Abstract
Objectives
The present study aimed to explore the eliciting effects of increasing concentrations (50, 100, and 200 µM) of methyl jasmonate (MeJA). We cultivated actively proliferating buds of Phoenix dactylifera L. cv. Barhee in a temporary immersion system and we monitored the bioactive compound accumulation after 7 days of culture.
Methods
Total phenolic (TPC) and flavonoid (TFC) contents were determined by high-performance liquid chromatography (HPLC), Fourier-transform infrared (FTIR), and radical scavenging activity using DPPH and ABTS assays. We also explored the activity of phenylpropanoid pathway enzymes, namely phenylalanine ammonia-lyase (PAL), tyrosine ammonia-lyase (TAL) and polyphenol oxidase (PPO).
Results
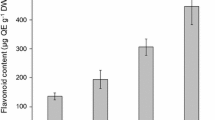
Our results revealed that MeJA treatment induced oxidative stress, and at the same time increased the activity of related defense enzymes in a dose-dependent manner. Exogenous application of MeJA at 200 µM increased ROS (two fold), hydrogen peroxide (3.7 fold), nitric oxide (14 fold), MDA (6.3 fold), superoxide dismutase (5.9 fold), catalase (4.4 fold) and guaiacol peroxidase (3.87 fold). Furthermore, the results demonstrated that 200 µM MeJA treatment enhanced the activities of PAL (3.65 fold), TAL (4.35 fold), PPO (threefold) and increased TPC (twofold) and TFC (1.75 fold) contents in buds cultures higher than the control. HPLC analysis showed that buds cultures exposed to 200 µM MeJA accumulated maximum amount of catechin (11 fold), 4-hydroxybenzoic acid (1.48 fold), caffeic acid (2.5 fold) and p-coumaric acid (1.76 fold) and demonstrate antioxidant capacity with the lowest DPPH (114.5 µg ml−1) and ABTS (90.2 µg ml−1) IC50 values on day 7 of culture as compared to the control. The MeJA in the culture medium directly reduced cell viability in a dose dependent manner up to 35% with the highest concentration.
Conclusion
The results of this study has revealed, for the first time, that MeJA offers a promising potential for the production of phenolic compound in Phoenix dactylifera L. buds.




Similar content being viewed by others
References
Abbas O, Compère G, Larondelle Y, Pompeu D, Rogez H, Baeten V (2017) Phenolic compound explorer: a mid-infrared spectroscopy database. Vib Spectrosc 92:111–118. https://doi.org/10.1016/j.vibspec.2017.05.008
Abbasi BH, Khan M, Guo B, Bokhari SA, Khan MA (2011) Efficient regeneration and antioxidative enzyme activities in Brassica rapa var. turnip. Plant Cell Tissue Organ Cult 105:337–344. https://doi.org/10.1007/s11240-010-9872-8
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Al-Khayri JM, Naik PM (2020) Elicitor-induced production of biomass and pharmaceutical phenolic compounds in cell suspension culture of date palm (Phoenix dactylifera L.). Molecules 25:46–69. https://doi.org/10.3390/molecules25204669
Anderson ME (1985) Determination of glutathione and glutathione disulfide biological samples. Meth Enzymol 113:548–555. https://doi.org/10.1016/S0076-6879(85)13073-9
Antoniou C, Savvides A, Georgiadou EC, Fotopoulos V (2018) Spectrophotometric quantification of reactive oxygen, nitrogen and sulfur species in plant samples. In: Alca´zar R, Tiburcio AF (eds) Methods in Molecular Biology. Springer, New York, pp 155–161. https://doi.org/10.1007/978-1-4939-7398-9_16
Ascacio-Valdés J, Buenrostro J, De la Cruz R, Sepúlveda L, Aguilera A, Prado A, Contreras JC, Rodríguez R, Aguilar CN (2014) Fungal biodegradation of pome-granate ellagitannins. J Basic Microbiol 54:28–34. https://doi.org/10.1002/jobm.201200278
