Abstract
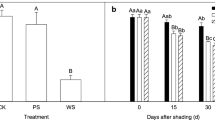
In shaded wheat (Triticum aestivum L.) leaves, the suppression of blue radiation (BR) triggers senescence. This phenomenon is correlated to an increase in oxidative stress symptoms and a decrease of catalase (CAT) activity, among other traits. Previous data suggest that the radiation signal transduction pathway may involve changes in Ca2+ and H2O2 homeostasis. For better a understanding of the interaction among the spectral composition of radiation, Ca2+ availability, and the antioxidant metabolism in the regulation of shade-induced senescence, detached wheat leaves were placed in a growth chamber and exposed to either blue (B, high BR transmittance) and/or green (G, very low BR transmittance) Lee® filters in the absence or presence of 0.8 mM verapamil (a Ca2+ channels blocker), 4.0 mM EGTA (a Ca2+chelator), or 8.0 mM 3-amino-1,2,4-triazole (a CAT inhibitor). At defined time points, the leaf samples were analyzed for changes in chlorophyll content, specific activities of CAT, ascorbate peroxidase (APX), and guaiacol peroxidase (POX), CAT isozymes, and gene expression of CAT1, CAT2, and two senescence markers (TaSAG1 and TaSAG3). BR transmittance decreased the chlorophyll degradation rate and SAG genes expression either in leaves continuously exposed under the B filter, as well as in leaves previously exposed under the G filter. The effect of BR was associated with the maintenance of a high CAT (but not APX and POX) activity, and it was suppressed either in the presence of 3-AT or when Ca2+ availability was decreased. BR altered the CAT activity both at the transcriptional and at the posttranscriptional level. Nevertheless, different responses of CAT isozymes and CAT genes expression profiles to specific treatment combinations indicate that they differed in their regulatory pathways.
Similar content being viewed by others
Abbreviations
- APX:
-
ascorbate peroxidase
- 3-AT:
-
3-amino-1,2,4-triazole
- BR:
-
blue radiation
- CAT:
-
catalase
- EGTA:
-
ethyleneglycol-bis-(β-amino-ethylether)-N,N,N′,N′-tetraacetic acid
- PFD:
-
photon flux density
- POX:
-
guaiacol peroxidase
- PVPP:
-
polyvinylpolypyrrolidone
References
Aebi, H.: Catalase in vitro. — Methods Enzymol. 105: 121–126, 1984.
Acevedo, A., Skadsen, R.W., Scandalios, J.G.: Two barley catalase genes respond differentially to light. — Physiol. Plant. 96: 369–374, 1996.
Azpillicueta, C.E., Pena, L.B., Tomaro, M.L., Gallego, S.M.: Modifications in catalase activity and expression in developing sunflower seedlings under cadmium stress. — Redox Rep. 13: 40–46, 2008.
Baum, G., Long, C., Jenkins, G.I., Trewavas, A.J.: Stimulation of the blue light phototropic receptor NPH1 causes a transient increase in cytosolic Ca2+. — Proc. nat. Acad. Sci. USA 96: 13554–13559, 1999.
Bradford, M.M.: A rapid and sensitive method for the quantification of microgram quantities of proteins utilizing the principle of protein-dye binding. — Anal. Biochem. 72: 248–254, 1976.
Causin, H.F., Barneix, A.J.: The role of oxidative metabolism in the regulation of leaf senescence by the light environment. — Int. J. Plant Develop. Biol. 1: 239–244, 2007.
Causin, H.F., Jauregui, R.N, Barneix, A.J.: The effect of light spectral quality on leaf senescence and oxidative stress in wheat. — Plant Sci. 171: 24–33, 2006.
Causin, H.F., Roberts, I.N., Criado, M.V., Gallego, S.M., Pena, L.B., Ríos, M. del C., Barneix, A.J.: Changes in hydrogen peroxide homeostasis and cytokinin levels contribute to the regulation of shade-induced senescence in wheat leaves. — Plant Sci. 177: 698–704, 2009.
Corpas, F.J., Barroso, J.B., Del Río, L.A.: Peroxisomes as a source of reactive oxygen species and nitric oxide signal molecules in plant cells. — Trends Plant Sci. 6: 145–150, 2001.
Demidchik, V., Bowen, H.C., Maathuis, F.J.M., Shabala, S.N., Tester, M.A., White, P.J., Davies, J.M.: Arabidopsis thaliana root non-selective cation channels mediate calcium uptake and are involved in growth. — Plant J. 32: 799–808, 2002
Dodd, A.N., Kudla, J., Sanders, D.: The language of calcium signaling. — Annu. Rev. Plant Biol. 61: 593–620, 2010.
