Abstract
Purpose
Identify the presence of a dysfunctional electroencephalographic (EEG) pattern in individuals with sickle cell disease (SCD) and hip osteonecrosis, and assess its potential associations with depression, anxiety, pain severity, and serum levels of brain-derived neurotrophic factor (BDNF).
Methods
In this cross-sectional investigation, 24 SCD patients with hip osteonecrosis and chronic pain were matched by age and sex with 19 healthy controls. Resting-state EEG data were recorded using 32 electrodes for both groups. Power spectral density (PSD) and peak alpha frequency (PAF) were computed for each electrode across Delta, Theta, Alpha, and Beta frequency bands. Current Source Density (CSD) measures were performed utilizing the built-in Statistical nonparametric Mapping Method of the LORETA-KEY software.
Results
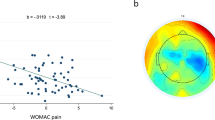
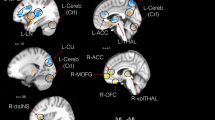
Our findings demonstrated that SCD individuals exhibited higher PSD in delta and theta frequency bands when compared to healthy controls. Moreover, SCD individuals displayed increased CSD in delta and theta frequencies, coupled with decreased CSD in the alpha frequency within brain regions linked to pain processing, motor function, emotion, and attention. In comparison to the control group, depression symptoms, and pain intensity during hip abduction were positively correlated with PSD and CSD in the delta frequency within the parietal region. Depression symptoms also exhibited a positive association with PSD and CSD in the theta frequency within the same region, while serum BDNF levels showed a negative correlation with CSD in the alpha frequency within the left insula.
Conclusion
This study indicates that individuals with SCD experiencing hip osteonecrosis and chronic pain manifest a dysfunctional EEG pattern characterized by the persistence of low-frequency PSD during a resting state. This dysfunctional EEG pattern may be linked to clinical and biochemical outcomes, including depression symptoms, pain severity during movement, and serum BDNF levels.




Similar content being viewed by others
References
Al-Otaibi ML (2022) An overview of pathophysiology and treatment options of osteonecrosis of femoral head in sickle cell Disease. Saudi Med J 43:1192–1199
Baliki MN, Mansour AR, Baria AT, Apkarian AV (2014) Functional reorganization of the default mode network across chronic pain conditions. PLoS ONE 9:e106133
Ballas SK, Darbari DS (2020) Review/overview of pain in sickle cell Disease. Complement Ther Med 49:102327
Boord P, Siddall PJ, Tran Y et al (2008) Electroencephalographic slowing and reduced reactivity in neuropathic pain following spinal cord injury. Spinal Cord 46:118–123
Buchanan DM, Ros T, Nahas R (2021) Elevated and slowed EEG oscillations in patients with post-concussive syndrome and Chronic Pain following a motor vehicle collision. Brain Sci 11. https://doi.org/10.3390/brainsci11050537
Campbell CM, Moscou-Jackson G, Carroll CP et al (2016) An evaluation of Central Sensitization in patients with Sickle Cell Disease. J Pain 17:617–627
Case M, Zhang H, Mundahl J et al (2017) Characterization of functional brain activity and connectivity using EEG and fMRI in patients with sickle cell Disease. Neuroimage Clin 14:1–17
Case M, Shirinpour S, Zhang H et al (2018) Increased theta band EEG power in sickle cell Disease patients. J Pain Res 11:67–76
Caumo W, Antunes LC, Elkfury JL et al (2017) The Central Sensitization Inventory validated and adapted for a Brazilian population: psychometric properties and its relationship with brain-derived neurotrophic factor. J Pain Res 10:2109–2122
Cavanna AE, Trimble MR (2006) The precuneus: a review of its functional anatomy and behavioural correlates. Brain 129:564–583
Da Silva JT, Letzen JE, Haythornthwaite JA et al (2019) Do chronic pain and comorbidities affect brain function in sickle cell patients? A systematic review of neuroimaging and treatment approaches. Pain 160:1933–1945
Dampier C, Palermo TM, Darbari DS et al (2017) AAPT Diagnostic Criteria for Chronic Sickle Cell Disease Pain. J Pain 18:490–498
