Abstract
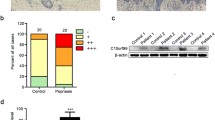
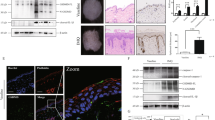
The mevalonate-diphosphate decarboxylase (MVD) gene, a member of the mevalonate pathway, plays a critical role in regulating the biosynthesis of cholesterol, steroid hormones, and non-steroid isoprenoids. Previous studies have suggested that the MVD c.746 T > C mutation is a major pathogenic gene of porokeratosis (PK), an autoinflammatory keratinization disease (AIKD) with unclear pathogenesis, few effective treatments, and no suitable animal model. To investigate the function of MvdF250S/+ mutation, we developed a novel MvdF250S/+ mouse model carrying an equivalent point mutation to the most common genetic variation among Chinese PK patients (MVDF249S/+) using CRISPR/Cas9 technology, which exhibited reduced cutaneous expression of Mvd protein. In the absence of external stimulation, MvdF250S/+ mice did not display specific phenotypes. However, upon induction with imiquimod (IMQ), MvdF250S/+ mice exhibited decreased susceptibility to skin acute inflammation compared to wild-type (WT) mice, as evidenced by reduced cutaneous proliferation and lower protein levels of IL-17a and IL-1β. Additionally, after IMQ induction, the MvdF250S/+mice exhibited downregulated collagen generation and upregulated expression of Fabp3 compared to WT mice, whereas no significant changes in the key genes related to cholesterol regulation were found. Furthermore, the MvdF250S/+ mutation activated autophagy. Our findings provided insights into the biological function of MVD in the skin.




Similar content being viewed by others
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- MVD:
-
Mevalonate diphosphate decarboxylase
- PK:
-
Porokeratosis
- AIKD:
-
Autoinflammatory keratinization disease
- IMQ:
-
Imiquimod
- WT:
-
Wild type
- sgRNA:
-
Single-guide RNA
- PASI:
-
Psoriasis area and severity index
- DEGs:
-
Differentially expressed genes
- ECM:
-
Extracellular matrix
References
Wong, W., Y. Huang, Z. Wu, et al. 2021. Mvda is required for zebrafish early development. Biological Research 54: 17. https://doi.org/10.1186/s40659-021-00341-7.
Martín Sánchez, C., J.M. Pérez Martín, J.-S. Jin, et al. 2015. Disruption of the mevalonate pathway induces dNTP depletion and DNA damage. Biochimica et Biophysica Acta 1851: 1240–1253. https://doi.org/10.1016/j.bbalip.2015.06.001.
Faulkner, R., Y. Jo. 2022. Synthesis, function, and regulation of sterol and nonsterol isoprenoids. Frontiers in Molecular Biosciences. 9: 1006822. Published 2022 Oct 5. https://doi.org/10.3389/fmolb.2022.1006822.
Zhang, Z., C. Li, F. Wu, et al. 2015. Genomic variations of the mevalonate pathway in porokeratosis. Elife 4: e06322. https://doi.org/10.7554/eLife.06322.
Li, M., Z. Li, J. Wang, et al. 2016. Mutations in the mevalonate pathway genes in Chinese patients with porokeratosis. Journal of the European Academy of Dermatology and Venereology 30: 1512–1517. https://doi.org/10.1111/jdv.13653.
Leng, Y., L. Yan, H. Feng, et al. 2018. Mutations in mevalonate pathway genes in patients with familial or sporadic porokeratosis. Journal of Dermatology 45: 862–866. https://doi.org/10.1111/1346-8138.14343.
Tao, L., Y.K. Huang, K.X. Yan, et al. 2022. A preliminary study of peripheral T-cell subsets in porokeratosis patients with MVK or MVD variants. Skin Health Disease 2: e82. https://doi.org/10.1002/ski2.82.
Takeichi, T., and M. Akiyama. 2019. Familial or sporadic porokeratosis as an autoinflammatory keratinization disease. Journal of Dermatology 46: e125–e126. https://doi.org/10.1111/1346-8138.14666.
Atzmony, L., and K.A. Choate. 2019. Second-hit somatic mutations in mevalonate pathway genes underlie porokeratosis. The Journal of Investigative Dermatology 139: 2409–2411. https://doi.org/10.1016/j.jid.2019.07.723.