Elzenga, J.T.M., Staal, M., Prins, H.B.A.: Calcium-calmodulin signalling is involved in light-induced acidification by epidermal leaf cells of pea, Pisum sativum L. — J. exp. Bot. 48: 2055–2060, 1997.
Folta, K.M., Lieg, E.J., Durham, T., Spalding, E.P.: Primary inhibition of hypocotyl growth and phototropism depend differently on phototropin-mediated increases in cytoplasmic calcium induced by blue light. — Plant Physiol. 133: 1464–1470, 2003.
Gepstein, S.: Leaf senescence — not just a ‘wear and tear’ phenomenon. — Genome Biol. 5: 212, 2004.
Guiamét, J.J., Willemoes, J.G., Montaldi, E.R.: Modulation of progressive leaf senescence by red:far-red ratio of incident light. — Bot. Gaz. 150: 148–151, 1989.
Guo, H., Mockler, T., Duong, H., Lin, C.: SUB1, an Arabidopsis Ca2+-binding protein involved in cryptochrome and phytochrome coaction. — Science 291: 487–490, 2001.
Huang, Y., Chen, C.T., Kao, C.H.: Senescence of rice leaves XXIV. Involvement of calcium and calmodulin in the regulation of senescence. — Plant Cell Physiol. 31: 1015–1520, 1990.
Huang, Y., Kao, C.H.: The importance of transmembrane flux of Ca2+ in regulating dark-induced senescence of detached corn leaves. — Bot. Bull. Acad. sin. 33: 17–21, 1992.
Jakoby, M., Weisshaar, B., Dröge-Laser, W., Vicente-Carbajosa, J., Tiedemann, J., Kroj, T., Parcy, F.: bZIP transcription factors in Arabidopsis. — Trends Plant Sci. 7: 106–111, 2002.
Jiang, M., Zhang, J.: Cross-talk between calcium and reactive oxygen species originated from NADPH oxidase in abscisic acid-induced antioxidant defense in leaves of maize seedlings. — Plant Cell Environ. 26: 929–939, 2003.
Kajimura, T., Mizuno, N., Takumi, S.: Utility of leaf senescence-associated gene homologs as developmental markers in common wheat. — Plant Physiol. Biochem. 48: 851–859, 2010.
Keskitalo, J., Bergquist, G., Gardeström, P., Jansson, S.: A cellular timetable of autumn senescence. — Plant Physiol. 139: 1635–1648, 2005.
Lim, P.O., Kim, H.J., Nam, H.G.: Leaf senescence. — Annu. Rev. Plant Biol. 58: 115–136, 2007.
Love, A.J., Milner, J.L., Sadanandom, A.: Timing is everything: regulatory overlap in plant cell death. — Trends Plant Sci. 13: 589–595, 2008.
Luna, C.M., Pastori, G.M., Driscoll, S., Groten, K., Bernard, S., Foyer, C.H.: Drought controls on H2O2 accumulation, catalase (CAT) activity and CAT gene expression in wheat. — J. exp. Bot. 56: 417–423, 2004.
Mhamdi, A., Noctor, G., Baker, A.: Plant catalases: peroxisomal redox guardians. — Arch.Biochem. Biophys. 525: 181–194, 2012.
Noctor, G., Veljovic-Jovanovic, S., Foyer, C.: Peroxide processing in photosynthesis: antioxidant coupling and redox signalling. — Phil. trans. roy. Soc. London B 355: 1465–1475, 2000.
Orendi, G., Zimmermann, P., Baar, C., Zentgraf, U.: Loss of stress-induced expression of catalase 3 during leaf senescence in Arabidopsis thaliana is restricted to oxidative stress. — Plant Sci. 161: 301–314, 2001.
Pena, L.B., Azpilicueta, C.E., Gallego, S.M.: Sunflower cotyledons cope with copper stress by inducing catalase subunits less sensitive to oxidation. — J. Trace Element med. Biol. 25: 125–129, 2011.
Polidoros, A.N., Scandalios, J.G.: Role of hydrogen peroxide and different classes of antioxidants in the regulation of catalase and glutathione S-transferase gene expression in maize (Zea mays L.). — Physiol. Plant. 106: 112–120, 1999.
Porra, R.J.: The chequered history of the development and use of simultaneous equations for the accurate determination of chlorophylls a and b. — Photosynth. Res. 73: 149–156, 2002.
Procházková, D., Sairam, R.K., Srivastava, G.C., Singh, D.V.: Oxidative stress and antioxidant activity as the basis of senescence in maize leaves. — Plant Sci. 161: 765–771, 2001.
Rouseaux, M.C., Hall, A.J., Sánchez, R.A.: Far-red enrichment and photosynthetically active radiation level influence leaf senescence in field-grown sunflower. — Physiol. Plant. 96: 217–224, 1996.