Darbari DS, Hampson JP, Ichesco E et al (2015) Frequency of hospitalizations for Pain and Association with altered Brain Network Connectivity in Sickle Cell Disease. J Pain 16:1077–1086
Delorme A, Makeig S (2004) EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including Independent component analysis. J Neurosci Methods 134:9–21
Di Pietro F, Macey PM, Rae CD et al (2018) The relationship between thalamic GABA content and resting cortical rhythm in neuropathic pain. Hum Brain Mapp 39:1945–1956
Fallon N, Chiu Y, Nurmikko T, Stancak A (2016) Functional connectivity with the default Mode Network is altered in Fibromyalgia patients. PLoS ONE 11:e0159198
Ficat P, Arlet J, Vidal R et al (1971) [Therapeutic results of drill biopsy in primary osteonecrosis of the femoral head (100 cases)]. Rev Rhum Mal Osteoartic 38:269–276
Fingelkurts AA, Fingelkurts AA (2015) Altered structure of dynamic electroencephalogram oscillatory pattern in major depression. Biol Psychiatry 77:1050–1060
Gallace A, Bellan V (2018) The parietal cortex and pain perception: a body protection system. Handb Clin Neurol 151:103–117
González-Roldán AM, Cifre I, Sitges C, Montoya P (2016) Altered dynamic of EEG oscillations in Fibromyalgia patients at Rest. Pain Med 17:1058–1068
Gram M, Erlenwein J, Petzke F et al (2017) The cortical responses to evoked clinical pain in patients with hip osteoarthritis. PLoS ONE 12:e0186400
Haukoos JS, Lewis RJ (2005) Advanced statistics: bootstrapping confidence intervals for statistics with difficult distributions. Acad Emerg Med 12:360–365
Inui K, Wang X, Qiu Y et al (2003) Pain processing within the primary somatosensory cortex in humans. Eur J Neurosci 18:2859–2866
Jensen MP, Sherlin LH, Fregni F et al (2014) Baseline brain activity predicts response to neuromodulatory pain treatment. Pain Med 15:2055–2063
Kanda M, Nagamine T, Ikeda A et al (2000) Primary somatosensory cortex is actively involved in pain processing in human. Brain Res 853:282–289
Karafin MS, Chen G, Wandersee NJ et al (2019) Chronic pain in adults with sickle cell Disease is associated with alterations in functional connectivity of the brain. PLoS ONE 14:e0216994
Kisler LB, Kim JA, Hemington KS et al (2020) Abnormal alpha band power in the dynamic pain connectome is a marker of chronic pain with a neuropathic component. Neuroimage Clin 26:102241
Llinás RR, Ribary U, Jeanmonod D et al (1999) Thalamocortical dysrhythmia: a neurological and neuropsychiatric syndrome characterized by magnetoencephalography. Proc Natl Acad Sci U S A 96:15222–15227
Llinás R, Urbano FJ, Leznik E et al (2005) Rhythmic and dysrhythmic thalamocortical dynamics: GABA systems and the edge effect. Trends Neurosci 28:325–333
Lopes TdaS, Silva WDS, Ribeiro SB et al (2017) Does Transcranial Direct Current Stimulation Combined with Peripheral Electrical Stimulation have an Additive Effect in the Control of Hip Joint Osteonecrosis Pain Associated with Sickle Cell Disease? A protocol for a one-Session double blind, Block-Randomized Clinical Trial. Front Hum Neurosci 11:633
Lopes Tda, Ballas S, Santana SK JERS, et al (2022) Sickle cell Disease chronic joint pain: clinical assessment based on maladaptive central nervous system plasticity. Front Med 9:679053
McLain NJ, Yani MS, Kutch JJ (2022) Analytic consistency and neural correlates of peak alpha frequency in the study of pain. J Neurosci Methods 368:109460
Meneses FM, Queirós FC, Montoya P et al (2016) Patients with Rheumatoid Arthritis and Chronic Pain Display enhanced Alpha Power Density at Rest. Front Hum Neurosci 10:395
Mussigmann T, Bardel B, Lefaucheur J-P (2022) Resting-state electroencephalography (EEG) biomarkers of chronic neuropathic pain. A systematic review. NeuroImage 258:119351
Orenius TI, Raij TT, Nuortimo A et al (2017) The interaction of emotion and pain in the insula and secondary somatosensory cortex. Neuroscience 349:185–194
Pinheiro ES, dos Queirós S, Montoya FC P, et al (2016) Electroencephalographic patterns in Chronic Pain: a systematic review of the literature. PLoS ONE 11:e0149085
