Abstract
Objective
To develop a cost-effective, non-toxic and xeno-free freezing solution for the preservation of adipose tissue-derived stem cells (hADSC) with a long shelf-life.
Results
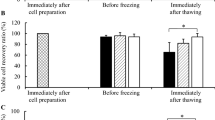
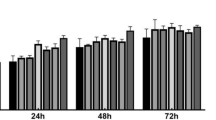
The potential of various hydrocolloids and organic osmolytes as cryoprotectants and individual components of phosphate buffered saline (PBS) as carrier media were evaluated to formulate a freezing solution for the cryopreservation of hADSCs. Among the hydrocolloids, the highest viability, 55 %, was achieved with post-thawed (after 48 h storage at −80 °C) hADSCs cryopreserved in 10 % (v/v) polyvinylpyrrolidone (PVP) using PBS as carrier media. 0.9 % NaCl was a superior carrier medium resulting an enhanced cell viability (70 %) when used in 10 % PVP than other components of PBS. A higher cell viability (81 %) was achieved when 10 % PVP/0.9 % NaCl was supplemented with 60 mM ectoin. The cryopreserved cells retained normal cytoskeletal distribution pattern and adipogenic and osteogenic differentiation ability during 14 and 21 days of incubation.
Conclusion
A serum-free and non-toxic 10 % PVP/0.9 % NaCl/60 mM ectoin freezing solution was developed for cryopreservation of hADSC for application in tissue engineering and regenerative medicine.






Similar content being viewed by others
References
Baust J, Baust J (2007) Advances in biopreservation. CRC/Taylor & Francis, Boca Raton
Bissoyi A, Pramanik K (2014) Role of the apoptosis pathway in cryopreservation-induced cell death in mesenchymal stem cells derived from umbilical cord blood. Biopreserv Biobank 12:246–254
Bunnell B, Flaat M, Gagliardi C et al (2008) Adipose-derived stem cells: isolation, expansion and differentiation. Methods 45:115–120
Freimark D, Sehl C, Weber C et al (2011) Systematic parameter optimization of a DMSO- and serum-free cryopreservation protocol for human mesenchymal stem cells. Cryobiology 63:67–75
Gimble J, Katz A, Bunnell B (2007) Adipose-derived stem cells for regenerative medicine. Circ Res 100:1249–1260
Grein TA, Freimark D, Weber C et al (2010) Alternatives to dimethylsulfoxide for serum-free cryopreservation of human mesenchymal stem cells. Int J Artif Organs 33:370–380
Hubálek Z (2003) Protectants used in the cryopreservation of microorganisms. Cryobiology 46:205–229
Khudyakov A, Polezhaeva T, Zaitseva O et al (2015) The cryoprotectant effect of polysaccharides from plants and microalgae on human white blood cells. Biopreserv Biobank 13:240–246
Liu B, McGrath J (2005) Response of cytoskeleton of murine osteoblast cultures to two-step freezing. Acta Biochim Biophys Sinica 37:814–818
Nair B (1998) Final report on the safety assessment of polyvinylpyrrolidone (PVP). Int J Toxicol 17:95–130
Pikuła M, Marek-Trzonkowska N, Wardowska A et al (2013) Adipose tissue-derived stem cells in clinical applications. Expert Opin Biol Ther 13:1357–1370
Soma P, Williams P, Lo Y (2009) Advancements in non-starch polysaccharides research for frozen foods and microencapsulation of probiotics. Front Chem Eng China 3:413–426
Sonnemann U, Möller M, Bilstein A (2014) Noninterventional open-label trial investigating the efficacy and safety of ectoine containing nasal spray in comparison with beclomethasone nasal spray in patients with allergic rhinitis. J Allergy (Cairo) 2014:1–12
Sun H, Hofmann N, Glasmacher B (2009) Biomolecules for the improvement of cryopreservation of human endothelial cells. Cryobiology 59:397
Sun H, Wolfes H, Glasmacher B, Hofmann N (2011) 107. l-Proline and ectoine stabilize proteins from denaturation. Cryobiology 63:335
Szurek EA, Eroglu A (2011) Comparison and avoidance of toxicity of penetrating cryoprotectants. PLoS ONE 6:e27604
Thirumala S, Wu X, Gimble J, Devireddy R (2010a) Evaluation of polyvinylpyrrolidone as a cryoprotectant for adipose tissue-derived adult stem cells. Tissue Eng Part C Methods 16:783–792
Thirumala S, Gimble J, Devireddy R (2010b) Evaluation of methylcellulose and dimethyl sulfoxide as the cryoprotectants in a serum-free freezing media for cryopreservation of adipose-derived adult stem cells. Stem Cells Dev 19:513–522
Windrum P, Morris T, Drake M et al (2005) Variation in dimethyl sulfoxide use in stem cell transplantation: a survey of EBMT centers. Bone Marrow Transpl 36:601–603
Yancey P (2005) Organic osmolytes as compatible, metabolic and counteracting cytoprotectants in high osmolarity and other stresses. Exp Biol J 208:2819–2830
Acknowledgments
The authors acknowledge to the Department of Biotechnology (DBT) and Department of Science and Technology, Ministry of Science and Technology, Government of India for providing research facility by sanctioning Program Support on Tissue Engineering Research (sanction no. BT/01/COE/09/1309062010) and research project (sanction no. SR/FTP/ETA-0106/2011) respectively.
Supporting Information
Supplementary Fig. 1—Phase contrast microscopic images shows a round cell morphology within 8 h of culture in DMEM containing 10 % FBS (a) and cell proliferation on 6th and 12th day (b, c). The cells reached confluency on 12th day of culture (scale bar 100 µm).
Supplementary Fig. 2—Flowcytometric analysis showing the expression of positive CD90 (99 %), CD73 (89 %), and CD105 (98 %) markers and negative HLA-DR (0.5 %), CD34 (1.2 %) and CD45 (2 %) markers representing the cells are mesenchymal stem cells.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare there is no conflict of interest.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
10529_2016_2111_MOESM1_ESM.tif
Supplementary material 1 (TIF 62 kb) Supplementary Fig 1. Phase contrast microscopic images shows a round cell morphology within 8 h of culture in DMEM containing 10% FBS (a) and cell proliferation on 6th and 12th day (b, c). The cells reached confluency on 12th day of culture. (Scale bar 100 µm).
Below is the link to the electronic supplementary material.
10529_2016_2111_MOESM2_ESM.tif
Supplementary material 1 (TIF 278 kb) Supplementary Fig 2. Flowcytometric analysis showing the expression of positive CD90 (99%), CD73 (89%), and CD105 (98%) markers and negative HLA-DR (0.5%), CD34 (1.2%) and CD45 (2%) markers representing the cells are mesenchymal stem cells.
Rights and permissions
About this article
Cite this article
Ray, S.S., Pramanik, K., Sarangi, S.K. et al. Serum-free non-toxic freezing solution for cryopreservation of human adipose tissue-derived mesenchymal stem cells. Biotechnol Lett 38, 1397–1404 (2016). https://doi.org/10.1007/s10529-016-2111-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-016-2111-6