Rousseaux, M.C., Ballaré, C.L., Jordan, E.T., Vierstra, R.D.: Directed overexpression of PHYA locally suppresses stem elongation and leaf senescence responses to far-red radiation. — Plant Cell Environ. 20: 1551–1558, 1997.
Schmidt, M., Grief, J., Feierabend, J.: Mode of translational activation of the catalase (CAT1) mRNA of rye leaves (Secalecereale L.) and its control through blue light and reactive oxygen. — Planta 223: 835–846, 2006.
Shang, W., Feierabend, J.: Dependence of catalase photoinactivation in rye leaves in light intensity and quality and characterization of a chloroplast-mediated inactivation in red light. — Photosynth. Res. 59: 201–213, 1999.
Shang, Z., Ma, L., Zhang, H., He, R., Wang, X., Cui, S.: Ca2+ influx into lily pollen grains through a hyperpolarizationactivated Ca2+-permeable channel which can be regulated by extracellular CaM. — Plant Cell Physiol. 46: 598–608, 2005.
Shimazaki, K., Misumi, T., Ayako, S.: Inhibition of the stomatal blue light response by verapamil at high concentration. — Plant Cell Physiol. 38: 747–750, 1997.
Shinkle, J.R., Jones, R.J.: Inhibition of stem elongation in Cucumis seedlings by blue light requires calcium. — Plant Physiol. 86: 960–966, 1988.
Smykowski, A., Zimmermann, P., Zentgraf, U.: G-box Binding Factor 1 reduces CATALASE 2 expression and regulates the onset of leaf senescence in Arabidopsis. — Plant Physiol. 153: 1321–1331, 2010.
Srivalli, B., Khanna-Chopra, R.: Induction of new isoforms of superoxide dismutase and catalase enzymes in the flag leaf of wheat during monocarpic senescence. — Biochem. biophys. Res. Commun. 288: 1037–1042, 2001.
Stoelzle, S., Kagawa, T., Wada, M., Rainer, H., Dietrich, P.: Blue light activates calcium-permeable channels in Arabidopsis mesophyll cells via the phototropin signaling pathway. — Proc. nat. Acad. Sci. USA 100: 1456–1446, 2003.
Vandenabeele, S., Vanderauwera, S., Vuylsteke, M., Rombauts, S., Langebartels, C., Seidlitz, H.K., Zabeau, M., Van Montagu, M., Inzé, D., Van Breusegem, F.: Catalase deficiency drastically affects gene expression induced by high light in Arabidopsis thaliana. — Plant J. 39: 45–58, 2004.
Willekens, H., Chamnongpol, S., Davey, M., Schraudner, M., Langebartels, C., Van Montagu, M., Inzé, D., Van Camp, W.: Catalase is a sink for H2O2 and is indispensable for stress defence in C3 plants. — EMBO J. 16: 4806–4816, 1997.
Woodbury, W., Spencer, A.K., Stahmann, M.A.: An improved procedure using ferricyanide for detecting catalase isozymes. — Anal. Biochem. 44: 301–305, 1971.
Yang, T., Poovaiah, B.W.: Hydrogen peroxide homeostasis: activation of plant catalase by calcium/calmodulin. — Proc. nat. Acad. Sci. USA 99: 4097–5002, 2002.
Yang, W.L., Liu, J.M., Chen, F., Liu. Q., Gong, Y.D., Zhao, N.M.: Identification of Festuca arundinacea Schreb Cat1 catalase gene and analysis of its expression under abiotic stresses. — J. integr. Plant. Biol. 48: 334–340, 2006.
Yang, Z.Q., Li, Y.X., Jiang, X.D., Zhu, J., Zhang, J.B.: Effects of different red to far-red radiation ratios on the senescence of greenhouse Chrysanthemum leaves. — Afr. J. agr. Res. 7: 1919–1925, 2012.
Zhong, H.H., Resnick, A.S., Straume, M., Robertson McClung, C.: Effects of synergistic signaling by phytochrome A and cryptochrome1 on circadian clock-regulated catalase expression. — Plant Cell 9: 947–955, 1997.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgements: We thank Dr. Ezequiel Petrillo for his valuable help in the determination of TaSAG genes expression. The present work was supported by CONICET (PIP 11220080101142), and the University of Buenos Aires (UBACyT 20020100200214 and UBACyT 20020100100059).
Rights and permissions
About this article
Cite this article
Causin, H.F., Marchetti, C.F., Pena, L.B. et al. Down-regulation of catalase activity contributes to senescence induction in wheat leaves exposed to shading stress. Biol Plant 59, 154–162 (2015). https://doi.org/10.1007/s10535-014-0480-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-014-0480-z