Abstract
The oxygen evolution reaction (OER) plays a vital role in electrolytic water splitting. It is urgent to explore highly active non-precious metal OER catalysts. Herein, the etching combined with the electrodeposition was used to synthesize the efficient Ni–Fe/p-CoAl catalyst, which exhibits ideal OER performance. A low overpotential of 282 mV and 296 mV are needed to achieve the current density of 50 mA/cm2 and 100 mA/cm2 in 1 M KOH, respectively. Moreover, the as-prepared catalyst shows good durability with no detectable degradation during long-term operation. The enhanced OER performance can be attributed to the large electrochemical surface area and the strong interaction between Ni–Fe nanosheets and active substrate.
Graphical Abstract
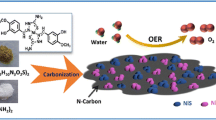
Etching combined with the electrodeposition method was used to synthesize an efficient Ni–Fe/p-CoAl catalyst for OER.








Similar content being viewed by others
References
Cen J, Wu Q, Liu M, Orlov A (2017) Developing new understanding of photoelectrochemical water splitting via in-situ techniques: A review on recent progress. Green Energy Environ 2:100–111
Luo Y, Zhang Z, Chhowalla M, Liu B (2022) Recent Advances in Design of Electrocatalysts for High-Current-Density Water Splitting. Adv Mater 34:2108133
Huang Z, Wang J, Peng Y, Jung C, Fisher A, Wang X (2017) Design of Efficient Bifunctional Oxygen Reduction/Evolution Electrocatalyst: Recent Advances and Perspectives. Adv Energy Mater 7:1700544
Sun H, Xu X, Kim H, Jung W, Zhou W, Shao Z (2023) Electrochemical Water Splitting: Bridging the Gaps Between Fundamental Research and Industrial Applications. Energy Environ Mater 6.
Zhao J, Zhang J, Li Z, Bu X (2020) Recent Progress on NiFe-Based Electrocatalysts for the Oxygen Evolution Reaction. Small 16:2003916
Liao H, Luo T, Tan P, Chen K, Lu L, Liu Y, Liu M, Pan J (2021) Unveiling Role of Sulfate Ion in Nickel-Iron (oxy)Hydroxide with Enhanced Oxygen-Evolving Performance. Adv Func Mater 31:2102772
Du Y, Liu D, Li T, Yan Y, Liang Y, Yan S, Zou Z (2022) A phase transformation-free redox couple mediated electrocatalytic oxygen evolution reaction. Applied Catalysis B-Environmental 306:121146
Liao Y, He R, Pan W, Li Y, Wang Y, Li J, Li Y (2023) Lattice distortion induced Ce-doped NiFe-LDH for efficient oxygen evolution. Chem Eng J 464:142669
Li X, Liu Y, Sun Q, Huangfu Z, Huang W-H, Wang Z, Chueh C-C, Chen C-L, Zhu Z (2022) Effects of Cationic and Anionic Defects on NiFe LDH in Electrocatalytic Oxygen Evolution. Acs Sustain Chem Eng 10:14474–14485
Zhang K, Wan T, Wang H, Luo Y, Shi Y, Zhang Z, Liu G, Li J (2023) Decorated Oxidation-resistive deficient Titanium oxide nanotube supported NiFe-nanosheets as high-efficiency electrocatalysts for overall water splitting. J Colloid Interface Sci 645:66–75
Jiang K, Luo M, Peng M, Yu Y, Lu YR, Chan TS, Liu P, de Groot FM, Tan Y (2020) Dynamic active-site generation of atomic iridium stabilized on nanoporous metal phosphides for water oxidation. Nat Commun 11:2701
Shi R, Wang J, Wang Z, Li T, Song Y-F (2019) Unique NiFe-NiCoO2 hollow polyhedron as bifunctional electrocatalysts for water splitting. J Energy Chem 33:74–80
