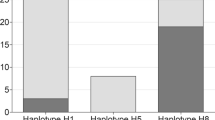
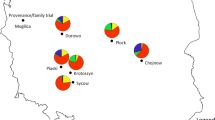
Abstract
Red spruce (Picea rubens Sarg.) is a coniferous tree with a highly fragmented range in eastern North American montane forests. It serves as a foundational species for many locally rare and threatened taxa and has therefore been the focus of large-scale reforestation efforts aimed at restoring these montane ecosystems, yet genetic input guiding these efforts has been lacking. To tackle this issue, we took advantage of a common garden experiment and a whole exome sequencing dataset to investigate the impact of different population genetic parameters on early-life seedling fitness in red spruce. The level of inbreeding, genetic diversity and genetic load were assessed for 340 mother trees sampled from 65 localities across the species range and compared to different fitness traits measured on 5100 of their seedlings grown in a controlled environment. We identified an overall positive influence of genetic diversity and negative impact of genetic load and population-level inbreeding on early-life fitness. Those associations were most apparent for the highly fragmented populations in the Central and Southern Appalachians, where lower genetic diversity and higher inbreeding were associated with lower germination rate, shorter height and reduced early-life fitness of the seedlings. These results provide unprecedented information that could be used by field managers aiming to restore red spruce forests and to maximize the success of future plantations.






Similar content being viewed by others
Data availability
A data table including the different genomic and trait values for each individual and family is available on Github (https://github.com/stephenrkeller/Prubens_Capblancq_etal_2020_ConsGen). together with the script used to perform the different analyses
Code availability
The script used to perform the different analyses is available on Github (https://github.com/stephenrkeller/Prubens_Capblancq_etal_2020_ConsGen).
References
Adams HS, Stephenson SL (1989) Old growth red spruce communities in the mid-Appalachians. Vegetatio 85:45–56
Agrawal AF, Whitlock MC (2012) Mutation load: the fitness of individuals in populations where deleterious alleles are abundant. Annu Rev Ecol Evol Syst 43(1):115–135. https://doi.org/10.1146/annurev-ecolsys-110411-160257
Allendorf FW, Luikart GH (2006) Conservation and the genetics of populations (Blackwell). Blackwell, London
Allendorf FW, Hohenlohe PA, Luikart G (2010) Genomics and the future of conservation genetics. Nat Rev Genet 11(10):697–709. https://doi.org/10.1038/nrg2844
Anderson JT (2015) Plant fitness in a rapidly changing world. New Phytol 210(1):81–87. https://doi.org/10.1111/nph.13693
Angeloni F, Ouborg NJ, Leimu R (2011) Meta-analysis on the association of population size and life history with inbreeding depression in plants. Biol Cons 144(1):35–43. https://doi.org/10.1016/j.biocon.2010.08.016
Baker HG (1972) Seed weight in relation to environmental conditions in California. Ecology 53(6):997–1010
Bashalkhanov S, Eckert AJ, Rajora OP (2013) Genetic signatures of natural selection in response to air pollution in red spruce (Picea rubens, Pinaceae). Mol Ecol 22(23):5877–5889. https://doi.org/10.1111/mec.12546
Birol I et al. (2013) Assembling the 20 Gb white spruce (Picea glauca) genome from whole-genome shotgun sequencing data. Bioinformatics, 29(12), 1492–1497. https://doi.org/10.1093/bioinformatics/btt178
Boisvert-Marsh L, Périé C, De Blois S (2014) Shifting with climate? Evidence for recent changes in tree species distribution at high latitudes. Ecosphere 5(7):1–33. https://doi.org/10.1890/ES14-00111.1
Butnor JR, Verrico BM, Johnsen KH, Maier CA, Vankus V, Keller SR (2019) Phenotypic variation in climate-associated traits of red spruce (Picea rubens Sarg) along elevation gradients in the Southern Appalachian Mountains. Castanea 84(2):128. https://doi.org/10.2179/0008-7475.84.2.128
Capblancq T et al. (2020) Whole-exome sequencing reveals a long-term decline in effective population size of red spruce (Picea rubens). Evolut Appl (2), 1–16. https://doi.org/10.1111/eva.12985
