Abstract
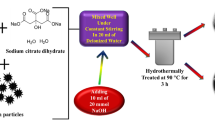
The degradation of organic pollutants in water has become a research hotspot in the environmental field. Development of photocatalysts with enhanced photocatalytic activity is of great concern for environmental and energy applications. Herein, different morphologies of ZnO with enhanced photocatalytic activity was synthesized by a facile hydrothermal method with the assistance of ethylene glycol (EG). EG is a key factor for the preparation of structures of ZnO materials. The volume ratio of EG to H2O also plays an important role on the morphology control of ZnO and different proportions have significantly different effects on the morphologies and performances. The obtained photocatalysts were characterized by X-ray powder diffraction, scanning electron microscopy, Brunauer, Emmett and Teller specific surface area (BET), nitrogen adsorption–desorption isotherm and Barrett-Joyner-Halenda pore diameter distribution, UV–Vis diffuse reflectance spectroscopy, photoluminescence spectroscopy and fluorescence lifetime spectroscopy, respectively. The photocatalytic activity of the photocatalyst was evaluated using Rhodamine B as a target pollutant. When the volume ratio of EG to H2O is 6:9, it is found that ZnO with the flower clusters like, average dimensions of 5.25 μm, crystalline size of 46.3 nm and BET-specific surface area of 91.0 m2/g, which photocatalytic efficiency is much higher than other samples, and the degradation rate of RhB reaches to 94.1%, photocatalytic rate is 0.015 min−1. The quantum yield of the photocatalytic reaction is 7.5%. This work could provide a facile way to fabricate the photocatalysts and has great potential in environmental remediation with efficient photocatalytic activity.










Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
D. Toloman, A. Popa, M. Stan, M. Stefan, O. Pana, J. Alloys Compds. 866, 159010 (2021)
A. Barnasas, M.V. Karavasilis, C. Aggelopoulos, C.D. Tsakiroglou, P. Poulopoulos, J. Nano Res. 62, 75–86 (2020)
S. Dong, J. Feng, M. Fan, Y. Pi, L. Hu, H. Xiao, M. Liu, J. Sun, J. Sun, RSC Adv. 5, 14610–14630 (2015)
B. Gsa, B. Aka, B. Ss, C. Mn, D. Pd, E. Dvnv, A. Fjs, Mater. Lett. 278, 128359 (2020)
Q. Chen, B. Jing, J. Li, H. Ke, X. Li, B. Zhou, W. Cai, Chem. Eng. J. 252, 89–94 (2014)
A. Fujishima, K. Honda, Nature 238, 37–38 (1972)
F.A. Fouad, M.A. Ahmed, M.S. Antonious, M.F. Abdel-Messih, J Mater. Sci. Mater. Electron. 31, 12355–12371 (2020)
A. Ismail, M.J. Abdullah, M.A. Qaeed, M.A. Khamis, W.A. Farooq, J. King Saud Univ. Sci. 33, 101229 (2021)
Shanshan, Xiao, Li, Liu, Jianshe, Lian J, Mater. Sci.: Mater. Electron. 25, 5518-5523 (2014)
M.A. Lahmer, Appl. Surf. Sci. 457, 315–322 (2018)
P. Dhamodharan, J. Chen, C. Manoharan, J. Mater. Sci. Mater. Electron. 32, 13418–13429 (2021)
Weilai, Zhang, Jinfeng, Peng, Tianyou, Appl. Catal., B. 181, 220-227 (2016)
M. Islam, A.K. Srivastava, B.M. Basavaraja, A. Sharma, Sens. Int. 2, 100084 (2021)
S.K. Kansal, M. Singh, D. Sud, J. Hazard. Mater. 141, 581–590 (2007)
N.H. Erdogana, T. Kutlub, N. Sedefogluc, H. Kavakb, J. Alloys Compds. 881, 160554 (2021)
A.M. Palve, S.S. Garje, Semicond. Sci. Technol. 36, 025007 (2021)