Assis JS, Maldonado R, Munoz T, Escribano MI, Merodio C (2001) Effect of high carbon dioxide concentration on PAL activity and phenolic contents in ripening cherimoya fruit. Postharvest Biol Technol 23:33–39. https://doi.org/10.1016/S0925-5214(01)00100-4
Baenas N, García-Viguera C, Moreno DA (2014) Elicitation: a tool for enriching the bioactive composition of foods. Molecules 19(9):13541–13563. https://doi.org/10.3390/molecules190913541
Balasundram N, Sundram K, Samman S (2006) Phenolic compounds in plants and agri-industrial by-products: antioxidant activity, occurrence, and potential uses. Food Chem 99:191–203. https://doi.org/10.1016/j.foodchem.2005.07.042
Beaudoin-Eagan LD, Thorpe TA (1985) Tyrosine and phenylalanine ammonia lyase activities during shoot initiation in tobacco callus cultures. Plant Physiol 78(3):438–441
Belchí-Navarro S, Abellan RM, Pedreño MA, Almagro L (2019) Production and localization of hydrogen peroxide and nitric oxide in grapevine cells elicited with cyclodextrins and methyl jasmonate. J Plant Physiol 237:80–86. https://doi.org/10.1016/j.jplph.2019.03.013
Bouissil S, Alaoui-Talibi ZE, Pierre G, Rchid H, Michaud P, Delattre C, El Modafar C (2020) Fucoidans of moroccan brown seaweed as elicitors of natural defenses in date palm roots. Mar Drugs 18(12):596. https://doi.org/10.3390/md18120596
Brand-Williams W, Culivier ME, Berset C (1995) Use of a free radical method to evaluate antioxidant activity. LWT—Food Sci Technol 28:25–30. https://doi.org/10.1016/S0023-6438(95)80008-5
Cai X, Wei H, Liu C, Ren X, Thi LT, Jeong BR (2020) Synergistic Effect of NaCl pretreatment and PVP on browning suppression and callus induction from petal explants of Paeonia Lactiflora pall ‘Festival Maxima.’ Plants 9(3):346. https://doi.org/10.3390/plants9030346
Cheynier V (2012) Phenolic compounds: from plants to foods. Phytochem Rev 11:153–177. https://doi.org/10.1007/s11101-012-9242-8
De Carlo A, Tarraf W, Lambardi M, Benelli C (2021) Temporary immersion system for production of biomass and bioactive compounds from medicinal plants. Agronomy 11(12):2414. https://doi.org/10.3390/agronomy11122414b
Efferth T (2019) Biotechnology applications of plant callus cultures. Engineering 5:50–59. https://doi.org/10.1016/j.eng.2018.11.006
Fki L, Bouaziz N, Kriaa W, Benjemaa-Masmoudi R, Gargouri-Bouzid R, Rival A, Drira N (2011) Multiple bud cultures of ‘Barhee’ date palm (Phoenix dactylifera) and physiological status of regenerated plants. J Plant Physiol 168(14):1694–1700. https://doi.org/10.1016/j.jplph.2011.03.013
Gantait S, El-Dawayati MM, Panigrahi J, Labrooy C, Verma SK (2018) The retrospect and prospect of the applications of biotechnology in Phoenix dactylifera L. Appl Microbiol Biotechnol 19:8229–8259. https://doi.org/10.1007/s00253-018-9232-x
Gay CA, Gebicki JM (2000) A critical evaluation of the effect of sorbitol on the ferric-xylenol orange hydroperoxide assay. Anal Biochem 284(2):217–220. https://doi.org/10.1006/abio.2000.4696
Giannopolites CN, Ries SK (1977) Superoxide dismutase occurrence in higher plants. Plant Physiol 59(2):309–314. https://doi.org/10.1104/pp.59.2.309
Gros-Balthazard M, Flowers JM, Hazzouri KM, Ferrand S, Aberlenc F, Sallon S, Purugganan MD (2021) The genomes of ancient date palms germinated from 2,000 y old seeds. Proc Natl Acad Sci USA 118(19):e2025337118. https://doi.org/10.1073/pnas.2025337118
Ho TT, Lee JD, Ahn MS, Kim SW, Park SY (2018) Enhanced production of phenolic compounds in hairy root cultures of Polygonum multiflorum and its metabolite discrimination using HPLC and FT-IR methods. Appl Microbiol Biotechnol 102:9563–9575
