabe. Entomol Exp Appl 101:89–92
Vane-Wright RI, Boppré M (1993) Visual and chemical signalling in butterflies: functional and phylogenetic perspectives. Phil Trans R Soc Lond B 340:197–205
Vetter RS, Rutowski RL (1978) External sex brand morphology of three sulphur butterflies (Lepidoptera: Pieridae). Psyche 85:383–393
Wallin EA, Kalinová B, Kindl J, Hedenström E, Valterová I(2020) Stereochemistry of two pheromonal components of the bumblebee wax moth, Aphomia sociella. Sci Rep 10:2094
Yildizhan S, van Loon J, Sramkova A, Ayasse M, Arsene C, ten Broeke C, Schulz S(2009) Aphrodisiac pheromones from the wings of the small cabbage white and large cabbage white butterflies, Pieris rapae and Pieris brassicae. Chem Bio Chem 10:1666–1667
Acknowledgements
We are thankful to Dr. Tomoko Amimoto, at the Natural Science Center for Basic Research and Development (N-BARD), Hiroshima University, for the GC–HR–TOFMS measurements.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Chemical analyses and morphological observations were performed by C. Okuda, K. Yoshimori, and H. Ômura. Bioassays were performed by K. Yoshimori and H. Ômura. Statistical analyses were performed by H. Ômura. The first draft of the manuscript was written by H. Ômura, with editorial inputs from K. Yoshimori, C. Okuda, and S. Ohta. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yoshimori, K., Okuda, C., Ohta, S. et al. Sex pheromones from male forewings of the Common Grass Yellow Eurema mandarina. J Chem Ecol 48, 518–530 (2022). https://doi.org/10.1007/s10886-022-01368-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-022-01368-0