Capblancq T, Fitzpatrick MC, Bay RA, Exposito-Alonso M, Keller SR (2020b) Genomic prediction of (Mal)adaptation across current and future climatic landscapes. Annu Rev Ecol Evol Syst 51(1):245–271. https://doi.org/10.1146/annurev-ecolsys-020720-042553
Charlesworth D, Willis JH (2009) The genetics of inbreeding depression. Nat Rev Genet 10(11):783–796. https://doi.org/10.1038/nrg2664
Cingolani P et al. (2012) A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff. Fly, 6(2), 80–92. https://doi.org/10.4161/fly.19695
Davis MB, Shaw RG (2001) Range shifts and adaptive responses to quaternary climate change. Science 292(5517):673–679. https://doi.org/10.1126/science.292.5517.673
de Lafontaine G, Bousquet J (2017) Asymmetry matters: a genomic assessment of directional biases in gene flow between hybridizing spruces. Ecol Evol 7(11):3883–3893. https://doi.org/10.1002/ece3.2682
De Lafontaine G, Prunier J, Gérardi S, Bousquet J (2015) Tracking the progression of speciation: Variable patterns of introgression across the genome provide insights on the species delimitation between progenitor-derivative spruces (Picea mariana × P. rubens). Mol Ecol 24(20):5229–5247. https://doi.org/10.1111/mec.13377
Eckert CG, Samis KE, Lougheed SC (2008) Genetic variation across species’ geographical ranges: the central-marginal hypothesis and beyond. Mol Ecol 17(5):1170–1188. https://doi.org/10.1111/j.1365-294X.2007.03659.x
Ellegren H, Galtier N (2016) Determinants of genetic diversity. Nat Publish Group 17(7):422–433. https://doi.org/10.1038/nrg.2016.58
Frankham R, Ballou JD, Briscoe DA (2010) Introduction to conservation genetics. Cambridge University Press, Cambridge
González-Martínez SC, Ridout K, Pannell JR (2017) Range expansion compromises adaptive evolution in an outcrossing plant. Curr Biol 27(16):2544–2551.e4. https://doi.org/10.1016/J.CUB.2017.07.007
Hampe A, Petit RJ (2005) Conserving biodiversity under climate change: the rear edge matters. Ecol Lett 8(5):461–467. https://doi.org/10.1111/j.1461-0248.2005.00739.x
Hampe A, Pemonge MH, Petit RJ (2013) Efficient mitigation of founder effects during the establishment of a leadingedge oak population. Proc R Soc B. https://doi.org/10.1098/rspb.2013.1070
Hendricks S et al. (2018) Recent advances in conservation and population genomics data analysis. Evol Appl 11(8), 1197–1211. https://doi.org/10.1111/eva.12659
Hewitt G (2000) The genetic legacy of the Quaternary ice ages. Nature 405(6789):907–913. https://doi.org/10.1038/35016000
Kardos M, Taylor HR, Ellegren H, Luikart G, Allendorf FW (2016) Genomics advances the study of inbreeding depression in the wild. Evol Appl 9(10):1205–1218. https://doi.org/10.1111/eva.12414
Keller LF, Waller DM (2002) Inbreeding effects in wild populations. Trends Ecol Evol 17(5):230–241. https://doi.org/10.1016/S0169-5347(02)02489-8
Khan S, Nabi G, Ullah MW, Yousaf M, Manan S, Siddique R, Hou H (2016) Overview on the role of advance genomics in conservation biology of endangered species. Int J Genomics. https://doi.org/10.1155/2016/3460416
Kimura M (1955) Solution of a process of random genetic drift with a continuous model. Proc Natl Acad Sci 41(2):144–150
Kimura M (1957) Some problems of stochastic processes in genetics. Ann Math Stat 28(4):882–901. https://doi.org/10.1214/aoms/1177706791
Koo KA, Madden M, Patten BC (2014a) Projection of red spruce (Picea rubens Sargent) habitat suitability and distribution in the Southern Appalachian Mountains, USA. Ecol Model 293:91–101. https://doi.org/10.1016/j.ecolmodel.2014.06.005
Koo KA, Patten BC, Teskey RO, Creed IF (2014b) Climate change effects on red spruce decline mitigated by reduction in air pollution within its shrinking habitat range. Ecol Model 293:81–90. https://doi.org/10.1016/j.ecolmodel.2014.07.017
Korneliussen TS, Albrechtsen A, Nielsen R (2014) ANGSD: analysis of next generation sequencing data. BMC Bioinform 15:356–368. https://doi.org/10.1007/978-1-4614-1174-1_8
Lande R (1995) Mutation and conservation. Conservation Biology. Conservation Biology 9(4):782–791
Latta RG (2008) Conservation genetics as applied evolution: from genetic pattern to evolutionary process. Evol Appl 1(1):84–94. https://doi.org/10.1111/j.1752-4571.2007.00008.x