Chen G, Zhu Y, Chen HM, Hu Z, Hung S, Ma N, Dai J, Lin H, Chen C, Zhou W, Shao Z (2019) An Amorphous Nickel-Iron-Based Electrocatalyst with Unusual Local Structures for Ultrafast Oxygen Evolution Reaction. Adv Mater 31:1900883
Zhao Y, Zhang X, Jia X, Waterhouse GIN, Shi R, Zhang X, Zhan F, Tao Y, Wu L, Tung C, O’Hare D, Zhang T (2018) Sub-3 nm Ultrafine Monolayer Layered Double Hydroxide Nanosheets for Electrochemical Water Oxidation. Adv Energy Mater 8:1703585
Liu J, Zhu D, Zheng Y, Vasileff A, Qiao S (2018) Self-Supported Earth-Abundant Nanoarrays as Efficient and Robust Electrocatalysts for Energy-Related Reactions. ACS Catal 8:6707–6732
Yao RQ, Shi H, Wan WB, Wen Z, Lang XY, Jiang Q (2020) Flexible Co–Mo–N/Au electrodes with a hierarchical nanoporous architecture as highly efficient electrocatalysts for oxygen evolution reaction. Adv Mater 32:1907214
Shi H, Zhou YT, Yao RQ, Wan WB, Ge X, Zhang W, Wen Z, Lang XY, Zheng WT, Jiang Q (2020) Spontaneously separated intermetallic Co3Mo from nanoporous copper as versatile electrocatalysts for highly efficient water splitting. Nat Commun 11:2940
Cai L, Bai H, Kao CW, Jiang K, Pan H, Lu YR, Tan Y (2024) Platinum–Ruthenium Dual‐Atomic Sites Dispersed in Nanoporous Ni0. 85Se Enabling Ampere‐Level Current Density Hydrogen Production. Small. 15:2311178.
Yao RQ, Zhou YT, Shi H, Wan WB, Zhang QH, Gu L, Zhu YF, Wen Z, Lang XY, Jiang Q (2021) Nanoporous surface high-entropy alloys as highly efficient multisite electrocatalysts for nonacidic hydrogen evolution reaction. Adv Func Mater 31:2009613
An Y, Tian Y, **ong S, Feng J, Qian Y (2021) Scalable and controllable synthesis of interface-engineered nanoporous host for dendrite-free and high rate zinc metal batteries. Acs Nano. 15:11828-11842
Zhao Y, Luo M, Chu S, Peng M, Liu B, Wu Q, Liu P, de Groot FMF, Tan Y (2019) 3D nanoporous iridium-based alloy microwires for efficient oxygen evolution in acidic media. Nano Energy 59:146–153
Qiao Y, Peng M, Lan J, Jiang K, Chen D, Tan Y (2023) Active-site engineering in dealloyed nanoporous catalysts for electrocatalytic water splitting. J Mater Chem A 11:495–511
Fu J, Welborn SS, Detsi E (2022) Dealloyed Air- and Water-Sensitive Nanoporous Metals and Metalloids for Emerging Energy Applications. Acs Appl Energy Mater 5:6516–6544
An Y, Tian Y, Wei C, Tao Y, Xi B, Xiong S, Feng J, Qian Y (2021) Dealloying: An effective method for scalable fabrication of 0D, 1D, 2D, 3D materials and its application in energy storage. Nano Today 37:101094
Guo X, Zhang C, Tian Q, Yu D (2021) Liquid metals dealloying as a general approach for the selective extraction of metals and the fabrication of nanoporous metals: A review. Mater Today Commun 26:102007
Lee S, Kim J, Joo H, Park C, Jeong S, Jung K, Kim Y, Kang K (2022) Effect of Plating Variables on Oxygen Evolution Reaction of Ni-Zn-Fe Electrodes for Alkaline Water Electrolysis. Catalysts 12:346
Loh A, Li X, Taiwo OO, Tariq F, Brandon NP, Wang P, Xu K, Wang B (2020) Development of Ni-Fe based ternary metal hydroxides as highly efficient oxygen evolution catalysts in AEM water electrolysis for hydrogen production. Int J Hydrogen Energy 45:24232–24247