Atzmony, L., H.M. Khan, Y.H. Lim, et al. 2019. Second-hit, postzygotic PMVK and MVD mutations in linear porokeratosis. JAMA Dermatology 155: 548–555. https://doi.org/10.1001/jamadermatol.2019.0016.
Biswas, A. 2015. Cornoid lamellation revisited: Apropos of porokeratosis with emphasis on unusual clinicopathological variants. American Journal of Dermatopathology 37: 145–155. https://doi.org/10.1097/DAD.0000000000000039.
Touitou, I. 2022. Twists and turns of the genetic story of mevalonate kinase-associated diseases: A review. Genes Dis 9: 1000–1007. https://doi.org/10.1016/j.gendis.2021.05.002.
Hivnor, C., N. Williams, F. Singh, et al. 2004. Gene expression profiling of porokeratosis demonstrates similarities with psoriasis. Journal of Cutaneous Pathology 31: 657–664. https://doi.org/10.1111/j.0303-6987.2004.00247.x.
Wang, M., S. Zhang, G. Zheng, et al. 2018. Gain-of-function mutation of Card14 leads to spontaneous psoriasis-like skin inflammation through enhanced keratinocyte response to IL-17A. Immunity 49: 66-79.e5. https://doi.org/10.1016/j.immuni.2018.05.012.
Son, S.-E., J.-M. Koh, and D.-S. Im. 2022. Free fatty acid receptor 4 (FFA4) activation ameliorates imiquimod-induced psoriasis in mice. International Journal of Molecular Sciences 23: 4482. https://doi.org/10.3390/ijms23094482.
Islam, A., Y. Kagawa, K. Sharifi, et al. 2014. Fatty acid binding protein 3 Is involved in n-3 and n-6 PUFA transport in mouse trophoblasts. Journal of Nutrition 144: 1509–1516. https://doi.org/10.3945/jn.114.197202.
Pang, B., Z. Zhu, C. Xiao, et al. 2021. Keratin 17 is required for lipid metabolism in keratinocytes and benefits epidermal permeability barrier homeostasis. Frontiers in Cell and Developmental Biology 9: 779257. https://doi.org/10.3389/fcell.2021.779257.
Wang, J., N. Kaplan, S. Wang, et al. 2020. Autophagy plays a positive role in induction of epidermal proliferation. The FASEB Journal 34: 10657–10667. https://doi.org/10.1096/fj.202000770RR.
Song, J., J. Jiang, L. Kuai, et al. 2022. TMT-based proteomics analysis reveals the protective effect of Jueyin granules on imiquimod-induced psoriasis mouse model by causing autophagy. Phytomedicine 96: 153846. https://doi.org/10.1016/j.phymed.2021.153846.
Pohl, C., and I. Dikic. 2019. Cellular quality control by the ubiquitin-proteasome system and autophagy. Science 366: 818–822. https://doi.org/10.1126/science.aax3769.
Chowdhari, S., and N. Saini. 2016. Gene expression profiling reveals the role of RIG1 like receptor signaling in p53 dependent apoptosis induced by PUVA in keratinocytes. Cellular Signalling 28: 25–33. https://doi.org/10.1016/j.cellsig.2015.10.015.
Tian, R., Y. Li, X. Yao. 2016. PGRN suppresses inflammation and promotes autophagy in keratinocytes through the Wnt/β-catenin signaling pathway. Inflammation 39: 1387–1394. https://doi.org/10.1007/s10753-016-0370-y.
Lee, H.-M., D.-M. Shin, J.-M. Yuk, et al. 2011. Autophagy negatively regulates keratinocyte inflammatory responses via scaffolding protein p62/SQSTM1. The Journal of Immunology 186: 1248–1258. https://doi.org/10.4049/jimmunol.1001954.
Kuzuyama, T., and H. Seto. 2012. Two distinct pathways for essential metabolic precursors for isoprenoid biosynthesis. Proceedings of the Japan Academy. Series B, Physical and Biological Sciences 88: 41–52. https://doi.org/10.2183/pjab.88.41.