Le HD, Smith C, Herbohn J, Harrison S (2012) More than just trees: assessing reforestation success in tropical developing countries. J Rural Stud. https://doi.org/10.1016/j.jrurstud.2011.07.006
Leimu R, Vergeer P, Angeloni F, Ouborg NJ (2010) Habitat fragmentation, climate change, and inbreeding in plants. Ann N Y Acad Sci 1195:84–98. https://doi.org/10.1111/j.1749-6632.2010.05450.x
Levin DA (2011) Mating system shifts on the trailing edge. Ann Bot 109:613–620
Li H (2011) Improving SNP discovery by base alignment quality. Bioinformatics 27(8):1157–1158. https://doi.org/10.1093/bioinformatics/btr076
Liu Y et al. (2013) Environmental factors influence both abundance and genetic diversity in a widespread bird species. Ecol Evol, 3(14), 4683–4695. https://doi.org/10.1002/ece3.856
Luikart G, Allendorf FW, Cornuet JM, Sherwin WB (1998) Distortion of allele frequency distributions provides a test for recent population bottlenecks. J Hered 89(3):238–247. https://doi.org/10.1093/jhered/89.3.238
Lynch M, Conery IJ, Burger R (1995) Mutation accumulation and the extinction of small populations. Am Nat 146(4):489–518
Major JE, Mosseler A, Johnsen KH, Campbell M, Malcolm J (2015) Growth and allocation of Picea rubens, Picea mariana, and their hybrids under ambient and elevated CO 2. Can J For Res 45(7):877–887. https://doi.org/10.1139/cjfr-2014-0525
Markert JA et al. (2010) Population genetic diversity and fitness in multiple environments. BMC Evol Biol, 10(1), 5–9. https://doi.org/10.1186/1471-2148-10-205
Marks CO (2007) The causes of variation in tree seedling traits: the roles of environmental selection versus chance. Evolution 61(2):455–469. https://doi.org/10.1111/j.1742-4658.2007.00021.x
Mathias JM, Thomas RB (2018) Disentangling the effects of acidic air pollution, atmospheric CO2, and climate change on recent growth of red spruce trees in the Central Appalachian Mountains. Glob Change Biol 24(9):3938–3953. https://doi.org/10.1111/gcb.14273
Miller ML, Kronenberger JA, Fitzpatrick SW (2019) Recent evolutionary history predicts population but not ecosystem-level patterns. Ecol Evolut 9(24):14442–14452. https://doi.org/10.1002/ece3.5879
Mosseler A et al. (2000) Indicators of population viability in red spruce, Picea rubens. I. Reproductive traits and fecundity. Can J Bot 78(7), 928–940. https://doi.org/10.1139/b00-065
Mosseler A, Major JE, Rajora OP (2003) Old-growth red spruce forests as reservoirs of genetic diversity and reproductive fitness. Theoret Appl Genet 106(5):931–937. https://doi.org/10.1007/s00122-002-1156-1
Naish KA, Seamons TR, Dauer MB, Hauser L, Quinn TP (2013) Relationship between effective population size, inbreeding and adult fitness-related traits in a steelhead (Oncorhynchus mykiss) population released in the wild. Mol Ecol 22(5):1295–1309. https://doi.org/10.1111/mec.12185
Nielsen R, Paul JS, Albrechtsen A, Song YS (2011) Genotype and SNP calling from next-generation sequencing data. Nat Rev Genet 12(6):443–451. https://doi.org/10.1038/nrg2986
Nowacki G, Carr R, Van Dyck M (2010) The current status of Red Spruce in the Eastern United States : distribution, population trends, and environmental drivers. Proceedings from the Conference on the Ecology and Management of High-Elevation Forests in the Central and Southern Appalachian Mountains, pp 140–162
Nystedt B et al. (2013) The Norway spruce genome sequence and conifer genome evolution. Nature, 497(7451), 579–584. https://doi.org/10.1038/nature12211
Peischl S, Dupanloup I, Kirkpatrick M, Excoffier L (2013) On the accumulation of deleterious mutations during range expansions. Mol Ecol 22(24):5972–5982. https://doi.org/10.1111/mec.12524
Peischl S, Kirkpatrick M, Excoffier L (2015) Expansion load and the evolutionary dynamics of a species range. Am Nat 185(4):E81–E93. https://doi.org/10.1086/680220
Pluess AR (2011) Pursuing glacier retreat: genetic structure of a rapidly expanding Larix decidua population. Mol Ecol 20(3):473–485. https://doi.org/10.1111/j.1365-294X.2010.04972.x
Ratnam W et al. (2014) Genetic effects of forest management practices: global synthesis and perspectives. For Ecol Manage 333, 52–65. https://doi.org/10.1016/j.foreco.2014.06.008
Reed DH, Frankham R (2003) Correlation between fitness and genetic diversity. Conserv Biol 17(1):230–237. https://doi.org/10.1046/j.1523-1739.2003.01236.x