Dong C, Han L, Zhang C, Zhang Z (2018) Scalable Dealloying Route to Mesoporous Ternary CoNiFe Layered Double Hydroxides for Efficient Oxygen Evolution. Acs Sustain Chem Eng 6:16096–16104
Hou X, Li J, Zheng J, Li L, Chu W (2022) Introducing oxygen vacancies to NiFe LDH through electrochemical reduction to promote the oxygen evolution reaction. Dalton Trans 51:13970–13977
Bao J, Zhang X, Fan B, Zhang J, Zhou M, Yang W, Hu X, Wang H, Pan B, Xie Y (2015) Ultrathin Spinel-Structured Nanosheets Rich in Oxygen Deficiencies for Enhanced Electrocatalytic Water Oxidation. Angewandte Chemie-International Edition 54:7399–7404
Zhao H, Zhang Y, Xie C, Wang J, Zhou T, Zhou C, Li J, Bai J, Zhu X, Zhou B (2023) Facile, Controllable, and Ultrathin NiFe-LDH In Situ Grown on a Ni Foam by Ultrasonic Self-Etching for Highly Efficient Urine Conversion. Environ Sci Technol 57:2939–2948
Zhang X, Wu A, Wang D, Jiao Y, Yan H, Jin C, Xie Y, Tian C (2023) Fine-tune the electronic structure in Co-Mo based catalysts to give easily coupled HER and OER catalysts for effective water splitting. Appl Catal B-Environ 328:122474
Liu Y, Luo X, Zhou C, Du S, Zhen D, Chen B, Li J, Wu Q, Iru Y, Chen D (2020) A modulated electronic state strategy designed to integrate active HER and OER components as hybrid heterostructures for efficient overall water splitting. Appl Catal B-Environ 260:118197
Zhu S, Wang J, Li H, Cai J, Li Y, Hu J, He Y, Zhou Y (2022) NiFe Layered Double Hydroxide Nanosheets Anchored on Cobalt Nanocrystal Matrixes as Electrocatalysts for Oxygen Evolution. Acs Appl Nano Mater 5:13047–13054
Lee J, Jung H, Park YS, Kwon N, Woo S, Selvam NCS, Han GS, Jung HS, Yoo PJ, Choi SM, Han JW, Lim B (2021) Chemical transformation approach for high-performance ternary NiFeCo metal compound-based water splitting electrodes. Appl Catal B-Environ 294:120246
Zhao Q, Yang J, Liu M, Wang R, Zhang G, Wang H, Tang H, Liu C, Mei Z, Chen H, Pan F (2018) Tuning Electronic Push/Pull of Ni-Based Hydroxides To Enhance Hydrogen and Oxygen Evolution Reactions for Water Splitting. ACS Catal 8:5621–5629
Han L, Guo L, Dong C, Zhang C, Gao H, Niu J, Peng Z, Zhang Z (2019) Ternary mesoporous cobalt-iron-nickel oxide efficiently catalyzing oxygen/hydrogen evolution reactions and overall water splitting. Nano Res 12:2281–2287
Son H, Park J, Uthirakumar P, Kuznetsov A, Lee I (2020) Impact of chloride surface treatment on nano-porous GaN structure for enhanced water-splitting efficiency. Appl Surf Sci 532:147465
Zong R, Fang Y, Zhu C, Zhang X, Wu L, Hou X, Tao Y, Shao J (2021) Surface Defect Engineering on Perovskite Oxides as Efficient Bifunctional Electrocatalysts for Water Splitting. ACS Appl Mater Interfaces 13:42852–42860
Acknowledgements
This work is supported by the Local Science and Technology Development Fund Projects Guided by the Central Government of China (2021ZYD0060), the Science and Technology Project of Southwest Petroleum University (2021JBGS03).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, L., Yan, S., Wang, H. et al. Ni–Fe Nanosheets Anchored on Porous Cobalt Substrate towards Oxygen Evolution Reaction. Catal Lett (2024). https://doi.org/10.1007/s10562-024-04657-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10562-024-04657-3