Buhaescu, I., and H. Izzedine. 2007. Mevalonate pathway: A review of clinical and therapeutical implications. Clinical Biochemistry 40: 575–584. https://doi.org/10.1016/j.clinbiochem.2007.03.016.
Fu, Y., J.D. Sander, D. Reyon, et al. 2014. Improving CRISPR-Cas nuclease specificity using truncated guide RNAs. Nature Biotechnology 32: 279–284. https://doi.org/10.1038/nbt.2808.
Shan, H., Z. Liu, Y. Jia, et al. 2021. Reduced off-target effect of NG-BE4max by using NG-HiFi system. Mol Ther Nucleic Acids 25: 168–172. https://doi.org/10.1016/j.omtn.2021.05.012.
Buccitelli, C., and M. Selbach. 2020. mRNAs, proteins and the emerging principles of gene expression control. Nature Reviews Genetics 21: 630–644. https://doi.org/10.1038/s41576-020-0258-4.
Schroll, S., T.J. Lange, M. Arzt, et al. 2013. Effects of simvastatin on pulmonary fibrosis, pulmonary hypertension and exercise capacity in bleomycin-treated rats. Acta Physiologica 208: 191–201. https://doi.org/10.1111/apha.12085.
Kim, MJ., KL. Bible, M. Regnier, et al. 2019. Simvastatin provides long-term improvement of left ventricular function and prevents cardiac fibrosis in muscular dystrophy. Physiological Reports 7: e14018. https://doi.org/10.14814/phy2.14018.
Jang, Y.O., S.H. Kim, M.-Y. Cho, et al. 2018. Synergistic effects of simvastatin and bone marrow-derived mesenchymal stem cells on hepatic fibrosis. Biochemical and Biophysical Research Communications 497: 264–271. https://doi.org/10.1016/j.bbrc.2018.02.067.
Acikgoz, Y., B. Can, K. Bek, et al. 2014. The effect of simvastatin and erythropoietin on renal fibrosis in rats with unilateral ureteral obstruction. Renal Failure 36: 252–257. https://doi.org/10.3109/0886022X.2013.836936.
Gesta, S., K. Guntur, I.D. Majumdar, et al. 2016. Reduced expression of collagen VI alpha 3 confers resistance to inflammation-induced MCP1 expression in adipocytes. Obesity (Silver Spring) 24: 1695–1703. https://doi.org/10.1002/oby.21565.
Kobayashi, S., H.T. Phung, Y. Kagawa, et al. 2021. Fatty acid-binding protein 3 controls contact hypersensitivity through regulating skin dermal Vγ4+ γ/δ T cell in a murine model. Allergy 76: 1776–1788. https://doi.org/10.1111/all.14630.
Meng, Y., S. Heybrock, D. Neculai, and P. Saftig. 2020. Cholesterol handling in lysosomes and beyond. Trends in Cell Biology 30: 452–466. https://doi.org/10.1016/j.tcb.2020.02.007.
Acknowledgements
We are grateful to Prof. Zhenghua Zhang as the leader of our team. Besides, we are grateful to Prof. Daqiang Li (Fudan University Shanghai Cancer Center and Institutes of Biomedical Sciences, Fudan University) and his lab for providing the experimental platform and guidance. And we thank Shanghai Model Organisms Center, Inc., and Genesky Biotechnologies Inc., (Shanghai, China) for their excellent technical support.
Funding
This work was supported by the National Natural Science Foundation of China (81974471).
Author information
Authors and Affiliations
Contributions
Zhenghua Zhang conceived this work and provided funding; Yumeng La performed experiments and wrote the original manuscript; Wenghong Wong also participated in experiments; Zhen Tian, Kexiang Yan, and Jing Luan provided technical assistance; Kexin Peng and Ruilin Sun performed statistical analysis; Qiaoan Zhang and Jiewen Pan performed pathology analysis. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Ethical clearance was obtained from the Animal Ethics Committee of Huashan Hospital, Fudan University.
Consent for Publication
All authors consent to publish this manuscript.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
La, Y., Wong, W., Peng, K. et al. Decreased Imiquimod-Induced Psoriasis-Like Skin Inflammation in a Novel MvdF250S/+ Knock-In Mouse Model. Inflammation 46, 1575–1586 (2023). https://doi.org/10.1007/s10753-023-01828-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-023-01828-z