Rentch JS, Schuler TS (2009) Pennsylvania boreal conifer forests and their bird communities: past, present, and potential. Proceedings from the Conference on the Ecology and Management of High-Elevation Forests in the Central and Southern Appalachian Mountains, 48–73
Rentch JS, Ford WM, Schuler TS, Palmer J (2016) Release of suppressed red spruce using canopy gap creation—ecological restoration in the central appalachians. Nat Areas J 36(1):29. https://doi.org/10.3375/043.036.0108
Ribeiro PJ, Diggle PJ (2016) geoR: A package for geostatistical data analysis using the R software. R News, 1(6), 15–18. Retrieved from http://www.leg.ufpr.br/geoR/
Roach DA, Wulff RD (1987) Maternal effects in plants. Ann Rev Ecol System. 18(1987):209–235. https://doi.org/10.1146/annurev.es.18.110187.001233
Sagarin RD, Gaines SD (2002) The “abundant centre” distribution: to what extent is it a biogeographical rule? Ecol Lett 5(1):137–147. https://doi.org/10.1046/j.1461-0248.2002.00297.x
Schauffler M, Jacobson GL (2002) Persistence of coastal spruce refugia during the Holocene in northern New England, USA, detected by stand-scale pollen stratigraphies. J Ecol 90(2):235–250. https://doi.org/10.1046/j.1365-2745.2001.00656.x
Singh J, Michelangeli JAC, Gezan SA, Lee H, Vallejos CE (2017) Maternal effects on seed and seedling phenotypes in reciprocal F1hybrids of the common bean (Phaseolus vulgaris L.). Front Plant Sci 8(1):1–13. https://doi.org/10.3389/fpls.2017.00042
Supple MA, Shapiro B (2018) Conservation of biodiversity in the genomics era. Genome Biol 19(1):1–12. https://doi.org/10.1186/s13059-018-1520-3
Wahid N, Bounoua L (2013) The relationship between seed weight, germination and biochemical reserves of maritime pine (Pinus pinaster Ait.) in Morocco. New Forests 44(3):385–397. https://doi.org/10.1007/s11056-012-9348-2
Wang J, Caballero A, Hill WG (1998) The effect of linkage disequilibrium and deviation from Hardy-Weinberg proportions on the changes in genetic variance with bottlenecking. Heredity 81(2):174–186. https://doi.org/10.1038/sj.hdy.6883900
Willi Y, Fracassetti M, Zoller S, Van Buskirk J (2018) Accumulation of mutational load at the edges of a species range. Mol Biol Evol 35(4):781–791. https://doi.org/10.1093/molbev/msy003
Wolf JB, Wade MJ (2009) What are maternal effects (and what are they not)? Philos Trans R Soc B 364(1520):1107–1115. https://doi.org/10.1098/rstb.2008.0238
Wright S (1931) Evolution in mendelian populations. Genetics 16:97–159. https://doi.org/10.4161/hv.21408
Zhang R, Baskin JM, Baskin CC, Mo Q, Chen L, Hu X, Wang Y (2017) Effect of population, collection year, after-ripening and incubation condition on seed germination of Stipa bungeana. Sci Rep 7(1):1–11. https://doi.org/10.1038/s41598-017-14267-2
Acknowledgments
We appreciate the help of partners who shared access to their seed collections: Barbara Crane, Matt Fitzpatrick, Robert Jetton, John Major, John Malcom, Dave Nelson, and Brittany Verrico. We also thank the members of the Keller lab and three anonymous reviewers for helpful comments on the manuscript. This project was supported by awards from the National Science Foundation (1656099) and USDA-HATCH (1006810) to SK, and a UVM SURF award to HM.
Funding
This project was supported by awards from the National Science Foundation (1656099) and USDA-HATCH (1006810) to Stephen Keller, and a UVM SURF award to Helena Munson.
Author information
Authors and Affiliations
Contributions
Stephen Keller conceived the study. SK and John Butnor collected the samples. Helena Munson germinated and grew the seedlings with help from SK and JB. HM measured fitness traits and Thibaut Capblancq analyzed genomic data. TC performed the statistical analyses with the help of HM. TC and HM wrote a first draft of the manuscript and all authors provided critical feedbacks.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Inforamtion
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Capblancq, T., Munson, H., Butnor, J.R. et al. Genomic drivers of early-life fitness in Picea rubens. Conserv Genet 22, 963–976 (2021). https://doi.org/10.1007/s10592-021-01378-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-021-01378-7