Roy N, Barry RJ, Fernandez FE et al (2020) Electrophysiological correlates of the brain-derived neurotrophic factor (BDNF) Val66Met polymorphism. Sci Rep 10:17915
Santos JG, Brito JO, de Andrade DC et al (2010) Translation to Portuguese and validation of the Douleur Neuropathique 4 questionnaire. J Pain 11:484–490
Sarnthein J, Stern J, Aufenberg C et al (2006) Increased EEG power and slowed dominant frequency in patients with neurogenic pain. Brain 129:55–64
Schmidt S, Naranjo JR, Brenneisen C et al (2012) Pain ratings, psychological functioning and quantitative EEG in a controlled study of chronic back pain patients. PLoS ONE 7:e31138
Simão AP, Mendonça VA, de Oliveira Almeida TM et al (2014) Involvement of BDNF in knee osteoarthritis: the relationship with inflammation and clinical parameters. Rheumatol Int 34:1153–1157
Simis M, Imamura M, Pacheco-Barrios K et al (2022) EEG theta and beta bands as brain oscillations for different knee osteoarthritis phenotypes according to Disease severity. Sci Rep 12:1480
Sitges C, Bornas X, Llabrés J et al (2010) Linear and nonlinear analyses of EEG dynamics during non-painful somatosensory processing in chronic pain patients. Int J Psychophysiol 77:176–183
Stern J, Jeanmonod D, Sarnthein J (2006) Persistent EEG overactivation in the cortical pain matrix of neurogenic pain patients. NeuroImage 31:721–731
Vlcek P, Bares M, Novak T, Brunovsky M (2020) Baseline difference in quantitative electroencephalography variables between responders and non-responders to low-frequency repetitive transcranial magnetic stimulation in Depression. Front Psychiatry 11:83
Vuckovic A, Hasan MA, Fraser M et al (2014) Dynamic oscillatory signatures of central neuropathic pain in spinal cord injury. J Pain 15:645–655
Walton KD, Dubois M, Llinás RR (2010) Abnormal thalamocortical activity in patients with Complex Regional Pain Syndrome (CRPS) type I. Pain 150:41–51
Yip DW, Lui F (2021) Physiology, motor cortical. In: StatPearls. StatPearls Publishing, Treasure Island (FL)
Zebhauser PT, Hohn VD, Ploner M (2023) Resting-state electroencephalography and magnetoencephalography as biomarkers of chronic pain: a systematic review. Pain 164:1200–1221
Zoon HFA, Veth CPM, Arns M et al (2013) EEG alpha power as an intermediate measure between brain-derived neurotrophic factor Val66Met and depression severity in patients with major depressive disorder. J Clin Neurophysiol 30:261–267
Acknowledgements
The authors also wish to thank the Research Support Foundation of the State of Bahia (FAPESB) for funding through the Support Program for Emerging Center, 8133/2014 (PNE0020/2014). TSL received a post-doctoral fellowship from the Coordination for the Improvement of Higher Education Personnel (CAPES) (Process: 88887.801519/2023-00). AFB and FJF receive a scholarship for scientific production from the Brazilian National Council for Scientific Development (CNPq). AFB is supported by CEPID/BRAINN-The Brazilian Institute of Neuroscience and Neurotechnology (Process: 13/07559 − 369 3) and by the LIM-54 HC FMUSP. JERSS received a Ph.D. scholarship from the CAPES. PM was supported by grant PSI2017-88388-C4-1-R funded by MCIN/AEI/ https://doi.org/10.13039/501100011033).
Author information
Authors and Affiliations
Contributions
The authors T.S, A.B, W.S, J.S, P.M, R.L and K.S were involved in study conception and method development. The T.S, W.S, and J.S contributed to all data collection, while F.F and Y.Z developed the data analysis. L.S and T.S prepared all figures and the authors T.S and A.B wrote the main manuscript text. All authors critically revised the final version of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Communicated by Franca Tecchio.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lopes, T.S., Santana, J.E., Silva, W.S. et al. Increased Delta and Theta Power Density in Sickle Cell Disease Individuals with Chronic Pain Secondary to Hip Osteonecrosis: A Resting-State Eeg Study. Brain Topogr (2023). https://doi.org/10.1007/s10548-023-01027-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10548-023-01027-x