Abstract
Purpose
Pterygium, a degenerative and hyperplastic lesion, has premalignant properties as a tumor analog. WWOX is a tumor suppressor gene and involved in many signal pathways, such as cell proliferation, embryonic development, metabolism and apoptosis. In many cancers, the loss of WWOX or the presence of abnormal transcripts indicates the tumor suppressor activity of WWOX. In this study, it was aimed to determine WWOX gene expression and protein levels in pterygium which may be a tumor analog.
Methods
For this purpose, the WWOX gene expression change in 27 pterygium tissue was investigated by real-time PCR method, and the change in WWOX protein was investigated using the Western blot method.
Results
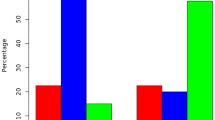
According to our results, it was found that the expression and protein levels of WWOX gene in pterygium tissue decreased significantly compared to control tissue (p < 0.05).
Conclusion
This information indicates that a decrease in expression and protein level in pterygium tissue of WWOX, a tumor suppressor gene, supports claims that pterygium may be a cancer analog tissue.

Similar content being viewed by others
References
Donald THT, Wen YT, Yan PL, Hak-Su G, Duncan RS (2000) Apoptosis and apoptosis related gene expression in normal conjunctiva and pterygium. Br J Ophthalmol 84:212–216
Cárdenas-Cantú E, Zavala J, Valenzuela J, Valdez-García JE (2016) Molecular basis of pterygium development. Semin Ophthalmol 31(6):567–583
Liu T, Liu Y, Xie L, He X, Ji Bai J (2013) Progress in the pathogenesis of pterygium. Curr Eye Res 38(12):1191–1197
Sebastiá R, Ventura MP, Solari HP, Antecka E, Orellana ME, Burnier MN (2013) Immunohistochemical detection of Hsp90 and Ki-67 in pterygium. Diagn Pathol 8:32
Demiryürek S, Saracaloglu A, Kimyon S, Mete A, Eronat O, Temiz E, Nacarkahya G, Tunca ZS, Düzen B, Saygili O, Güngör K, Karakök M, Demiryürek AT (2019) Increased expressions of ICAM-2 and ICAM-3 in pterygium. Curr Eye Res. https://doi.org/10.1080/02713683.2019.1570527
Liang K, Jiang Z, Ding B, Cheng P, Huang D, Tao L (2011) Expression of cell proliferation and apoptosis biomarkers in pterygia and normal conjunctiva. Mol Vis 17:1687–1693
Girolamo ND, Chui J, Coroneo MT, Wakefield D (2004) Pathogenesis of pterygia: role of cytokines, growth factors, and matrix metalloproteinases. Prog Retin Eye Res 23:195–228
Huang YH, Chang NS, Tseng SH (2015) Expression of WW domain-containing oxidoreductase WWOX in pterygium. Mol Vis 21:711–717
Tung JN, Chiang CC, Tsai YY, Chou YY, Yeh KT, Lee H, Cheng YW (2010) CyclinD1 protein expressed in pterygia is associated with β-catenin protein localization. Mol Vis 16:2733–2738
Baryła I, Styczen-Binkowska E, Bednarek AK (2015) Alteration of WWOX in human cancer, a clinical view. Exp Biol Med 240:305–314
Schrock MS, Huebner K (2015) WWOX: a fragile tumor suppressor. Exp Biol Med 240:296–304
Yang Z, Lan H, Chen X, Li P, Li S, Mo W, Tang A (2014) Molecular alterations of the WWOX gene in nasopharyngeal carcinoma. Neoplasma 61(2):170–176
Lin JT, Li HY, Chang NS, Lin CH, Chen YC, Lu PJ (2015) WWOX suppresses prostate cancer cell progression through cyclin D1-mediated cell cycle arrest in the G1 phase. Cell Cycle 14(3):408–416
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3(6):1101–1108
Adıguzel U, Karabacak T, Sarı A, Oz O, Cınel L (2007) Cyclooxygenase-2 expression in primary and recurrent pterygium. Eur J Ophthalmol 17(6):879–884
Spandidos DA, Sourvinos G, Kiaris H, Tsamparlakis J (1997) Microsatellite instability and loss of heterozygosity in human pterygia. Br J Ophthalmol 81:493–496
Weinstein O, Rosenthal G, Zirkin H, Monos T, Lifshitz T, Argov S (2002) Overexpression of p53 tumor suppressor gene in pterygia. Eye 16:619–621
Wistuba II, Behrens C, Virmani AK (2000) High resolution chromosome 3p allelotyping of human lung cancer and preneoplastic/preinvasive bronchial epithelium reveals multiple, discontinuous sites of 3p allele loss and three regions of frequent breakpoints. Cancer Res 60:1949–1960
Nowakowska M, Płuciennik E, Wujcicka W, Sitkiewicz A, Kazanowska B, Zielińska E, Bednarek AK (2014) The correlation analysis of WWOX expression and cancer related genes in neuroblastoma—a real time RT-PCR study. Acta Biochim Pol 61(1):91–97
Abdeen SK, David UB, Shweiki A, Maly B, Aqeilan RI (2018) Somatic loss of WWOX is associated with TP53 perturbation in basal-like breast cancer. Cell Death Dis 9:832
Aldaz CM, Ferguson BW, Abba MC (1846) WWOX at the crossroads of cancer, metabolic syndrome related traits and CNS pathologies. Biochim Biophys Acta 2014:188–200
Płuciennik E, Nowakowska M, Wujcicka WI, Sitkiewicz A, Kazanowska B, Zielińska E, Bednarek AK (2012) Genetic alterations of WWOX in Wilms’ tumor are involved in its carcinogenesis. Oncol Rep 28(4):1417–1422
Qin HR, Iliopoulos D, Nakamura T, Costinean S, Volinia S, Druck T, Sun J, Okumura H, Huebner K (2007) Wwox suppresses prostate cancer cell growth through modulation of ErbB2-mediated androgen receptor signaling. Mol Cancer Res 5(9):957–965
Yang J, Zhang W (2008) WWOX tumor suppressor gene. Histol Histopathol 23:877–882
Nunez MI, Ludes-Meyers J, Abba MC, Kil H, Abbey NW, Page RE, Sahin A, Klein-Szanto AJ, Aldaz CM (2005) Frequent loss of WWOX expression in breast cancer: correlation with estrogen receptor status. Breast Cancer Res Treat 89:99–105
Chang R, Song L, Xu Y, Wu Y, Dai C, Wang X, Sun X, Hou Y, Li W, Zhan X, Zhan L (2018) Loss of Wwox drives metastasis in triple-negative breast cancer by JAK2/STAT3 axis. Nat Commun 9(1):3486
Ekizoglu S, Bulut P, Karaman E, Kilic E, Buyru N (2015) Epigenetic and genetic alterations affect the WWOX gene in head and neck squamous cell carcinoma. PLoS ONE. https://doi.org/10.1371/journal.pone.0115353
Acknowledgements
The authors thank Helin Deniz Demir for the critical review of the article and the kind assistance in obtaining the patient’s samples. This study was supported by Tokat Gaziosmanpaşa University Scientific Research Projects Unit and TUBITAK. Project Nos. 2015/26 (Tokat Gaziosmanpaşa University) and 215S692 (TUBITAK).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bozkurt, N., Ates, O. Analysis of WWOX gene expression and protein levels in pterygium. Int Ophthalmol 40, 1949–1953 (2020). https://doi.org/10.1007/s10792-020-01368-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-020-01368-7