G.P. Singh, M.K. Roy, Mater. Today: Proc. (2021). https://doi.org/10.1016/j.matpr.2021.02.741
F. Wang, Y. Zhou, X. Pan, B. Lu, Z. Ye, Phys. Chem. Chem. Phys. 20, 6959–6969 (2018)
W. Si, L. Pei, Micro Nano Lett. 15, 751–754 (2020)
S. Sathiya, J. Vijayapriya, K. Parasuraman, D.B. Anburaj, S.J. Gnanamuthu, G. Nedunchezian, J. Metastable, Nanocryst. Mater. 32, 33–43 (2021)
M.Z. Mobaraki, A. Haghighatzadeh, Bull. Mater. Sci. 43, 1–11 (2020)
Y.N. Wang, J. Li, Opt. Mater. 108, 110203 (2020)
M.N. Rumyantseva, S.A. Vladimirova, V.B. Platonov, A.S. Chizhov, M. Batuk, J. Hadermann, N.O. Khmelevsky, A.M. Gaskov, Sens. Actuators B. 307, 127624 (2021)
Z. Wu, L. Wang, Mater. Lett. 234, 287–290 (2019)
H. Wen, H.V. Ngoc, D.J. Kang, J. Photochem. Photobiol. A. 356, 212–218 (2018)
J.M. Jang, S.D. Kim, H.M. Choi, J.Y. Kim, W.G. Jung, Mater. Chem. Phys. 113, 389–394 (2009)
I. Boukhoubza, M. Khenfouch, L. Leontie, M. Achehboune, A. Jorio, Mater. Today: Proc. (2021). https://doi.org/10.1016/j.matpr.2021.04.634
H. Li, S. Jiao, S. Bai, H. Li, S. Gao, J. Wang, Q. Yu, F. Guo, L. Zhao, Phys. Status Solidi A. 211, 595–600 (2014)
M. Kamruzzaman, J.A. Zapien, Crystallogr. Rep. 63, 456–471 (2018)
P. Scherrer, Nachr. Ges. Wiss. Göttingen. 26, 98 (1918)
J.I. Langford, A.J.C. Wilson, J. Appl. Cryst. 11, 102 (1978)
M.N. Ghazzal, N. Barthen, N. Chaoui, Appl. Catal. B. 103, 85–90 (2012)
F. Sun, X. Qiao, F. Tan, W. Wang, X. Qiu, Appl. Surf. Sci. 263, 704–711 (2012)
R. Singh, K. Verma, A. Patyal, I. Sharma, P.B. Barman, D. Sharma, Solid State Sci. 89, 1–14 (2019)
M. Taheri, H. Abdizadeh, M.R. Golobostanfard, J. Alloys Compd. 725, 291–301 (2017)
S. Zarezadeh, A. Habibi-Yangjeh, M. Mousavi, S. Ghosh, J. Photochem. Photobiol. A. 389, 112247 (2019)
Tongguang, Xu, Liwu, Zhang, Hanyun, Cheng, Yongfa, Appl. Catal., B. 101, 382–387 (2011)
Y. Zhang, J. Xu, P. Xu, Y. Zhu, W. Yu, Nanotechnol. 21, 285501 (2010)
Y. Chen, D. Zeng, K. Zhang, A. Lu, L. Wang, D.L. Peng, Nanoscale 6, 1–9 (2013)
A. Saljooqi, T. Shamspur, A. Mostafavi, Environ. Sci. Pollut. Res. 28, 9146–9156 (2020)
K.S. Al-Namshah, S.M. Mariappan, M. Shkir, M.S. Hamdy, Appl. Phys. A. 127, 1–10 (2021)
K. Qi, B. Cheng, J. Yu, W. Ho, J. Alloys Compds. 727, 792–820 (2017)
Acknowledgements
This work was supported by the Fundamental Research Funds for the Central Universities (Grant Nos. 2412021QD008), the Industrial Technology Research and Development Projects of Jilin Province (No.2019C059), Research and Development Project of Ministry of Housing and Urban-Rural Development (2019-K-050)and the Science and Technology Development Project of Jilin Province (No. 20180520217JH).
Author information
Authors and Affiliations
Contributions
Investigation, data curation, formal analysis and writing-original draft (MZ); supervision [GG]; Writing-review (FB); conceptualization (LW].
Corresponding authors
Ethics declarations
Conflict of interest
There are no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, M., Gai, G., Bi, F. et al. Synthesis of different morphologies of ZnO via hydrothermal method for enhanced photocatalytic degradation of wastewater. J Mater Sci: Mater Electron 33, 4523–4534 (2022). https://doi.org/10.1007/s10854-021-07642-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-021-07642-3