Ho T-T, Murthy HN, Park SY (2020) Methyl jasmonate induced oxidative stress and accumulation of secondary metabolites in plant cell and organ cultures. Int J Mol Sci 21(3):716. https://doi.org/10.3390/ijms21030716
Jones AMP, Saxena PK (2013) Inhibition of phenylpropanoid biosynthesis in Artemisia annua L: a novel approach to reduce oxidative browning in plant tissue culture. PLoS ONE 8(10):e76802. https://doi.org/10.1371/journal.pone.0076802
Joshi DD (2012) FTIR spectroscopy: herbal drugs and fingerprints. In: Joshi DD (ed) Herbal drugs and fingerprints. Springer, India, pp 121–146. https://doi.org/10.1007/978-81-322-0804-4_7
Kolewe ME, Gaurav V, Roberts SC (2008) Pharmaceutically active natural product synthesis and supply via plant cell culture technology. Mol Pharm 5(2):243–256
Kuk YI, Shin JS, Burgos NR, Hwang TE, Han O, Cho BH, Jung S, Guh JO (2003) Antioxidative enzymes offer protection from chilling damage in rice plants. Crop Sci 43(6):2109–2117. https://doi.org/10.2135/cropsci2003.2109
Kwon YK, Ahn MS, Park JS, Liu JR, In DS, Min BW, Kim SW (2014) Discrimination of cultivation ages and cultivars of ginseng leaves using Fourier transform infrared spectroscopy combined with multivariate analysis. J Ginseng Res 38(1):52–58
Li ZG (2019) Measurement of signaling molecules calcium ion, reactive sulfur species, reactive carbonyl species, reactive nitrogen species, and reactive oxygen species in plants. In: Khan MR, Reddy PS, Ferrante A, Khan NA (eds) Plant signaling molecules: role and regulation under stressful environments. Elsevier, Cambridge, pp 83–103. https://doi.org/10.1016/B978-0-12-816451-8.00005-8
Manivannan A, Soundararajan P, Park YG, Jeong BR (2016) Chemical elicitor-induced modulation of antioxidant metabolism and enhancement of secondary metabolite accumulation in cell suspension cultures of Scrophularia kakudensis Franch. Int J Mo Sci 17(3):399. https://doi.org/10.3390/ijms17030399
Meloni DA, Oliva MA, Martinez CA, Cambraia J (2003) Photosynthesis and activity of superoxide dismutase, peroxidase and glutathione reductase in cotton under salt stress. Environ Exp Bot 49:69–76. https://doi.org/10.1016/S0098-8472(02)00058-8
Naik PM, Al-Khayri JM (2017) Extraction and estimation of secondary metabolites from date palm cell suspension cultures. Methods Mol Biol 1637:319–332. https://doi.org/10.1007/978-1-4939-7156-5_26
Naik PM, Al-Khayri JM (2020) Elicitor-induced production of biomass and pharmaceutical phenolic compounds in cell suspension culture of date palm (Phoenix dactylifera L.) extraction and estimation of secondary metabolites from date palm cell suspension cultures. Molecules 25:4669. https://doi.org/10.3390/molecules25204669
Nandy S, Hazra AK, Pandey DK, Ray P, Dey A (2021) Elicitation of industrially promising vanillin type aromatic compound 2-hydroxy 4-methoxy benzaldehyde (MBAlD) yield in the in-vitro raised medicinal crop Hemidesmus indicus (L) R. Br. by methyl jasmonate and salicylic acid. Ind Crops Prod 164:113–375. https://doi.org/10.1016/j.indcrop.2021.113375
Othmani A, Bayoudh C, Sellemi A, Drira N (2017) Temporary immersion system for date palm micropropagation. Methods Mol Biol 1637:239–249. https://doi.org/10.1007/978-1-4939-7156-5_20
Per TS, Khan MIR, Anjum NA, Masood A, Hussain SJ, Khan NA (2018) Jasmonates in plants under abiotic stresses: crosstalk with other phytohormones matters. Environ Exp Bot 145:104–120. https://doi.org/10.1016/j.envexpbot.2017.11.004
Pesaraklua A, Radjabiana A, Salamib SA (2021) Methyl jasmonate and Ag+ as effective elicitors for enhancement of phenolic acids contents in Salvia officinalis and Salvia verticillata, as two traditional medicinal plants. S Afr J Bot 141:105–115. https://doi.org/10.1016/j.sajb.2021.04.032
Putter J (1974) Peroxidase. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Verlag Chemie, Weinhan, pp 685–690
Re R, Pellegrini N, Proteggente A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med 26:1231–1237. https://doi.org/10.1016/S0891-5849(98)00315-3
Rybaczek D, Musialek MW, Balcerczyk A (2015) Caffeine-induced premature chromosome condensation results in the apoptosis-like programmed cell death in root meristems of Vicia faba. PLoS ONE 10(11):e0142307. https://doi.org/10.1371/journal.pone.0142307
Sachadyn-Król M, Agriopoulou S (2020) Ozonation as a method of abiotic elicitation improving the health-promoting properties of plant products—a review. Molecules 25(10):2416. https://doi.org/10.3390/molecules25102416
Sánchez-Sampedro MA, Fernández-Tárrago J, Corchete P (2005) Yeast extract and methyl jasmonate-induced silymarin production in cell cultures of Silybum marianum (L.) Gaertn. Journal Biotechnol 119(1):60–69
Soliva RC, Elez P, Sebastian M, Martin O (2001) Evaluation of browning effect on avocado puree preserved by combined methods. Innov Food Sci Emerg 1:261–268
Suzuki N, Mittler R (2006) Reactive oxygen species and temperature stresses: a delicate balance between signaling and destruction. Physiol Plant. https://doi.org/10.1111/j.0031-9317.2005.00582.x
Thakur M, Bhattacharya S, Khosla PK, Puri S (2019) Improving production of plant secondary metabolites through biotic and abiotic elicitation. J Appl Res Med Aromat Plants 12:1–12. https://doi.org/10.1016/j.jarmap.2018.11.004
Udomsin O, Yusakul G, Kitisripanya T, Juengwatanatrakul T, Putalun W (2020) The deoxymiroestrol and isoflavonoid production and their elicitation of cell suspension cultures of Pueraria candollei var. mirifica: from shake flask to bioreactor. Appl Biochem Biotechnol 190:57–72. https://doi.org/10.1007/s12010-019-03094-y
Veersham C (2004) Elicitation: medicinal plant biotechnology. CBS Publisher, India, pp 270–293
Venugopalan A, Srivastava S (2015) Endophytes as in vitro production platforms of high value plant secondary metabolites. Biotechnol Adv 33:873–887. https://doi.org/10.1016/j.biotechadv.2015.07.004
Włodek L (2002) Beneficial and harmful effects of thiols. Polish J Pharmacol 54(3):215–223
Zhishen J, Mengcheng T, Jianming W (1999) The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem 64:555–559. https://doi.org/10.1016/S0308-8146(98)00102-2
Acknowledgements
We gratefully acknowledge the support of Mr. Alain Rival (CIRAD, Jakarta-Indonesia) for his valuable help and Mr. Kamel Maaloul and Mr. Hatem Rebaii, professors of English at the University of Sfax, for their careful proofreading of the manuscript.
Funding
This research did not benefit from any specific grant or funding from any agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Amal Ben Romdhane, Lotfi Fki and Yassine Chtourou conceived and designed the research. Amal Ben Romdhane, Mohamed Maalej, Yassine Chtourou and Lotfi Fki conducted experiments, analyzed the data and drafted the manuscript. Haifa Sebii performed HPLC. Emna Baklouti, Ameni Nasri, Riadh Drira, Mohamed Maalej, Noureddine Drira and Alain Rival contributed to experiments related to the establishment of cell cultures. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ben Romdhane, A., Chtourou, Y., Sebii, H. et al. Methyl jasmonate induces oxidative/nitrosative stress and the accumulation of antioxidant metabolites in Phoenix dactylifera L.. Biotechnol Lett 44, 1323–1336 (2022). https://doi.org/10.1007/s10529-022-03299-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-022-03299-y